RESPIRATORY GAS TRANSPORT Biochemistry Departement Medical Faculty Of

RESPIRATORY GAS TRANSPORT Biochemistry Departement Medical Faculty Of Andalas University Padang
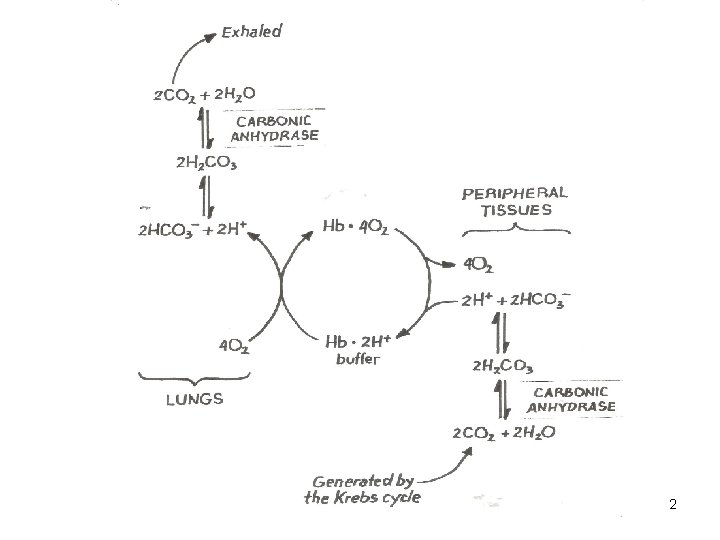
2

Oxygen Transport 3

Total Body Oxygen Stores • Oxygen in the Lung (~500 ml O 2). • Oxygen in the Blood (~850 ml O 2). • Oxygen in the Cells (very little except Mb-bound).
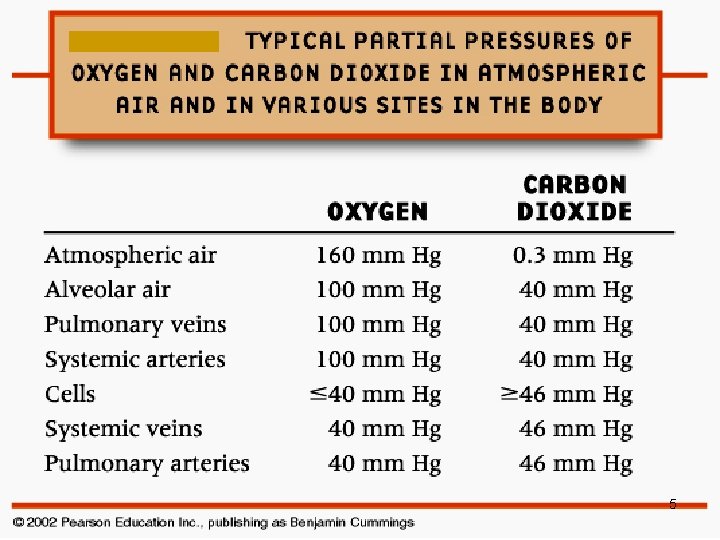
5
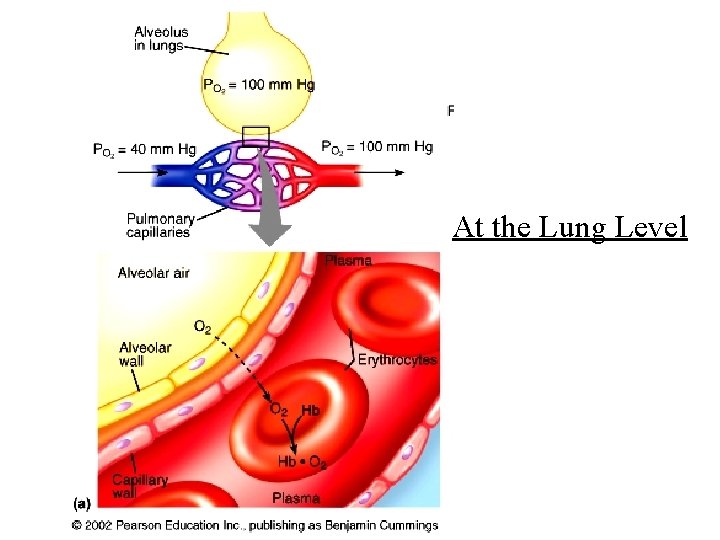
At the Lung Level
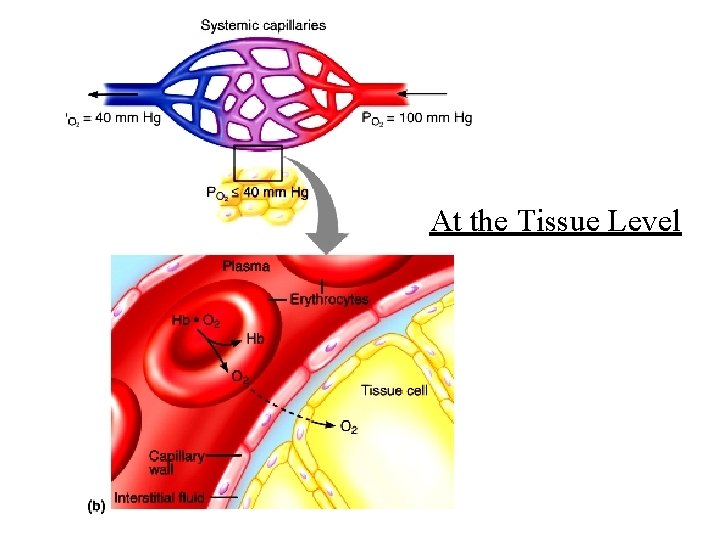
At the Tissue Level

