Restriction Mapping Southern Blotting Made Simple Class instructions

Restriction Mapping & Southern Blotting Made Simple Class instructions

In this lesson • • • Importance of complementary base pairing Importance of 5’ and 3’ ends of DNA Electrophoresis separates by size Blotting keeps the DNA in the same position The bands visible on the blot are the labeled ones but other DNA bands are still present Southern blotting can be used to find exact matches or similar matches Dye labels are used instead of radioisotopes due to safety considerations How to interpret restriction maps and Southern blots How restriction enzymes produce sticky ends
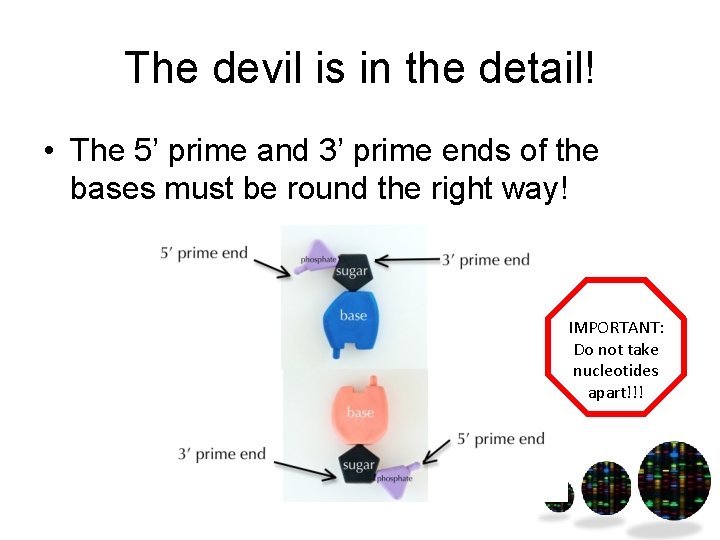
The devil is in the detail! • The 5’ prime and 3’ prime ends of the bases must be round the right way! IMPORTANT: Do not take nucleotides apart!!!

Correct base pairing is critical! • Green (Guanine) pairs • Blue (Adenine) pairs with yellow (Cytosine) with orange (Thymine)
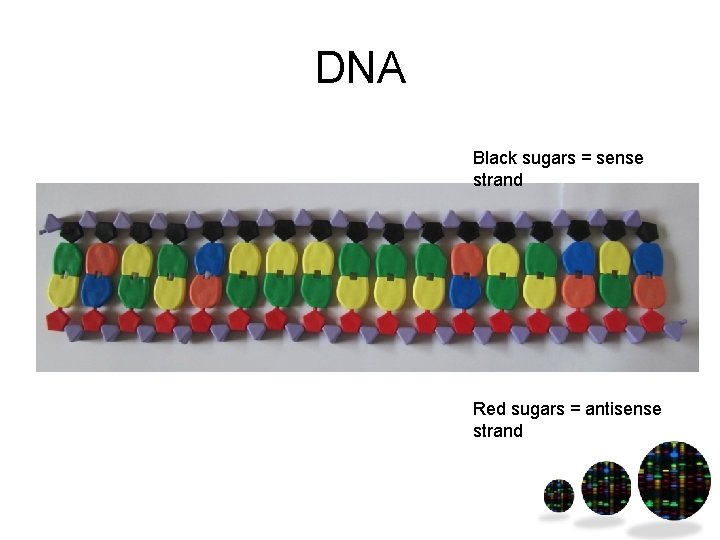
DNA Black sugars = sense strand Red sugars = antisense strand
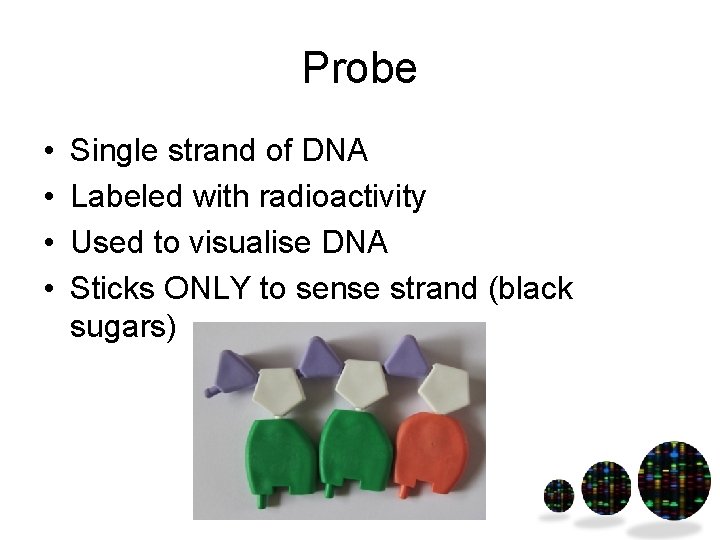
Probe • • Single strand of DNA Labeled with radioactivity Used to visualise DNA Sticks ONLY to sense strand (black sugars)

