RESPIRATORY FAILURE Presented by Ahmad Bataineh Abdelrazzaq Malhas

RESPIRATORY FAILURE Presented by : • Ahmad Bataineh • Abdelrazzaq Malhas • Abdalrahman • Noor Zahran Adeen Abo Salameh Supervised by: Dr. Lina Al Shadfan

INTRODUCTION Respiratory failure occurs when oxygenation and ventilation are insufficient to meet the metabolic demands of the body. Although respiratory failure is traditionally defined as respiratory dysfunction resulting in arterial partial pressure of oxygen (Pa. O 2 ) <60 mm Hg when breathing room air and Pa. CO 2 >50 mm Hg resulting in acidosis, the patient's general state, respiratory effort, and potential for impending exhaustion are more important indicators than ABG values. So it is diagnosed when the patient’s respiratory system loses the ability to provide sufficient oxygen to the blood, and hypoxemia develops, or when the patient is unable to adequately ventilate, and hypercarbia and hypoxemia develop.

TYPES OF RESPIRATORY FAILURE Type I respiratory failure involves low oxygen, and normal or low carbon dioxide levels. Type II respiratory failure involves low oxygen, with high carbon dioxide. Type III Type IV

Type I respiratory failure (hypoxic respiratory failure) occurs because of damage to lung tissue. This lung damage prevents adequate oxygenation of the blood (hypoxaemia); however, the remaining normal lung is still sufficient to excrete the carbon dioxide being produced by tissue metabolism. This is possible because less functioning lung tissue is required for carbon dioxide excretion than is needed for oxygenation of the blood.

Type II respiratory failure (hypercarbic respiratory failure) is also known as ‘ventilatory failure’. It occurs when alveolar ventilation is insufficient to excrete the carbon dioxide being produced. Inadequate ventilation is due to reduced ventilatory effort, or inability to overcome increased resistance to ventilation – it affects the lung as a whole, and thus carbon dioxide accumulates. Complications include: damage to vital organs due to hypoxaemia, CNS depression due to increased carbon dioxide levels, respiratory acidosis (carbon dioxide retention). This is ultimately fatal unless treated. Complications due to treatment may also occur.


PATHOPHYSIOLOGY

PATHOPHYSIOLOGY Systemic venous (pulmonary arterial) blood is arterialized after equilibration with alveolar gas in the pulmonary capillaries and is carried back to the heart by pulmonary veins. The ABG is influenced by the composition of inspired gas , effectiveness of alveolar ventilation, pulmonary capillary perfusion, and diffusion capacity of the alveolar capillary membrane. Abnormality in any of these steps can result in respiratory failure.

Hypoxemia is caused by one of the following abnormalities: 1. Mismatch between alveolar ventilation (V) and pulmonary perfusion (Q) 2. Intrapulmonary shunt 3. Hypoventilation 4. Abnormal diffusion of gases at the alveolarcapillary interface 5. Reduction in inspired oxygen concentration 6. Increased venous desaturation with cardiac dysfunction plus one or more of the above 5 factors

VENTILATION-PERFUSION MISMATCH, INTRAPULMONARY SHUNT, AND HYPOVENTILATION Alveolar gas exchange depends not only on ventilation of the alveoli but also on circulation of blood through the alveolar capillaries. In other words it depends both on ventilation and perfusion. This makes sense. You need both oxygen in the alveoli, and adequate blood flow past alveoli to pick up oxygen, other wise oxygen cannot be delivered. When the proper balance is lost between ventilated alveoli and good blood flow through the lungs, ventilation perfusion mismatch is said to exist. The ventilation/perfusion ratio is often abbreviated V/Q mismatch is common and often effects our patient’s ventilation and oxygenation. There are 2 types of mismatch: dead space and shunt. Shunt is perfusion of poorly ventilated alveoli. Physiologic dead space is ventilation of poor perfused alveoli.

WHAT IS THE DEAD SPACE? Dead space is the portion of the respiratory system where tidal volume doesn’t participate in gas exchange: it is ventilated but not perfused. There are three types of dead space: anatomic, physiologic, and that dead space belonging to any airway equipment being used to assist ventilation.