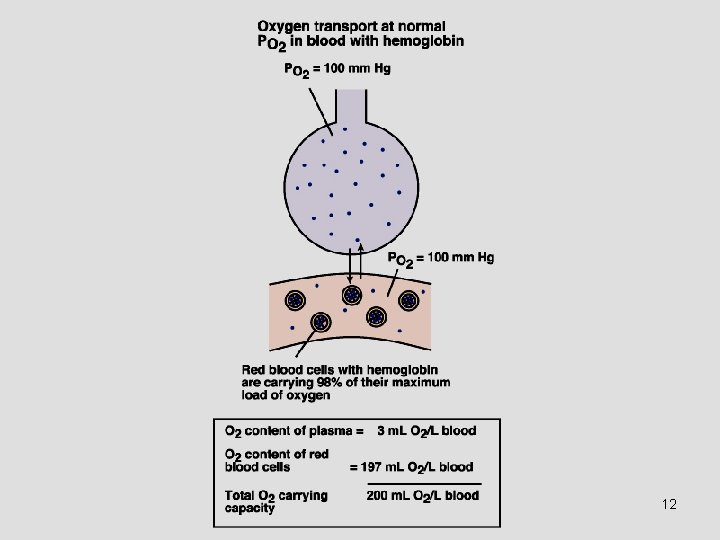
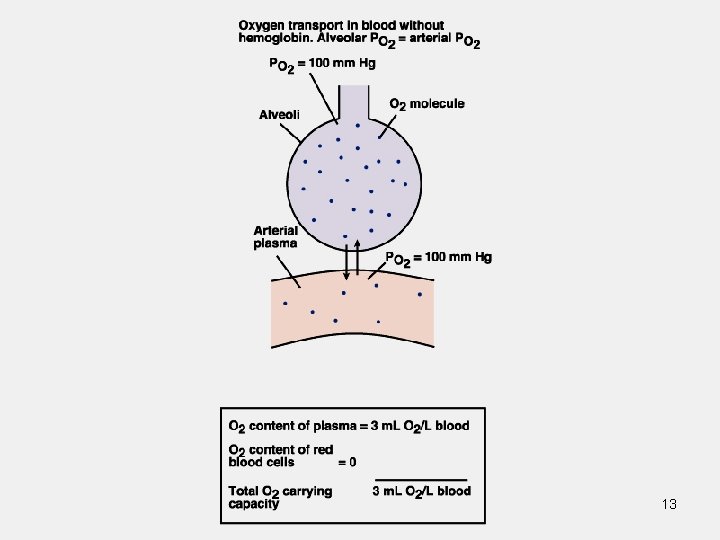
Oxygen Is Carried in Blood in 2 Forms • Bound to hemoglobin in red blood cells. • Dissolved in plasma. Normally insignificant.