The activity • Divide class into three groups - each group gets a double stranded section of DNA • Group 1 digest DNA with Taq 1 • Group 2 digest DNA with Sma 1 • Group 3 digest DNA with Taq 1 and Sma 1

The activity overview • Cut the template with enzyme and separate the DNA at these sites leaving sticky ends. • How many fragments do you get and how big are they (measure the black sense strand only)? • Separate the fragments using DNA gel electrophoresis. Put fragments in size order (measure the black strand) from largest to smallest as in a DNA gel. • Blot the DNA bands onto a membrane. Denature the DNA so it is single stranded by removing the red antisense strands from the bands leaving the black sense strands. • DNA is blotted onto a nitrocellulose or nylon membrane so you have a replica of the positions of the DNA in the gel preserved on the membrane. • Hybridise the DNA probe. In our example, the probe binds to one region. The probe has white sugars. Remember to keep the 5’ to 3’ prime orientation the correct way round. • Visualise the band. Probe is labeled so it can be visualised after hybridisation. Which band does the probe bind to?

Restriction Digest Sites Taq 1 5’ - T C G A - 3' 3’ - A G C T - 5’ Sma 1 5’ - C C C 3’ - G G G - 3' C C C - 5’
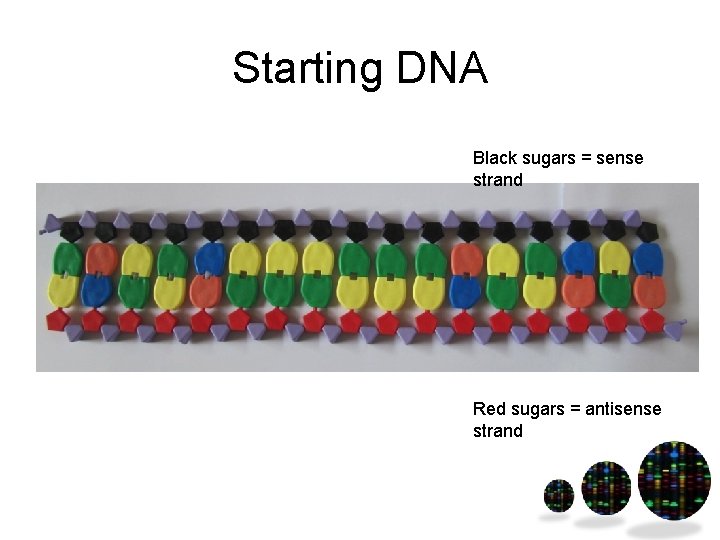
Starting DNA Black sugars = sense strand Red sugars = antisense strand

