Resonant SFG Line Shapes on Single Crystal Surfaces






- Slides: 18

Resonant SFG Line Shapes on Single Crystal Surfaces 63 rd OSU International Symposium on Molecular Spectroscopy Friday, June 20 th 2008 - Columbus, Ohio Scott K. Shaw, A. Laguchev, D. Dlott, A. Gewirth Department of Chemistry University of Illinois at Urbana-Champaign

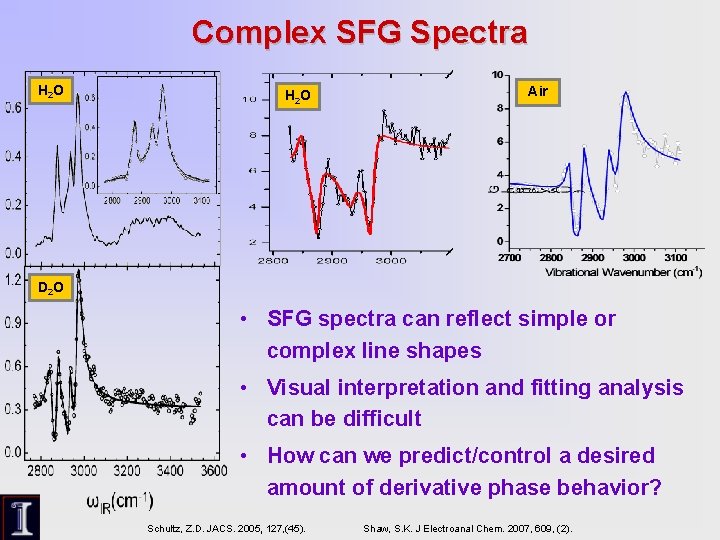
Complex SFG Spectra H 2 O Air D 2 O • SFG spectra can reflect simple or complex line shapes • Visual interpretation and fitting analysis can be difficult • How can we predict/control a desired amount of derivative phase behavior? Schultz, Z. D. JACS. 2005, 127, (45). Shaw, S. K. J Electroanal Chem. 2007, 609, (2).

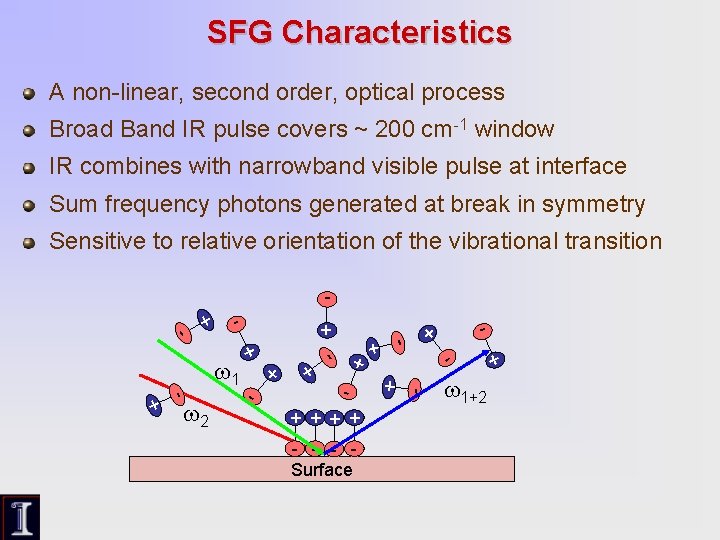
SFG Characteristics A non-linear, second order, optical process Broad Band IR pulse covers ~ 200 cm-1 window IR combines with narrowband visible pulse at interface Sum frequency photons generated at break in symmetry Sensitive to relative orientation of the vibrational transition + + + - - - Surface + - - + + - - 1+2 + - + + - 2 - + + - 1 +