Hemoglobin • Each “heme” molecule is capable of binding with 1 O 2 molecule and each “globin” molecule is capable of binding with 1 CO 2 molecule. • So, each molecule of Hb can bind to either 4 molecules of O 2 and 1 molecule of CO 2 • 100 ml of blood has about 15 gm of Hb, at Hct = 0. 45 9

• Binding of O 2 to 4 heme sites given by: Equilibrium constants for different reactions different Binding of first O 2 relatively low affinity 2 nd, 3 rd and 4 th - much higher affinity 10

Oxygen as Oxyhemoglobin • Each gram of Hb can store about 1. 34 ml of O 2: • 1 L of blood (150 gm of Hb) can store about 208 ml of O 2 Oxygen Capacity of Hb. • With normal cardiac output, about 1040 ml of O 2 can be carried in blood per minute. (4 times of the metabolic demands). 11
12
13

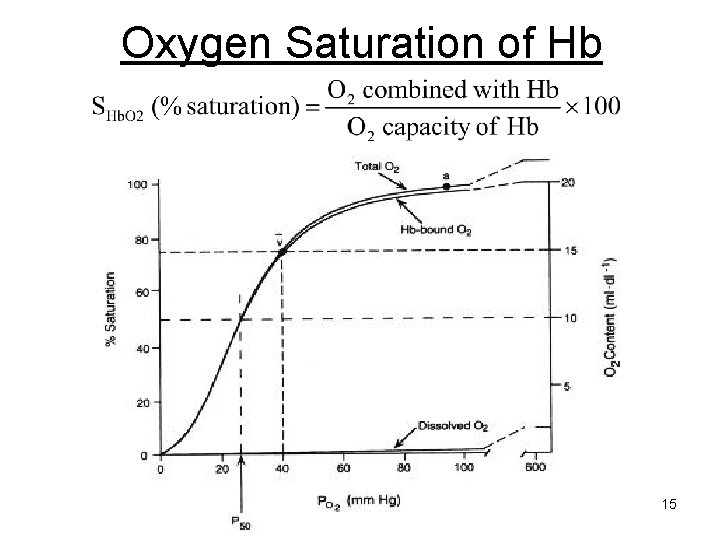
O 2 Saturation. • Units: percent. • Fraction or percentage of all the hemoglobin binding sites that are currently occupied by oxygen.
Oxygen Saturation of Hb 15

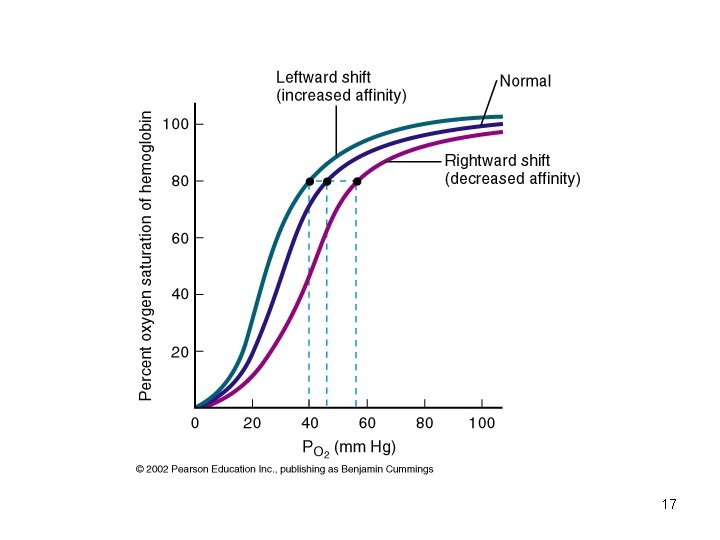
Four (5 -6? ) Things Change Oxyhemoglobin Affinity 1. • • • Hydrogen Ion Concentration, [H+] Carbon Dioxide Partial Pressure, PCO 2 Temperature [2, 3 -DPG] • • Special Case: Carbon Monoxide Hemoglobin variants
17