ANATOMIC DEAD SPACE Anatomic deadspace consists of the parts of the respiratory tract that are ventilated but not perfused. It consists of conducting airways such as the trachea, bronchi, and bronchioles —structures that don’t have alveoli. It’s called anatomic because it’s fixed by anatomy and doesn’t change. About a third of each normal breath we take is anatomic dead space, which means that a third of each breath is essentially wasted. Dead space is age dependent. It’s highest in the infant at 3 ml/kg ideal body weight and is about 2 ml/kg in older children and adults. An adequate tidal volume must include enough volume to also fill the deadspace, otherwise not enough air enters the alveoli and the patient hypoventilates.


Let’s say our baby is sick with nausea, vomiting, and a fever of 101 F (38 C). She starts to hypoventilate and is now breathing tidal volumes of 10 ml. She’s still moving 10 ml of gas in and out of her mouth and you can feel her breathing and see her chest move, even though it looks shallow. Her dead space is still 8 so now the amount of gas reaching her alveoli is 2 ml (10 ml – 8 ml). That’s not enough. If you’re providing our baby with extra oxygen, she may not become hypoxic right away because enough oxygen will still reach her alveoli to maintain her oxygen saturation for a while. However, she is barely moving her dead space gas back and forth so her ventilation is poor. As a result, her carbon dioxide starts to rise. Hypoventilation leads to increased Pa. CO 2

PHYSIOLOGIC DEAD SPACE 1. 2. 3. A second type of dead space, physiologic dead space, consists of alveoli that are ventilated but lack capillary blood flow to pick up oxygen and drop off carbon dioxide. In other words, they are not perfused. Dead space can change from minute to minute with alterations in cardiac output and pulmonary blood flow. Many things can impair alveolar perfusion and increase physiologic dead space such as: cardiovascular shock (blood flow to the lungs is decreased), emphysema (lots of enlarged alveoli with less surface area and fewer alveolar capillaries) pulmonary embolus (flow is blocked by clot).


Let’s go back to the baby in our clinical scenario. Perhaps the baby is hypoventilating because she is in shock from diarrhea. Now she has two reasons for respiratory failure … She’s hypoventilating, and barely exceeding her anatomic dead space. She’s in cardiovascular shock. Hypovolemia and acidosis is decreasing her cardiac output and lung perfusion. Her physiologic dead space has increased and she is not perfusing all of the alveoli that are still getting ventilated. Remember, hypovolemia and shock increases physiologic dead space

WHAT IS PULMONARY SHUNT? Another contributor to ventilation perfusion mismatch is shunt. Shunt is the opposite of dead space and consists of alveoli that are perfused, but not ventilated. Blood flowing past poorly ventilated alveoli doesn’t pick up additional oxygen. This poorly oxygenated blood returns to the heart and mixes with oxygenated blood coming from other areas of the lungs that are ventilated. The mixture lowers the total oxygen content of the arterial blood, producing hypoxemia. The larger the shunt, the lower the oxygen content. Giving a patient with an intrapulmonary shunt 100% oxygen to breathe won’t increase the Pa. O 2 much.


1. 2. 3. 4. 5. 1. 2. 3. Common causes of shunt occur in lung tissue disease and include: pneumonia and pulmonary edema: some alveoli filled with fluid tissue trauma: alveolar wall swelling atelectasis: collapse of alveoli from failure to expand, or absorbsion of the air out of the alveoli without replacing it mucous plugging: air can’t get into the alveoli pulmonary arteriovenous fistulas Other factors cause atelectasis : painful breathing from surgery or trauma depressed levels of consciousness such as from drug, injury, or illness the disease process itself

DIFFUSION Even if ventilation and perfusion are matched, gas exchange requires diffusion across the interstitial space between alveoli and pulmonary capillaries. Under normal conditions, there is sufficient time for the pulmonary capillary blood to equilibrate with alveolar gas across the interstitial space. When the interstitial space is filled with inflammatory cells or fluid, diffusion is impaired. Because the diffusion capacity of CO 2 is 20 times greater than that of O 2 , diffusion defects manifest as hypoxemia rather than hypercarbia. Even with the administration of 100% oxygen, PAO 2 increases to approximately 660 mm Hg from 100 mm Hg at sea level, and the concentration gradient for diffusion of O 2 is increased by only 6. 6 times.