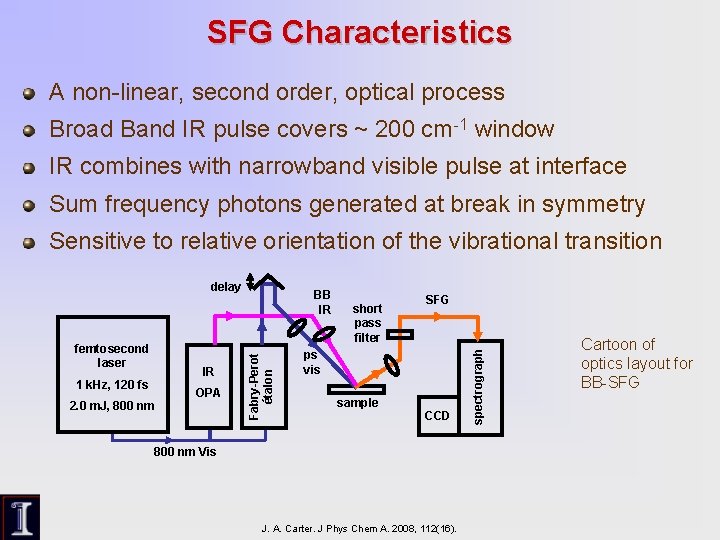
SFG Characteristics A non-linear, second order, optical process Broad Band IR pulse covers ~ 200 cm-1 window IR combines with narrowband visible pulse at interface Sum frequency photons generated at break in symmetry Sensitive to relative orientation of the vibrational transition IR 1 k. Hz, 120 fs 2. 0 m. J, 800 nm OPA Fabry-Perot étalon femtosecond laser BB IR short pass filter SFG ps vis sample CCD 800 nm Vis J. A. Carter. J Phys Chem A. 2008, 112(16). spectrograph delay Cartoon of optics layout for BB-SFG

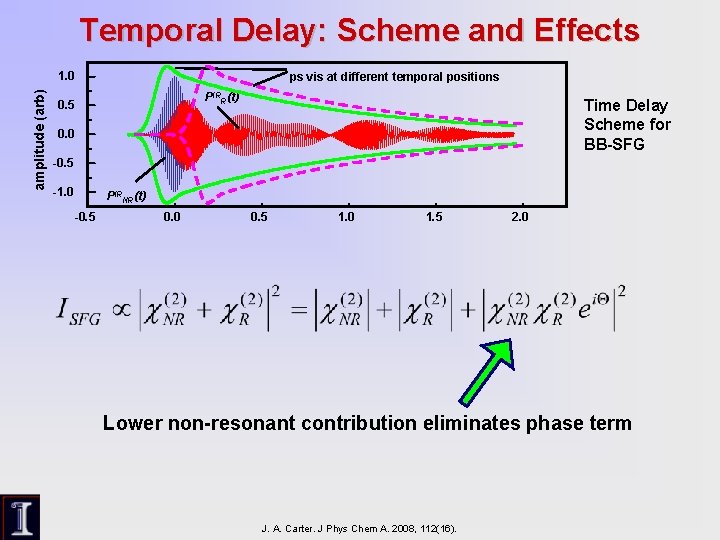
SFG Characteristics Parameters of resonant transitions are extracted by fitting SFG signal to the equation: Non-resonant background Gaussian IR profile Phase factor Contribution from resonant transitions

SFG Characteristics Varying magnitude of XNR is directly related to changing resonant line shapes • Energy of Incident Radiation • Substrate Material • Angle of Radiation Incidence • Media above sample (solvent) • Applied Potential • Azimuthal Rotation • Temporal Overlap Top: Octadecanethiol on Au in 532 and 1064 nm radiation Bottom: Octadecanethiol on Ag in 532 and 1064 nm radiation Potterton. Bain. J. Electroanal Chem. (409) 1996.

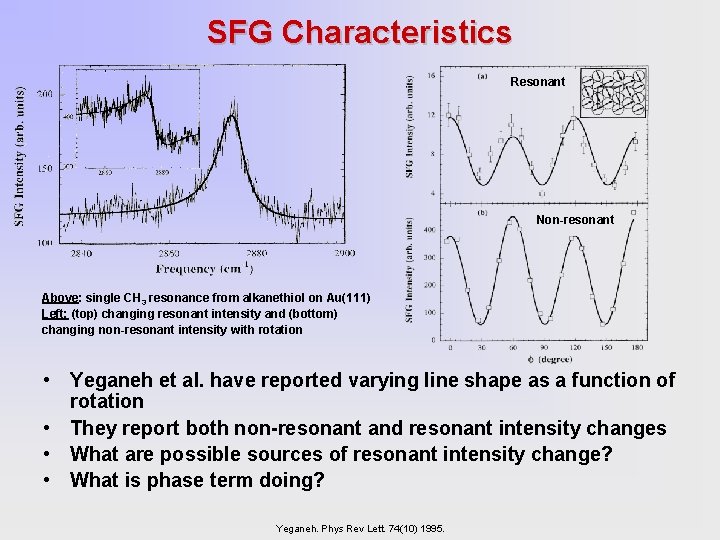
SFG Characteristics Resonant Non-resonant Above: single CH 3 resonance from alkanethiol on Au(111) Left: (top) changing resonant intensity and (bottom) changing non-resonant intensity with rotation • Yeganeh et al. have reported varying line shape as a function of rotation • They report both non-resonant and resonant intensity changes • What are possible sources of resonant intensity change? • What is phase term doing? Yeganeh. Phys Rev Lett. 74(10) 1995.

