Responsible Conduct of Research RCR Farida Lada Associate

Responsible Conduct of Research (RCR) Farida Lada Associate University Provost for Research Administration & Compliance October 3, 2017 http: //www. cuny. edu/research/compliance. html 1

Overview Rules • Federal regulations & State laws • Sponsor requirements • Institutional policies & procedures Ethics & Good Practice • International, national and institutional standards • Standards of relevant research community 2

Image borrowed from: http: //undsci. berkeley. edu/article/socialsideofscience_06 3

Research Misconduct - US Fabrication • Making up data or results, and recording or reporting them Falsification • Manipulating research materials, equipment or processes, or changing or omitting data or results, such that the research is not accurately represented in the research record Plagiarism • Appropriation of another person’s ideas, processes, results or words without giving appropriate credit 4
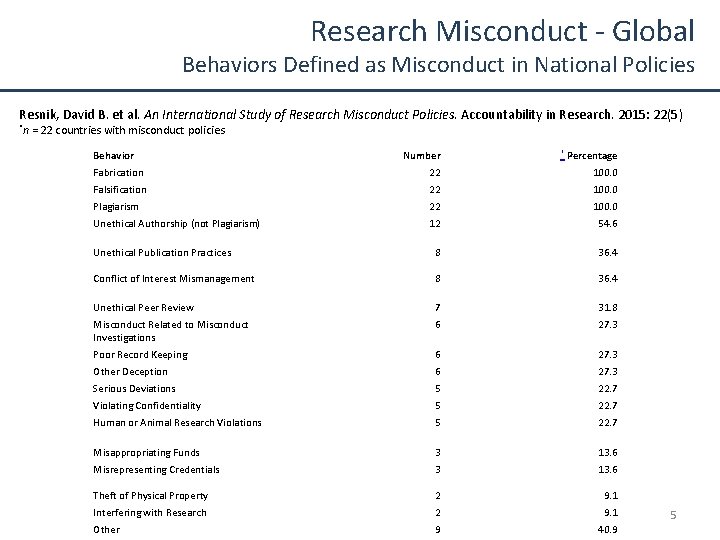
Research Misconduct - Global Behaviors Defined as Misconduct in National Policies Resnik, David B. et al. An International Study of Research Misconduct Policies. Accountability in Research. 2015: 22(5) *n = 22 countries with misconduct policies Behavior Number * Percentage Fabrication 22 100. 0 Falsification 22 100. 0 Plagiarism 22 100. 0 Unethical Authorship (not Plagiarism) 12 54. 6 Unethical Publication Practices 8 36. 4 Conflict of Interest Mismanagement 8 36. 4 Unethical Peer Review 7 31. 8 Misconduct Related to Misconduct Investigations 6 27. 3 Poor Record Keeping 6 27. 3 Other Deception 6 27. 3 Serious Deviations 5 22. 7 Violating Confidentiality 5 22. 7 Human or Animal Research Violations 5 22. 7 Misappropriating Funds 3 13. 6 Misrepresenting Credentials 3 13. 6 Theft of Physical Property 2 9. 1 Interfering with Research 2 9. 1 Other 9 40. 9 5

Research Misconduct – Does it Happen? 2014: NSF Case # A 13050061 Plagiarism: NSF found that the PI knowingly plagiarized 153 lines & 42 embedded references into an NSF proposal Outcome: PI must: – Comply with requirements imposed by the PI’s University (extensive) – Complete comprehensive RCR training in interactive format – Submit certifications that any proposals or reports submitted by the PI to NSF do not contain plagiarized, falsified or fabricated materials for 3 years – Submit assurances by a responsible official of the employer that any proposals or reports submitted by the PI to NSF do not contain plagiarized, falsified or fabricated material for 3 years http: //www. nsf. gov/oig/search/A 13050061. pdf 6

Research Misconduct – Does it Happen? 2015: ORI Case – Venkata J. Reddy, U of Minnesota Falsification / Fabrications: ORI found preponderance of evidence that Reddy intentionally & knowingly falsified and/or fabricated data provided to his mentor to include in an NIH grant. Outcome: Reddy: – Debarred for federal funding for a period of 5 years – Prohibited from serving in any advisory capacity (peer review, advisory committee, consultant) to PHS https: //ori. hhs. gov/content/case-summary-reddy-venkata-j 7