Therefore, with diffusion defects, lethal hypoxemia will set in before clinically significant CO 2 retention results. In fact, in such situations, Pa. CO 2 is often decreased because of the hyperventilation that accompanies hypoxemia. Presence of hypercarbia in diseases that impair diffusion is indicative of alveolar hypoventilation from coexisting airway obstruction, exhaustion, or CNS depression. Examples of disease that impair diffusion are interstitial pneumonia, ARDS, scleroderma, and pulmonary lymphangiectasia.

SUMMARY Respiration involves the nervous, cardiovascular, musculoskeletal , and respiratory systems. The causes of respiratory failure can come from any of these systems and are expansive. The pathophysiologic mechanisms that lead to respiratory failure involve primarily either V/Q mismatch or impairment of oxygen transfer at the alveolar-capillary membrane. A high V/Q ratio is when the alveoli are well ventilated but are not well perfused. High V/Q ratios act like dead space. A low V/Q ratio is when the alveoli units are well perfused but are not well ventilated. Low V/Q ratios act like shunts. These 2 common pathophysiologic mechanisms of respiratory failure are observed in a variety of diseases. Pulmonary conditions that involve the bronchi (eg, status asthmaticus and bronchiolitis) or inflammation or infection of the parenchyma (eg, pneumonia, aspiration, cystic fibrosis, and ciliary dysmotility) result in airway obstruction and/or parenchymal loss, leading to V/Q mismatch and impaired gas exchange.

ASTHMA AND RESPIRATORY FAILURE Status asthmaticus occurs because of progression of airway inflammation, bronchospasm, and mucous plugging during days to weeks or sudden onset of asphyxia from bronchospasm. In either case, airway obstruction causes V/Q mismatch, incomplete alveolar gas exchange, and lung hyperinflation. End-expiratory alveolar pressure increases as a result, creating an autopositive end-expiratory pressure state. Work of breathing is increased to overcome the autopositive end-expiratory pressure for inspiratory flow to occur, eventually leading to inspiratory muscle fatigue and respiratory failure.

CYSTIC FIBROSIS AND RESPIRATORY FAILURE In cystic fibrosis, similarly, an acute pulmonary exacerbation can result in airway obstruction and mucous plugging, leading to V/Q mismatch and reduced functional residual capacity. Increased work of breathing results in respiratory muscle fatigue, leading to hypoventilation, hypercarbia, and respiratory failure.

EPIDEMIOLOGY Approximately half of respiratory failure cases seen in the neonatal period, WHY? WHY I. Smaller upper airway (comparing with adults) with subglottic area is the narrowest. II. Immature stage of lung development present with fewer number of alveoli , smaller intrathoracic airway caliber , and underdeveloped collateral ventilation III. Infant respiratory muscle have reduced Type I muscle fibers(specially the diaphragm) IV. Bradypnea , Apnea, or tachypnea results commonly from immaturity of respiratory center. v

CAUSES v Lung & Airway disorder: � Lung Parenchyma( Bronchiolitis , Severe asthma, Etc) � Airway ( Croup, Laryngo. Tracheo. Broncho. Malagia, Etc) v Respiratory Pump Failure: Restrictive lung disorder ( Kyphoscoliosis) • Chest wall abnormality ( Flat chest) • Neuromuscular disorder ( Phrenic nerve paralysis) • v Respiratory Center failure: � Brain Injury � CNS Infection � Drug overdose v Failure to meet increased metabolic demands � Septic Shock


APPROACH

EMERGENCY Respiratory support should be urgently provided for a patient with significant tachypnea, retractions, grunting, nasal flaring, and head bobbing ( De Musset’s sign) Delay in respiratory support may lead to increase in fatigue , shallow breathing , reduced consciousness and cyanosis.

CLINICAL FINDINGS Signs and symptoms of Acute respiratory failure reflects the underlying disease and the associated hypoxemia or hypercapnia. Type I ( Hypoxia)

Type II ( Hypercapnia) :

Other signs: 1. Asterixis (flapping tremor) : sever Hypercapnia 2. Tachycardia & Arthythmias : Hypoxemia &acidosis 3. Cyanosis 4. Excessive Respiratory effort 5. Somnolence( state of strong desire for sleep) 6. Seizures & myoclonus 7. Hepatomegaly & peripheral Edema (Cor pulmonale Sequance)

HISTORY The diagnosis of Acute or chronic respiratory failure begins with clinical suspicion of its presence. Confirmation of the diagnosis is based on ABG. Evaluation of the underlying cause must initiate early , Evident after careful history and examination.