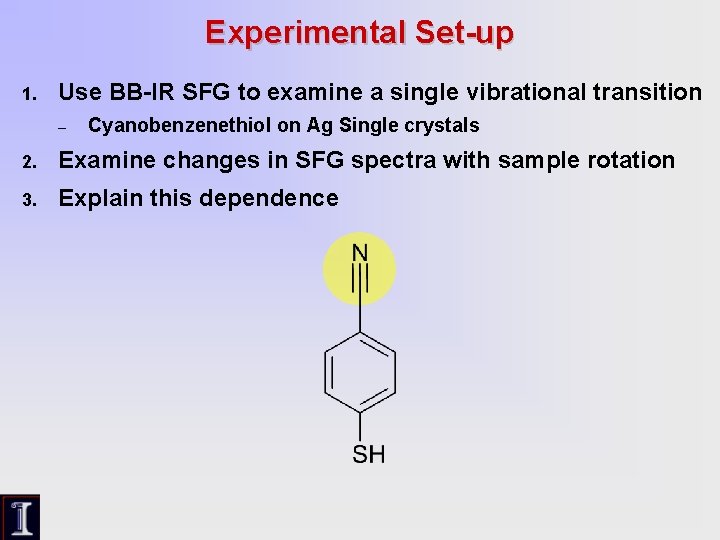
Experimental Set-up 1. Use BB-IR SFG to examine a single vibrational transition – Cyanobenzenethiol on Ag Single crystals 2. Examine changes in SFG spectra with sample rotation 3. Explain this dependence

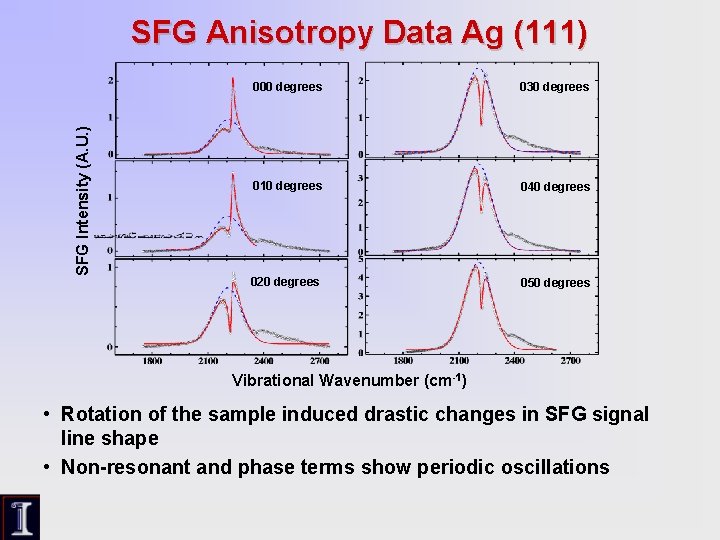
SFG Intensity (A. U. ) SFG Anisotropy Data Ag (111) 000 degrees 030 degrees 010 degrees 040 degrees 020 degrees 050 degrees Vibrational Wavenumber (cm-1) • Rotation of the sample induced drastic changes in SFG signal line shape • Non-resonant and phase terms show periodic oscillations

SFG Anisotropy Data Ag(110) SFG Intensity (A. U. ) 000 degrees 060 degrees 020 degrees 080 degrees 040 degrees 100 degrees Vibrational Wavenumber (cm-1) • Rotation of the sample induced drastic changes in SFG signal line shape • Non-resonant and phase terms show periodic oscillations