Factors Affecting Hb-O 2 Affinity: Summary • Hydrogen Ion: – Increased H+ (decreased p. H) increases H+ binding to Hb and reduces O 2 affinity (Hb. O 2+H+ Hb. H++O 2). • Carbon Dioxide (Bohr effect): – Increased PCO 2 increases CO 2 binding to Hb and reduces O 2 affinity (increased O 2 delivery to tissue). – Increased PCO 2 increases H+ and reduces O 2 affinity (fixed acid Bohr effect). • Temperature and 2, 3 -DPG (diphosphoglycerate): 18 – Increased temperature and 2, 3 -DPG reduces O 2 affinity.
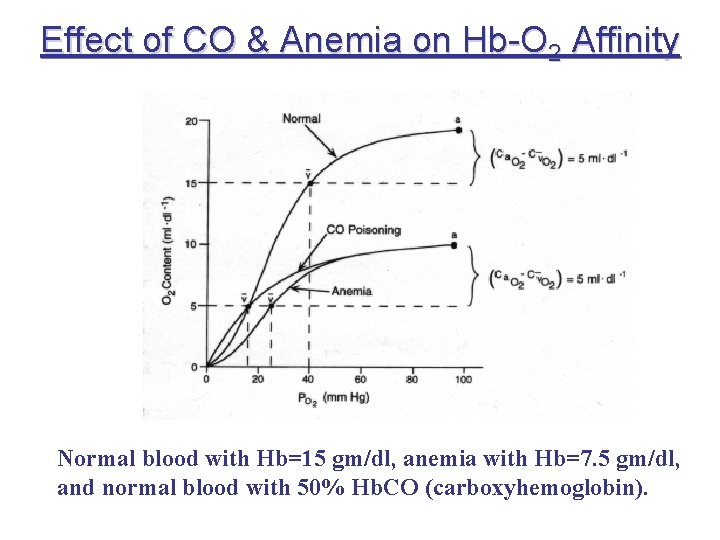
Effect of CO & Anemia on Hb-O 2 Affinity Normal blood with Hb=15 gm/dl, anemia with Hb=7. 5 gm/dl, and normal blood with 50% Hb. CO (carboxyhemoglobin).

Exercise • Increase temperature • Increased PCO 2 and • Decreased p. H (acidosis)

2, 3 -DPG • 2, 3 -DPG is a glycolytic intermediate – accumulates to uniquely high levels in RBCs -Increased 2, 3 -DPG right shift -Decreased 2, 3 -DPG left shift • Increased 2, 3 -DPG associated with hypoxia.

Conditions with Increased 2, 3 -DPG • • acclimatization to high altitudes. chronic lung disease; emphysema. anemia. hyperthyroidism. right to left shunt. congenital heart disease. pulmonary vascular disease.