RISK FACTORS Prematurity Immunodeficiency Anatomical abnormality Chronic Disorders (pulmonary , cardiac , Neuromuscular disorder): � Cystic fibrosis � Asthma � Unrepaired congenital heart disease � Myasthenia graves Hx of Fever , respiratory infection, seizures , head trauma

RELATION BETWEEN ACUTE RESPIRATORY FAILURE AND KIDNEY TRANSPLANT: Canet et al Study determined hat 200 of 6819 kidney transplant recipient required admission to ICU for acute respiratory failure (due to Graft failure or use of antilymphocyte globulin for rejection) which was associated with high mortality rates How to improve the outcome: 1. Early ICU admission 2. Increased Bacterial & Pneumocystis prophylaxis(trimethoprim/sulfamethoxazole)

PHYSICAL EX General Look Vitals : Tachypnea is sensitive indicator of respiratory failure & one of the earliest compensatory mechanisms Heart rate Also INCREASE Blood Pressure Normal or High Tempreture

CHEST EX Respiratoy rate from vitals: - Tachypnia with nasal flaring and retractions needs respiratory support - Bradypnia usually sign of Respiratory center failure & Neuromuscular disorders. Inspection of Chest wall while breathing to determine if there is - Asymmetrical chest wall movement which indicates Pnemothorax , Sever Empyema, Plural Effusion Or Chest trauma. - Paradoxical Movement of chest and abdomen indicate respiratory distress

Ascultation: - Expiratory wheeze reflects disease of the lower airways, such as asthma - A local or asymmetric wheeze can indicate possible airway obstruction due to a foreign body or a mass - Stridor is a high-pitched inspiratory wheeze usually caused by upper airway narrowing or obstruction in such conditions as laryngomalacia, croup, tracheitis, subglottic stenosis - Crackles or rales are heard in pneumonia, congestive heart failure, pulmonary fibrosis

Heart ( Abnormal Heart sounds) Neurological - Glasgow coma scale - Muscle strength ( decreased in Guillain-barr Syndrome OR Duchenne Muscular Dystrophy)

WORK UP Respiratory Failure may be associated with verity of clinical manifestations however these are non specific , and very significant RF may be present without dramatic S&Sx. This is why its very important to do more investigations to confirm the diagnosis and reveals the cause.

LAB CBC (anemia & Polycythemia) Blood , Urine Cultures & PCR ( Identify the cause of infection) KFT LFT Electrolytes ( K, Mg, P, Na) Creatine Kinase Troponin I TSH

ARTERIAL BLOOD GAS ABG offers a valuable assistance in Diagnosis, Monitoring and management. Due to Technical Difficulty in obtaining an arterial sample from children , Capillary Blood Gas (CBG) obtained in emergency room CBG provides good estimation of Pa. Co 2 & Arterial PH But less for Pa. O 2.

Acute Respiratory Failure: ABG Result: Pa. O 2: <60 mm. Hg PH: < 7. 35 Pa. Co 2 : > 50 mm. Hg HCO 3: low or Normal Ø

Chronic Respiratory Failure: ABG Result: Pa. Co 2: > 50 mm. Hg PH: Normal HCO 3: Increased ( > 26 m. Eq/L ) Ø