Taq 1 digest Result after electrophoresis and denaturation Blot probe is visible

Sma 1 digest Result after electrophoresis and denaturation Blot probe is visible

Taq 1& Sma 1 double digest Result after electrophoresis and denaturation probe is visible
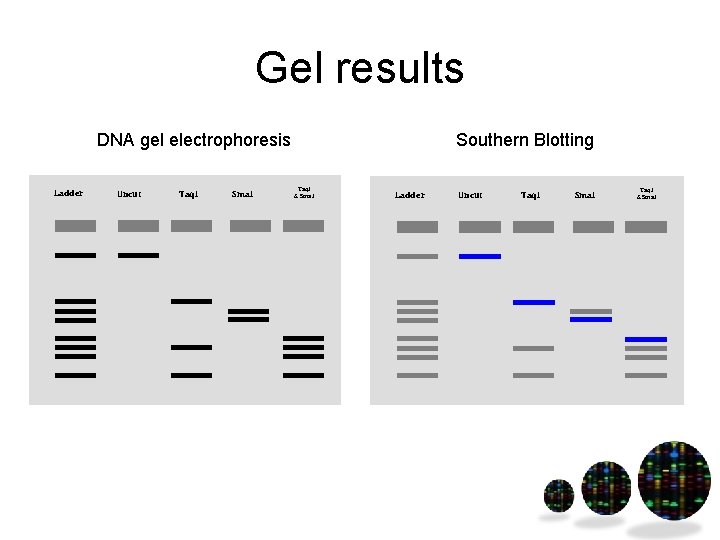
Gel results DNA gel electrophoresis Ladder Uncut Taq. I Sma. I Southern Blotting Taq. I & Sma. I Ladder Uncut Taq. I Sma. I Taq. I & Sma. I

Restriction mapping • Find restriction enzyme sites in DNA • Combine single & double digest results • Use to localize where the region of interest is on a piece of DNA (such as a genome) Hint – start with Sma. I, then double digest and use Taq. I result to put them in the correct order
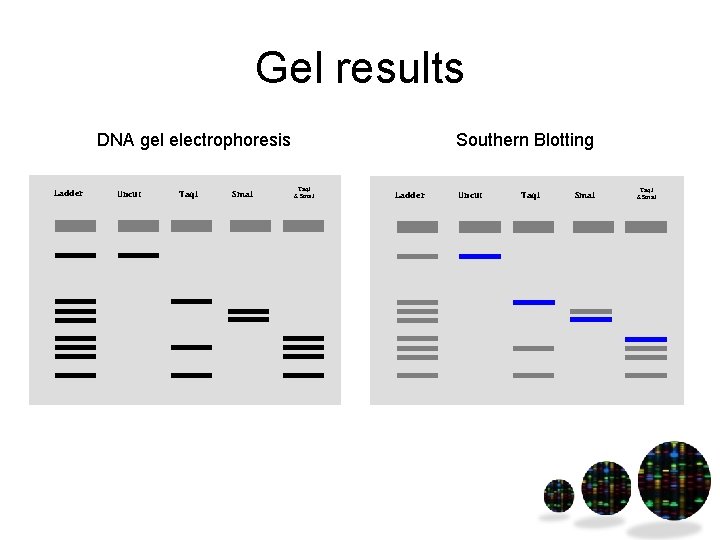
Gel results DNA gel electrophoresis Ladder Uncut Taq. I Sma. I Southern Blotting Taq. I & Sma. I Ladder Uncut Taq. I Sma. I Taq. I & Sma. I

Restriction Mapping Uncut Taq. I digest Sma. I digest Taq. I and Sma. I digest
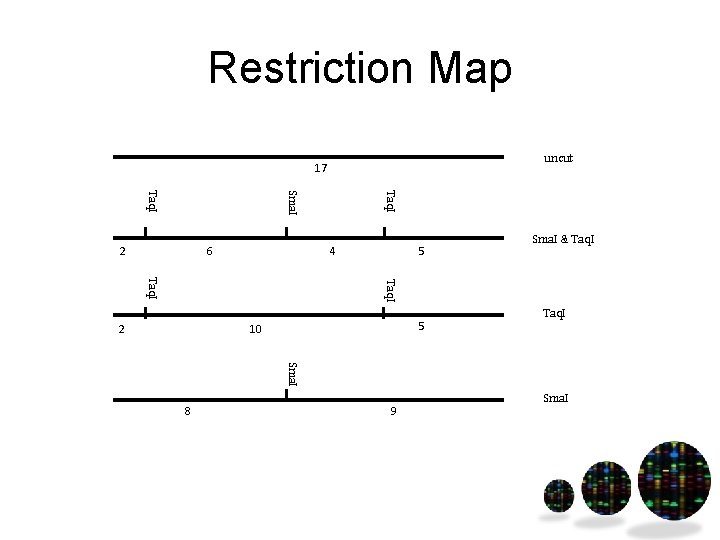
Restriction Map uncut 17 6 Taq. I Sma. I Taq. I 2 4 5 Sma. I & Taq. I 2 5 10 Taq. I Sma. I 8 9 Sma. I
- Slides: 18