Carbon Dioxide Transport 24
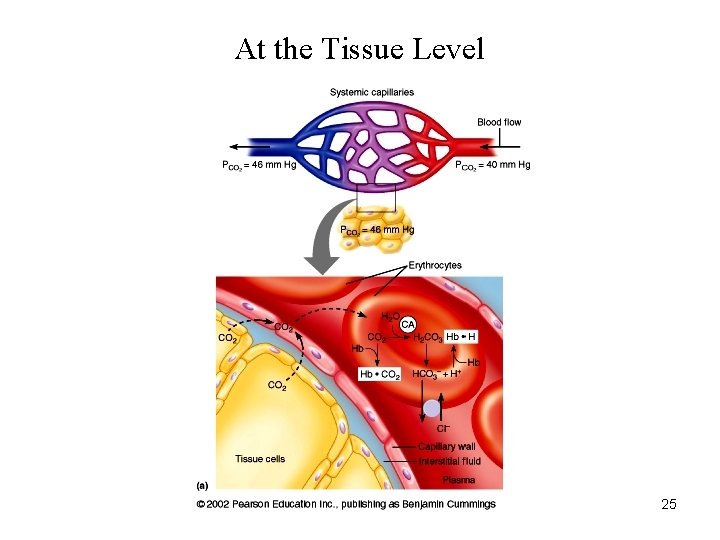
At the Tissue Level 25
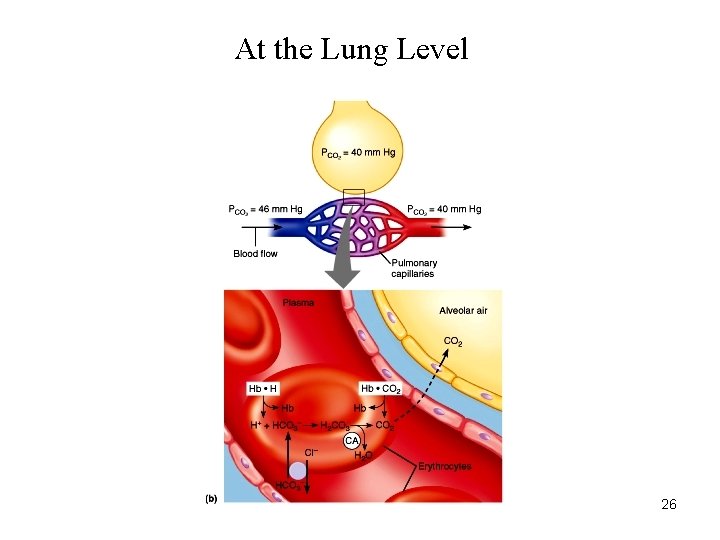
At the Lung Level 26

Carbon Dioxide Transport • CO 2 is transported in blood in dissolved form, as bicarbonate ions, and as protein-bound carbamino compound. • Protein-bound CO 2 (carbamino compounds): • Amount of CO 2 stored as carbamino compounds is about 21 ml/L (4% of the total art CO 2). 27

Carbon Dioxide Transport • A majority amount of CO 2 is transported in the form of bicarbonate ions (HCO 3 -): • Amount of CO 2 in HCO 3 - form at PCO 2=40 mm. Hg is about 420 ml/L (90% of the total arterial CO 2). 28

Carbon Dioxide Transport • Haldane Effect: Increasing O 2 -saturation reduces CO 2 content and shifts the CO 2 dissociation curve to right. This is because, increasing PO 2 leads to : – Decrease in the formation of carbamino compound. – Release of H+ ions from the hemoglobin and resulting in dehydration of HCO 3 -. 29
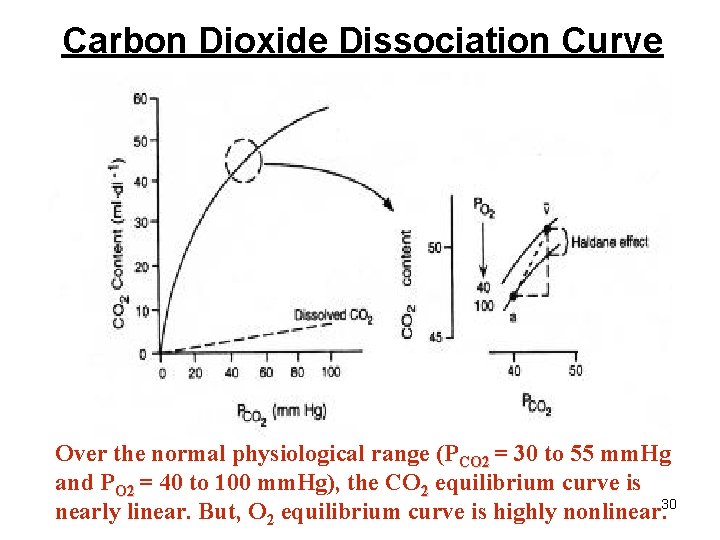
Carbon Dioxide Dissociation Curve Over the normal physiological range (PCO 2 = 30 to 55 mm. Hg and PO 2 = 40 to 100 mm. Hg), the CO 2 equilibrium curve is nearly linear. But, O 2 equilibrium curve is highly nonlinear. 30
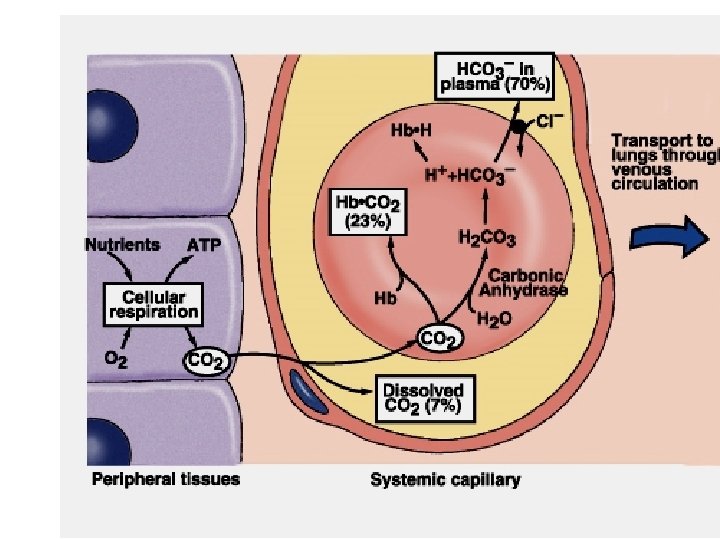