Chest X-Ray Essential in evaluation of respiratory failure, it mostly reveal the cause. ARDS: Bilateral airspace infiltrate v
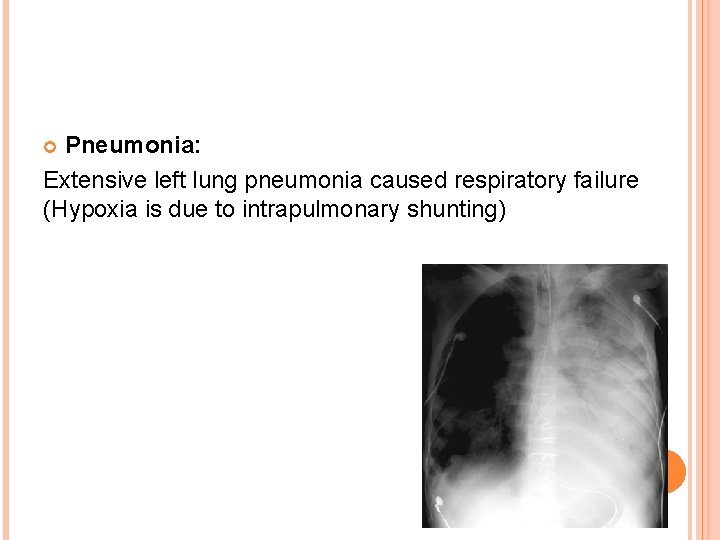
Pneumonia: Extensive left lung pneumonia caused respiratory failure (Hypoxia is due to intrapulmonary shunting)
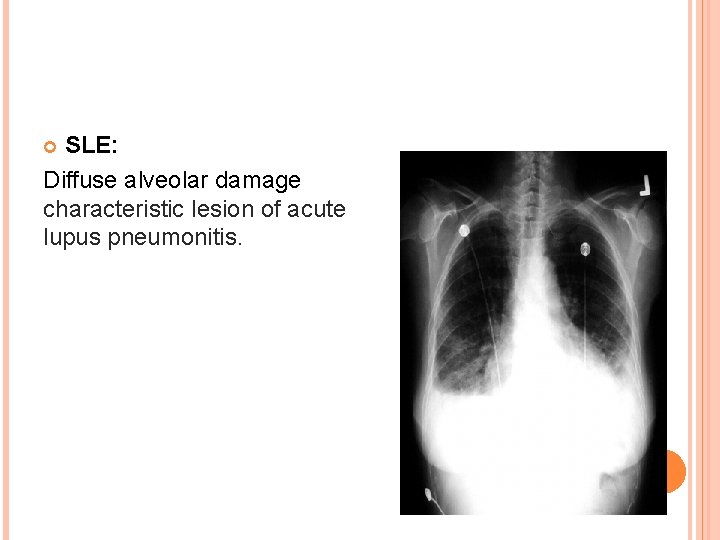
SLE: Diffuse alveolar damage characteristic lesion of acute lupus pneumonitis.

Echocardiogram: Not routinely performed , but its useful test for cardiac causes. Cardiogenic pulmonary edema : Left ventricular dilatation, sever mitral regurgitation. ARDS: Normal Heart size, normal systolic and diastolic function in patient with pulmonary edema v

Pulmonary Function Test: Generally Patient with acute respiratory failure are unable to perform PFT. However these tests are useful in evaluation of Chronic Respiratory failure. v Tests : spirometry lung volumes diffusion capacity and maximal respiratory pressures v
MANAGEMENT

The goal of management for respiratory distress and respiratory failure is to ensure a patent airway and provide necessary support for adequate oxygenation of the blood and removal of CO 2. Compared with hypercapnia, hypoxemia is a lifethreatening condition; therefore initial therapy for respiratory failure should be aimed at ensuring adequate oxygenation.

Oxygen Administration Supplemental oxygen administration is the least invasive and most easily tolerated therapy for hypoxemic respiratory failure. Nasal cannula oxygen provides low levels of oxygen supplementation and is easy to administer. Oxygen is humidified in a bubble humidifier and delivered via nasal prongs inserted in to the nares. In children, a flow rate <5 L/min is most often used because of increasing nasal irritation with higher flow rates.

The typical FIO 2 value (expressed as percentage rather than fraction of 1) is between 23% and 40%, although the FIO 2 varies according to the size of the child, the respiratory rate, and the volume of air moved with each breath.

simple mask may be used, which consists of a mask with open side ports and a valveless oxygen source. Variable amounts of room air are entrained through the ports and around the side of the mask, depending on the fit, size, and minute volume of the child. Oxygen flow rates vary from 5 -10 L/min, yielding typical FIO 2 values (expressed as percentage rather than fraction of 1) between 30% and 65%. If more precise delivery of oxygen is desired, other mask devices should be used.