Research Misconduct – Does it Happen? 2010: ORI Case - Sezen, Bengu (former graduate student at Columbia University); research supported by NIH Falsification, fabrication & plagiarism: ORI made 21 findings of research misconduct including fabrication, falsification and plagiarism across 3 papers and doctoral thesis. Outcome: ORI took administrative action that for 5 years, i) Dr. Sezen is debarred from eligibility for any contracting or subcontracting with any agency of the United States Government and from eligibility or involvement in nonprocurement programs of the United States Government; ii) Dr. Sezen is barred from service in any advisory capacity to the PHS. http: //ori. hhs. gov/content/case-summary-sezen-bengu 8

Research Misconduct - Case Summaries • US Department of Health & Human Services Office of Research Integrity http: //ori. dhhs. gov/case_summary • National Science Foundation http: //www. nsf. gov/oig/closeouts. jsp 9

Research Misconduct Causes of research misconduct • • Publish or perish pressures Need to obtain research funding Seeking promotion Reputational motivations 10

Research Misconduct at CUNY • CUNY policy for disposition of allegations of research misconduct • CUNY policy on training in responsible conduct of research • Research Integrity Officers http: //www. cuny. edu/research/compliance/Responsible-Conduct-of. Research. html 11

RCR Scenario – Self Plagiarism 12

RCR Scenario – Making an Error 13
14

Conflict of Interest • Financial Conflict of Interest • CUNY policy, procedures and disclosure forms at http: //www. cuny. edu/research/compliance/conflictofinte restpolicy. html • Conflict of Commitment • Relationships with individuals or entities • Use of resources / funds • Honor time commitments made • College Conflicts Officers • CUNY Conflicts Committee 15

16

Protection of Human Subjects • HRPP / IRB review is required when: • Activity involves research (systematic investigation for generalizable knowledge); and • Activity involves human subjects (living individual about whom data is collected through intervention or interaction; or private identifiable information is collected); and • CUNY is engaged • CUNY Human Research Protection Program (HRPP) • HRPP Coordinators / IRB Administrators (Research Compliance Administrators) • University Integrated (UI) Institutional Review Boards (IRB) • http: //www. cuny. edu/research/compliance/human-subjectsresearch-1. html 17

18

Animal Welfare • Humane treatment of animals used in research and educational activities • Institutional Animal Care and Use Committees (IACUC) • Veterinarians, animal facilities staff, & IACUC administrators 19

20

Biosafety • Environmental Health, Safety & Risk Management • Policies and procedures regarding laboratory safety, hazardous materials, health safety & radiation safety http: //www. cuny. edu/about/administration/offices/ehsr m. html • Responsible for training, oversight and audits • College specific Institutional Biosafety Committees (IBC) • Recombinant DNA & gene therapy research • Risk group 2 or higher infectious agent use • Use of transgenic animals 21

Biosafety • Other biosafety categories to be aware of: • Select agents & toxins: biological agents and toxins that are identified as having the potential to pose a severe threat to public health and safety, animal health and safety, plant health and safety, or to the safety of animal or plant products • Dual use research of concern (DURC): life sciences research that, based on current understanding, can be reasonably anticipated to provide knowledge, information, products, or technologies that could be directly misapplied to pose a significant threat with broad potential consequences to public health and safety, agricultural crops and other plants, animals, the environment, materiel or national security. 22

Image borrowed from: http: //www. dhra. mil/perserec/osg/counterintelligence/espionage-indicators. htm 23

Export Control • Controls • State, Commerce & Treasury Controls • Hardware, software, materials, equipment, technology & technological data that have civilian and inherent military or defense application • Defense articles designed or modified for defense or military application without civil equivalent and defense services. • Restricted transactions; for example, those with Cuba, Iran, Syria or Sudan • CUNY policy, procedures, guidance & forms • http: //www. cuny. edu/research/compliance/Export. Control. html • Export Control Administrators 24