Bicarbonate in RBCs. • Carbonic anhydrase is present in RBCs • CO 2 forms carbonic acid which dissociates to H+ and HCO 3 • Released H+ is buffered by histidine residues (imidazole group) • Percent of the total Pa. CO 2: 70%

Carbamino Compounds in RBCs. • Approximately 30% of RBC contents is Hb • CO 2 forms carbamino hemoglobin • Released H+ is buffered by histidine residues (imidazole group) • Percent of the total Pa. CO 2: 23 %

CO 2 Formation in Plasma • Carbamino compounds – CO 2 binds the amine groups of plasma proteins to form carbamino compounds.

35
36

Chloride Shift (Hamburger Shift) • Newly formed HCO 3 - passes out of RBC • Cl- diffuses into RBC to maintain electroneutrality – Chloride shift is rapid – Complete before the RBCs exit capillary

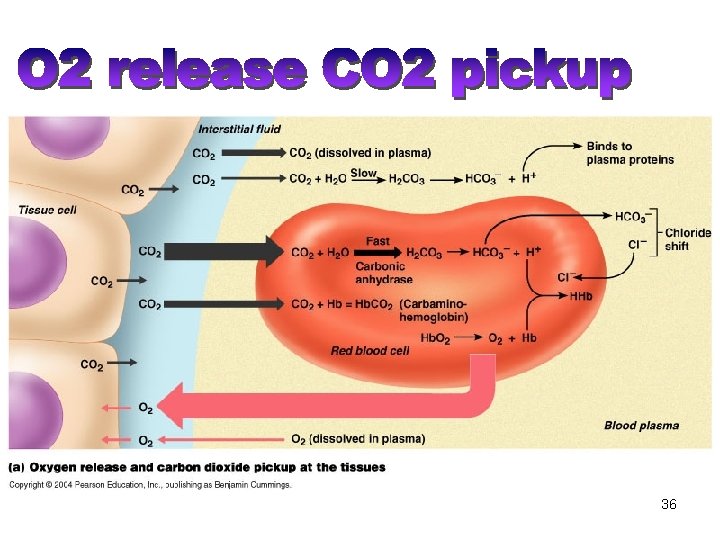
Tissue-Gas Exchange: Summary • Gas exchange processes in the peripheral organs are essentially opposite those in the lungs. • O 2 is released from the capillary blood to the tissues and diffuses to the mitochondria where O 2 is converted to CO 2 and energy (ATP) through cellular metabolism. • CO 2 diffuses from the tissues to the blood stream and is transported to the lungs for elimination. • The exchange of O 2 and CO 2 in the blood-tissue exchange unit depends on PO 2, PCO 2, and also on O 2 and CO 2 saturation curves. 38