A Venturi mask provides preset FIO 2 through a mask and reservoir system by entraining precise flow rates of room air into the reservoir along with high-flow oxygen. The adapter at the end of each mask reservoir determines the flow rate of entrained room air and the subsequent FIO 2. (Adapters provide FIO 2 of 0. 30 - 0. 50. ) Oxygen flow rates of 5 -10 L/min are recommended to achieve the desired FIO 2 and to prevent rebreathing. Partial rebreather and non-rebreather masks use a reservoir bag attached to a mask to provide higher FIO 2.


Partial rebreather masks have 2 open exhalation ports and contain a valveless oxygen reservoir bag. Some exhaled gas can mix with reservoir gas, although most exhaled gas exits the mask via the exhalation ports. Through these same ports, room air is entrained, and the partial rebreather mask can provide FIO 2 up to 0. 60, for as long as oxygen flow is adequate to keep the bag from collapsing (typically 10 -15 L/ min). As with nasal cannulas, smaller children with smaller tidal volumes entrain less room air, and their Fio 2 values will be higher.

Non-rebreather masks include 2 one-way valves, 1 between the oxygen reservoir bag and the mask and the other on 1 of the 2 exhalation ports. This arrangement minimizes mixing of exhaled and fresh gas and entrainment of room air during inspiration. The 2 nd exhalation port has no valve, a safeguard to allow some room air to enter the mask in the event of disconnection from the oxygen source. A non- rebreather mask can provide FIO 2 up to 0. 95. The use of a non-rebreather mask in conjunction with an oxygen blender allows delivery of FIO 2 between 0. 50 and 0. 95 (Table 89. 8 ). When supplemental oxygen alone is inadequate to improve oxygenation, or when ventilation problems coexist, additional therapies may be necessary.


AIRWAY ADJUNCTS Maintenance of a patent airway is a critical step in maintaining adequate oxygenation and ventilation. Artificial pharyngeal airways may be useful in patients with oropharyngeal or nasopharyngeal airway obstruction and in those with neuromuscular weakness in whom inherent extrathoracic airway resistance contributes to respiratory compromise. An oropharyngeal airway is a stiff plastic spacer with grooves along each side that can be placed in the mouth to run from the teeth along the tongue to its base just above the vallecula. The spacer prevents the tongue from opposing the posterior pharynx and occluding the airway. Because the tip sits at the base of the tongue, it is usually not tolerated by patients who are awake or whose gag reflex is strong.

The nasopharyngeal airway , or nasal trumpet , is a flexible tube that can be inserted into the nose to run from the nasal opening along the top of the hard and soft palate with the tip ending in the hypopharynx. It is useful in bypassing obstruction from enlarged adenoids or from contact of the soft palate with the posterior nasopharynx. Because it is inserted past the adenoids, a nasopharyngeal airway should be used with caution in patients with bleeding tendencies.

INHALED GASES Helium-oxygen mixture (heliox) is useful in overcoming airway obstruction and improving ventilation. Helium is much less dense and slightly more viscous than nitrogen. When substituted for nitrogen, helium helps maintain laminar flow across an obstructed airway, decreases airway resistance, and improves ventilation. It is especially helpful in diseases of large airways obstruction in which turbulent airflow is more common, such as acute laryngotracheobronchitis, subglottic stenosis, and vascular ring. It is also used in patients with severe status asthmaticus. To be effective, helium should be administered in concentrations of at least 60%, so associated hypoxemia may limit its use in patients requiring >40% oxygen

Inhaled nitric oxide (i. NO) is a powerful inhaled pulmonary vasodilator. Its use may improve pulmonary blood flow and V/Q mismatch in patients with diseases that elevate pulmonary vascular resistance, such as occurs in persistent pulmonary hypertension of the newborn, primary pulmonary hypertension, and secondary pulmonary hypertension as a result of chronic excess pulmonary blood flow (e. g. , ventriculoseptal defect) or collagen vascular diseases. i. NO is administered in doses ranging from 5 to 20 parts per million of inspired gas. Although administration of i. NO to unintubated patients is possible, it is usually administered to patients undergoing mechanical ventilation via an endotracheal tube, because of the need for precision in i. NO dosing

NON INVASIVE POSITIVE PRESSURE SUPPORT It is useful in treating both hypoxemic and hypoventilatory (hypercapnic) respiratory failure. Positive airway pressure helps with aeration of partially atelectatic or filled alveoli, prevention of alveolar collapse at end-exhalation, and increase in functional residual capacity (FRC). These actions improve pulmonary compliance and hypoxemia, as well as decrease intrapulmonary shunt. In addition, it is useful in preventing collapse of extrathoracic airways by maintaining positive airway pressure during inspiration. Improving compliance and overcoming airway resistance also improves tidal volume and therefore ventilation.