Export Control • Requirements • Prior authorization or license may be required • Screening of all parties involved • Exceptions for fundamental research activities • No publication or citizenship restrictions accepted 25

RCR Scenario – Sensitive Data Collection 26

27

Mentor / Trainee Responsibilities • • • Proactively set expectations Clarify evaluation criteria Clear distribution of responsibilities Standard operating procedures Criteria for establishing authorship & ownership Awareness of and compliance with institutional requirements 28

Peer Review • Responsibility • Assess project for quality • Make judgment regarding importance of research being proposed • Responsible Conduct • Timely review • Constructive feedback • Free from personal bias • Maintain confidentiality 29

Responsible Authorship • Authors • Made significant contributions to the research • Assumed responsibility for data collection & analysis • Participated in drafting the publication • Approved the final publication 30

Responsible Authorship • Inappropriate Authorship • Honorary authorship: “…instances of listing a senior colleague or a chairperson, providing facilities and technical support without contributing creatively or playing an insignificant role…” • Gift authorship: “…a senior or a junior colleague’s name is added as a gesture of amicable relations, or in an attempt to boost his/her profile or to receive a similar ‘gift’ in response. ” • Ghost authorship: “. . . misconduct related to the omission of a substantive contributor’s name. . . ” • Prevalence • Gasparyan et al conducted a survey of 360 corresponding authors in Annals of Internal Medicine, JAMA, The Lancet, Nature Medicine, The New England Journal of Medicine & PLo. S Medicine: estimated the prevalence of both honorary and ghost authorship at Gasparyan, A. , Ayvazyan, L. & Kitas, G. Authorship problems in scholarly journals: considerations for authors, peer reviewers and editors. Rheumatol Intl (2013) 33: 277 -284. 31

Authorship Causes of inappropriate authorship • False expectations, unclear arrangements or poor communication • Publish or perish pressures • Power differentials Authorship has impact on: • • Obtaining research funds Promotions Selection of reviewers and editors Demotivating effects Gasparyan, A. , Ayvazyan, L. & Kitas, G. Authorship problems in scholarly journals: considerations for authors, peer reviewers and editors. Rheumatol Intl (2013) 33: 277 -284. Swiss Academies of Arts and Sciences. Authorship in scientific publications: analysis and recommendations. Swiss Medical Weekly (2015) 32

Data Management • Data Ownership • Funding agency – grants vs. contracts • Institutional policy • CUNY Intellectual Property Policy: http: //www. cuny. edu/about/administration/offices/la/intellectual_pr operty_9. 20. 11. pdf • Data Source • Data Collection • Methods are appropriate for type of research • Data recorded such that it can be validated • Appropriate authorization • Human subjects; animal subjects; hazardous material & biological agent use; proprietary data; copyrighted or patented materials 33

Data Management • Data Protection • Proper storage to avoid accidental damage, loss or theft • Confidentiality & privacy agreements honored • Data retention according to contract &/or institutional practice • CUNY use of computer resources policy: http: //www. cuny. edu/about/administration/offices/CIS/policies/Compute r. Use. Policy. pdf • Data Sharing • We may enter into various types of data sharing agreements (data use, data transfer, etc. ) http: //www. cuny. edu/research/compliance/Research-Agreements. html 34

Collaborative Research • Roles & Relationships • Define each collaborator’s role in the project in a written agreement • Ensure common understanding of the research goals • Be familiar with each institution’s intellectual property & ownership policies • Management Plan • Financial Management: ensure that all collaborators are in compliance with funding agency’s financial management rules • Training & Supervision: address training & supervision of all research team members & be familiar with institutional requirements • Compliance: address how compliance with regulatory & institutional requirements will be ensured 35

Collaborative Research • Formal Agreements • Contact research compliance staff prior to signing these • Ensure that your collaboration is documented in a formal Agreement • Examples include: Memorandum of Understanding; Memorandum of Agreement; Material Transfer Agreement; Data Transfer Agreement • Signatory authority defined by policy: http: //www. cuny. edu//research-policies/Signatory. Authority. html 36

RCR Scenario – Competing Interests 37

RCR Scenario – Collaborative Research 38

RCR Scenario – Data Management 39
- Slides: 39