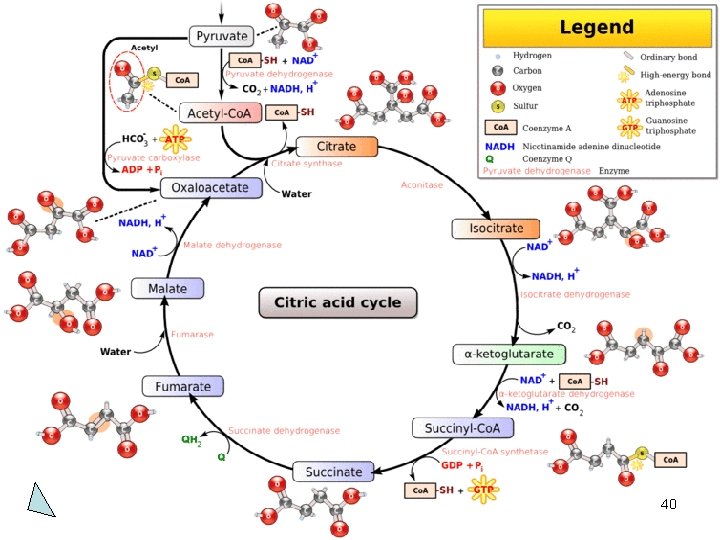
Gas Transport in Cell 39
40

Pelepasan CO 2 • Dilakukan oleh: 1. isositrat dehidrogenase 2. α-ketoglutarat dehidrogenase • Pelepasan CO 2 tidak mengkonsumsi oksaloasetat. 41

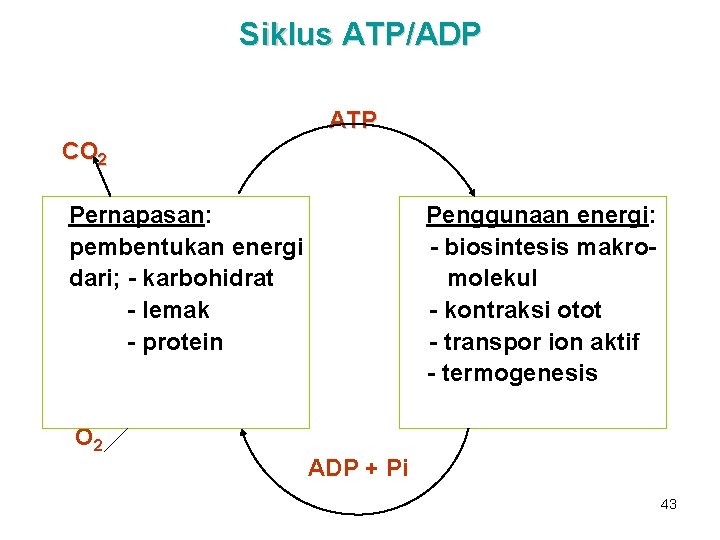
Siklus ATP/ADP • • • Berperan untuk menghubungkan proses yg menghasilkan P-berenergi-tinggi dgn proses yg menggunakan P-berenergitinggi. ATP dikonsumsi & dibentuk kembali secara kontinu. Depot ATP/ADP sangat kecil, sehingga hanya cukup untuk mempertahankan jaringan aktif dlm waktu beberapa detik saja. 42
Siklus ATP/ADP ATP CO 2 Pernapasan: pembentukan energi dari; - karbohidrat - lemak - protein Penggunaan energi: - biosintesis makromolekul - kontraksi otot - transpor ion aktif - termogenesis O 2 ADP + Pi 43

Fosforilasi Oksidatif • Adalah sistem dalam mitokondria yang memasangkan respirasi dengan proses pembentukan intermediat berenergi tinggi, ATP. • Sistem ini memungkinkan organisme aerob menangkap energi bebas dari substrat respiratorik dalam jumlah lebih besar dibanding organisme anaerob. 44
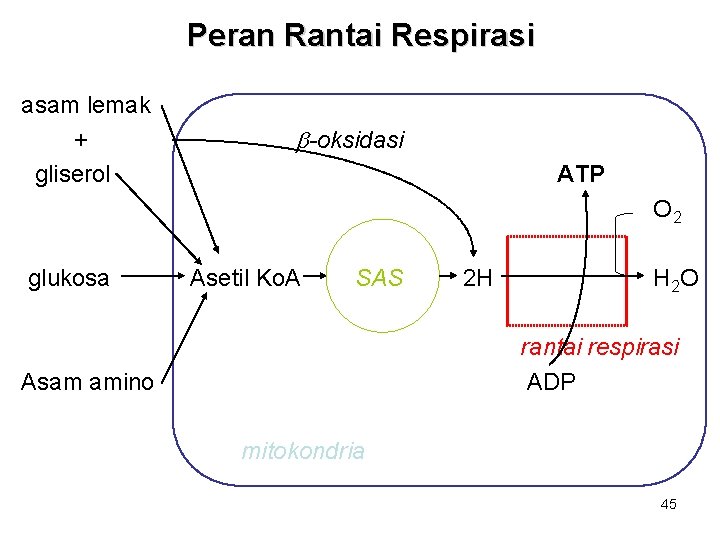
Peran Rantai Respirasi asam lemak + gliserol b-oksidasi ATP O 2 glukosa Asetil Ko. A SAS 2 H H 2 O rantai respirasi ADP Asam amino mitokondria 45
46