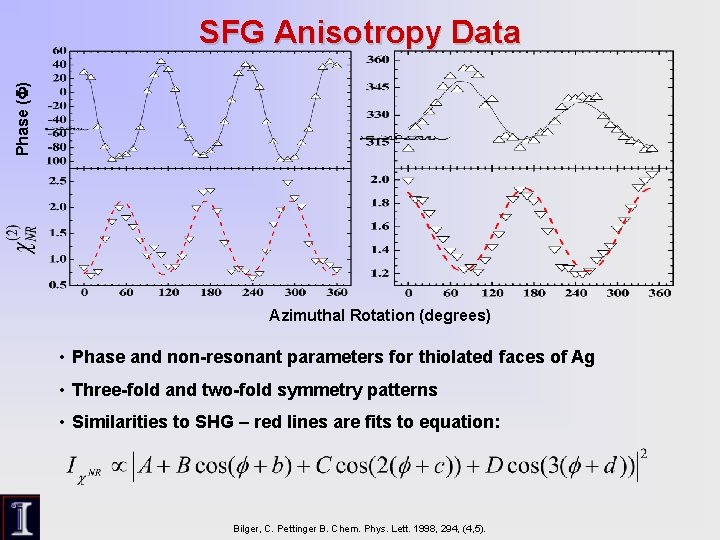
Phase (F) SFG Anisotropy Data Azimuthal Rotation (degrees) • Phase and non-resonant parameters for thiolated faces of Ag • Three-fold and two-fold symmetry patterns • Similarities to SHG – red lines are fits to equation: Bilger, C. Pettinger B. Chem. Phys. Lett. 1998, 294, (4, 5).

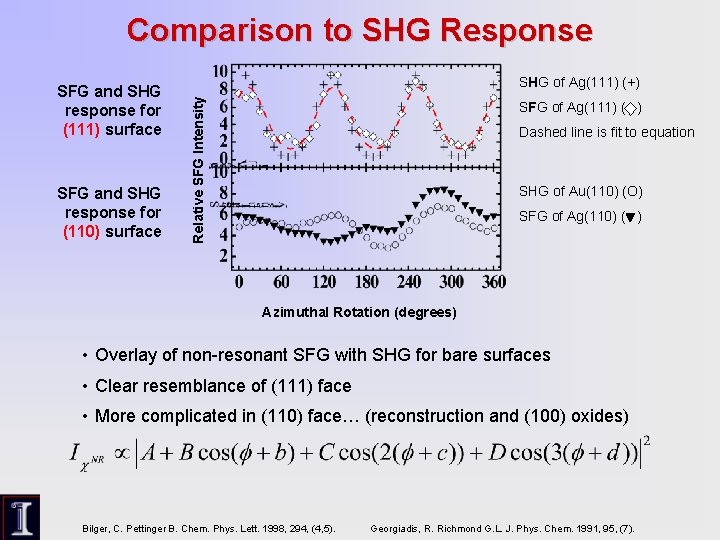
Comparison to SHG Response SFG and SHG response for (110) surface SHG of Ag(111) (+) Relative SFG Intensity SFG and SHG response for (111) surface SFG of Ag(111) ( ) Dashed line is fit to equation SHG of Au(110) (O) SFG of Ag(110) ( ) Azimuthal Rotation (degrees) • Overlay of non-resonant SFG with SHG for bare surfaces • Clear resemblance of (111) face • More complicated in (110) face… (reconstruction and (100) oxides) Bilger, C. Pettinger B. Chem. Phys. Lett. 1998, 294, (4, 5). Georgiadis, R. Richmond G. L. J. Phys. Chem. 1991, 95, (7).

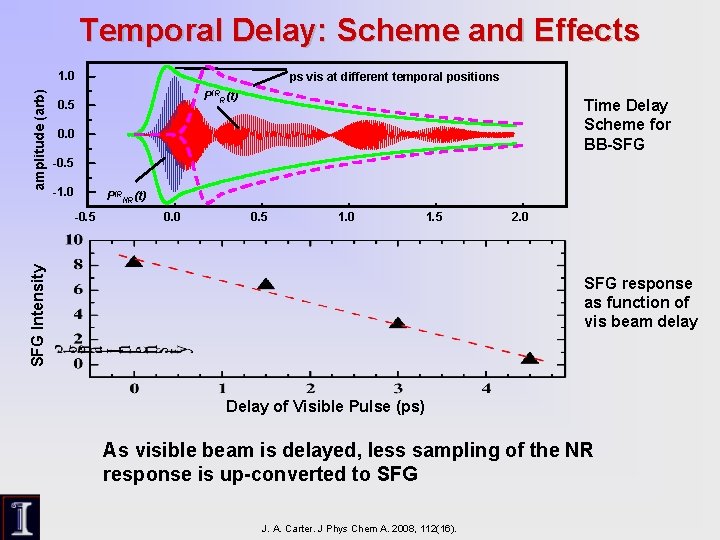
Temporal Delay: Scheme and Effects amplitude (arb) 1. 0 ps vis at different temporal positions PIRR(t) 0. 5 Time Delay Scheme for BB-SFG 0. 0 -0. 5 -1. 0 PIRNR(t) 0. 0 0. 5 1. 0 1. 5 SFG Intensity -0. 5 2. 0 SFG response as function of vis beam delay Delay of Visible Pulse (ps) As visible beam is delayed, less sampling of the NR response is up-converted to SFG J. A. Carter. J Phys Chem A. 2008, 112(16).