CON. A highflow nasal cannula delivers gas flow at 4 -16 L/min and up to 60 L/min, with newer systems for older children and adolescents, capable of providing significant continuous positive airway pressure (CPAP). In this setting, the amount of CPAP provided is not quantifiable and varies with each patient, depending on the percentage of total inspiratory flow that is delivered from the cannula, airway anatomy, and degree of mouth breathing. The FIO 2 (of room air = 21%) can be adjusted by provision of gas flow through an oxygen blender. Another benefit of a highflow nasal cannula system is the washout of CO 2 from the nasopharynx, which decreases rebreathing of CO 2 and dead space ventilation.

CON. Noninvasive CPAP is most useful in diseases of mildly decreased lung compliance and low FRC, such as atelectasis and pneumonia. Patients with diseases of extrathoracic airway obstruction, in which extrathoracic negative airway pressures during inspiration lead to airway narrowing (e. g. , laryngotracheitis, obstructive sleep apnea, postextubation airway edema), may also benefit from CPAP. Potential risks include nasal irritation, hyperinflation from excessive CPAP in smaller patients, and abdominal distention from swallowed air (possibly aspiration).

INVASIVE POSITIVE PRESSURE SUPPORT ENDOTRACHEAL INTUBATION AND MECHANICAL VENTILATION Mechanical ventilation: use of a ventilator to assist or completely replace spontaneous breathing When hypoxemia or significant hypoventilation persists despite the interventions already described, endotracheal intubation and mechanical ventilation are indicated. Additional indications for intubation include maintaining airway patency in patients who have the potential for airway compromise, such as those with actual or potential neurologic deterioration, and in patients with hemodynamic instability. Mechanical ventilation increases minute ventilation and decreases dead space. This approach is the mainstay of treatment for acute hypercapnia and severe hypoxemia.

CON. Administration of a sedative and analgesic followed by a paralytic agent is a common pharmacologic regimen for facilitating intubation. In fact, sedation and paralysis with neuromuscular blocking agents should be considered standard unless contraindicated. Dexmedetomidine has been a standard sedating agent for maintenance during mechanical ventilation. An alternative to this pharmacologic approach is rapid sequence intubation , used when endotracheal intubation is urgent, or the patient is suspected of having a full stomach and at increased risk of aspiration. Once adequate sedation and/or paralysis have been achieved, ventilation should be assisted with a bagmask device.

CON. After insertion, Rapid confirmation of ETT placement is essential and should be assessed by as many of the following steps as possible: presence of Pet. CO 2 determined by a monitor attached in-line with ETT; auscultation of both lung fields as well as the epigastrium for equal breath sounds; and, good air movement and evaluation of the abdomen for increasing distention. Adequate, bilateral chest expansion and misting inside the ETT with each breath confirm proper tube placement. An increasing heart rate, if heart rate has decreased during the attempt, and a rising or normal Sp. O 2 reading are suggestive of successful tube placement. A chest radiograph should also be obtained to confirm proper placement of the ETT, which should lie with the tip about halfway between the glottis and the carina

TRANSIENT MANUAL VENTILATION IN IMMEDIATE PREINTUBATION AND POSTINTUBATION PERIODS Establishment of supportive ventilation via bag-mask or bag-ETT is required before transport of the patient to a setting of continued critical care. The technique of manual ventilation should take into account the underlying pathology. Mechanical ventilation of patients with diseases characterized by low FRC (e. g. , pneumonia, pulmonary edema, ARDS) should include the application of PEEP to prevent alveolar derecruitment. Lung volume recruitment can be accomplished with a PEEP valve on a self-inflating ventilation bag or by careful manipulation of exhaust gas using an anesthesia bag. Such diseases are also characterized by a short time constant for lung deflation and therefore are best managed with relatively small tidal volumes and high ventilation rates. In contrast, diseases characterized by airway obstruction have prolonged deflation time constants and are therefore best managed with relatively slow ventilation rates and high tidal volumes.


THANK YOU
- Slides: 73