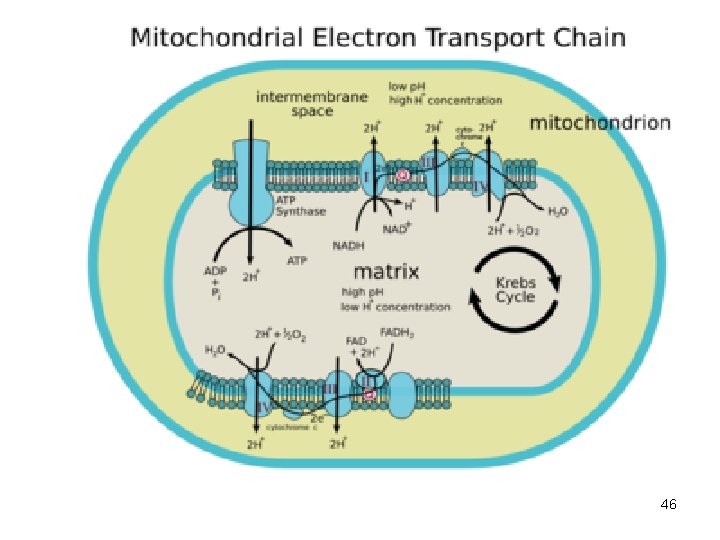
Produk ATP pada Fosforilasi Oksidatif Berdasarkan hipotesis kimiosmotik dari Mitchell yaitu; rantai bekerja --> proton dipompa keluar dari membran dlm mitokondria --> p. H antar membran turun --> proton balik ke dalam matrik lewat tonjolan ATP-sintase--> fosforilasi ADP menjadi ATP. 47

Produk ATP pada Fosforilasi Oksidatif • • • Diperkirakan satu ATP disintesis setiap dua proton melewati tonjolan tsb. Hasilnya ialah; - 3 mol. ATP utk oksidasi 1 mol. NADH - 2 mol. ATP utk oksidasi 1 mol. FADH 2 Laju fosforilasi oksidatif dikendalikan oleh; NADH, oksigen, ADP 48

Resources • BIOEN 589: Integrative Physiology. Download 24 jan 05. • Kennelly, PJ. , Rodwell, V W. Proteins: Myoglobin & Hemoglobin. In: Harper’s Illustrated Biochemistry. 27 th Ed. 41 - 8. • Miliefsky, M. Respiratory System Ch. 23. Download 24 Nov 10. • Sheardown, H. Blood Biochemistry. Mc. Master University. Download 20 Mei 07. • Irvin, CG. Respiratory Physiology. Lecture 4 A CO 2 Transport. In: MEDICAL PHYSIOLOGY 30. Download 22 Jun 09. • Marks, DB. , Marks, AD. , Smith CM. Basic medical biochemistry: a clinical approach. 1996. Dalam: B. U. Pendit, penerjemah. Biokimia Kedokteran Dasar: Sebuah Pendekatan Klinis. Eds. J. Suyono. , V. Sadikin. , L. I. Mandera. Jakarta: EGC, 2000 • R. K. Murray, D. K. Granner, P. A. Mayes, V. W. Rodwell Harper’s Biochemistry. 27 th ed. Mc. Graw-Hill Companies, New York. 2006. 49
- Slides: 48