Temporal Delay: Scheme and Effects amplitude (arb) 1. 0 ps vis at different temporal positions PIRR(t) 0. 5 Time Delay Scheme for BB-SFG 0. 0 -0. 5 -1. 0 PIRNR(t) -0. 5 0. 0 0. 5 1. 0 1. 5 2. 0 Lower non-resonant contribution eliminates phase term J. A. Carter. J Phys Chem A. 2008, 112(16).

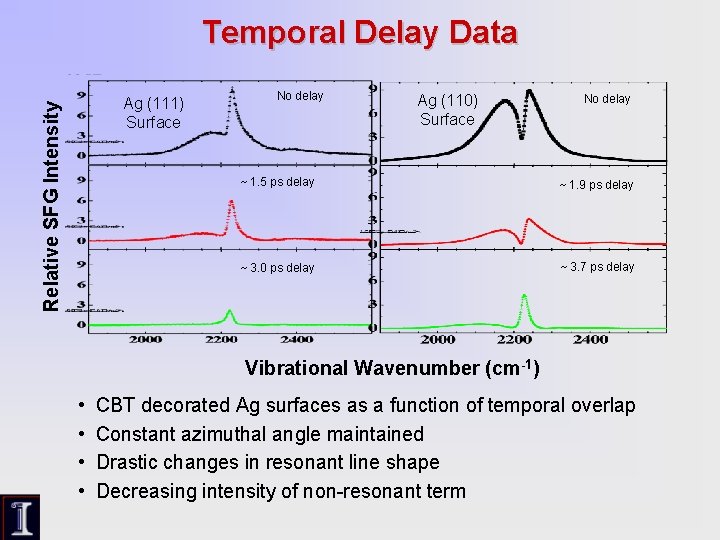
Temporal Delay Data Relative SFG Intensity Ag (111) Surface No delay Ag (110) Surface No delay ~ 1. 5 ps delay ~ 1. 9 ps delay ~ 3. 0 ps delay ~ 3. 7 ps delay Vibrational Wavenumber (cm-1) • • CBT decorated Ag surfaces as a function of temporal overlap Constant azimuthal angle maintained Drastic changes in resonant line shape Decreasing intensity of non-resonant term

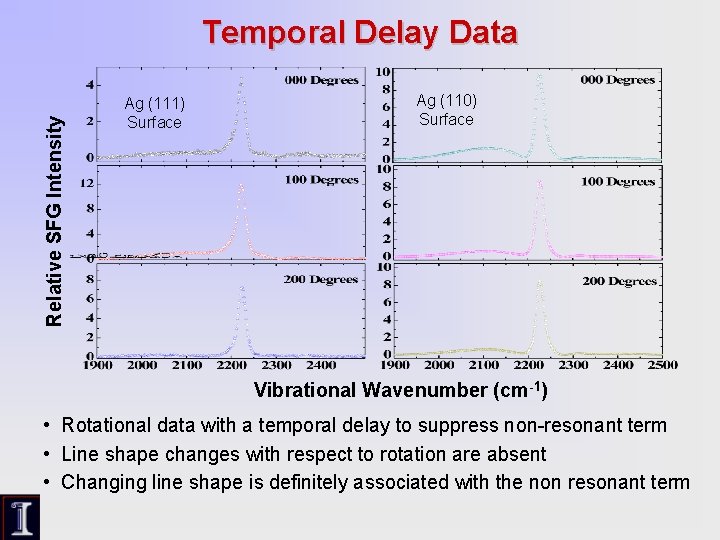
Relative SFG Intensity Temporal Delay Data Ag (111) Surface Ag (110) Surface Vibrational Wavenumber (cm-1) • Rotational data with a temporal delay to suppress non-resonant term • Line shape changes with respect to rotation are absent • Changing line shape is definitely associated with the non resonant term

Conclusions: • SFG from single crystal surfaces is azimuthally dependent • Can minimize non-resonant response to simply resonant line shapes • Allows more consistent approach to future vibrational SFG studies • Will simplify analysis of SFG spectra • Explains discrepancies in previous data

Acknowledgements: • Jonathan Arambula (synthesis of CBT) • Alexi Lagutchev, Dana Dlott, Andrew Gewirth* • Mauro Sardela (X-ray work) • – DMR 050438 – CHE-06 -03675 • Air Force Office of Scientific Research – FA 9550 -06 -1 -0235