ReSOLVE antibiotic resistance initiative Antimicrobial resistance in ophthalmology

Re-SOLVE antibiotic resistance initiative Antimicrobial resistance in ophthalmology – an urgent global healthcare priority 1 1. Antibacterial resistance in ophthalmic infections: a multi-centre analysis across UK care settings//BMC Infectious Diseases. volume 19, Article number: 768 (2019) EDUCATIONAL MODULE NOVEMBER 2020 NP-FOTE-EMEA-0186
Contents Antimicrobial resistance in ophthalmology Development of antibiotic resistance Classification and pharmacology of antibiotics Principles and challenges of modern antibiotic therapy
01 Antimicrobial resistance in ophthalmology

What are antibiotics? • Antibiotics (from ‘anti’ and ‘bios’. i. e. ‘life’ in Greek) are chemotherapeutic agents with the ability to destroy or prevent the growth of pathogens • Antibiotics can be formed by live microorganisms or obtained by artificial synthesis 1 • Commonly used antibiotics for the prevention of eye infections include: cefuroxime, cefazolin, vancomycin, gentamicin, tobramycin and moxifloxacin 2 1. Mandell: Principles and Practice of Infectious Diseases, 5 th ed. , 2000 Churchill Livingstone, Inc; 2. Barry P, et al. ESCRS Guidelines for Prevention and Treatment of Endophthalmitis Following Cataract Surgery: Data, Dilemmas and Conclusions; 2013. Available from: https: //www. escrs. org/downloads/Endophthalmitis-Guidelines. pdf. Accessed January 2020.

What is antimicrobial resistance? WHO definition: the ability of a microorganism to maintain vital functions, including reproduction, despite contact with a drug that was once effective against that microorganism 1 Resistance is a property of the microbe, not the infected organism 1 Antibiotic resistance is a subset of antimicrobial resistance, linked to pathogenic bacteria 2 WHO, World Health Organization 1. WHO 2014. Antimicrobial resistance. Available from: https: //www. who. int/en/news-room/fact-sheets/detail/antimicrobial-resistance. Accessed January 2020; 2. Mac. Gowan A, Macnaughton E. Medicine 2017; 45(10): 622– 628.
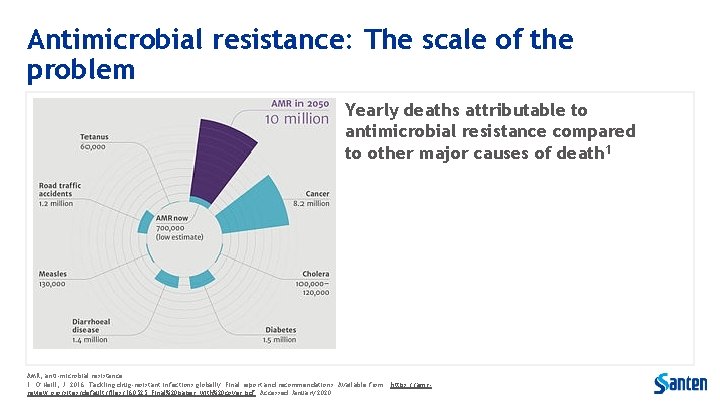
Antimicrobial resistance: The scale of the problem Yearly deaths attributable to antimicrobial resistance compared to other major causes of death 1 AMR, anti-microbial resistance 1. O’Neill, J. 2016. Tackling drug-resistant infections globally: Final report and recommendations. Available from: https: //amrreview. org/sites/default/files/160525_Final%20 paper_with%20 cover. pdf. Accessed January 2020.

A public health challenge Antimicrobial resistance is a major threat to public health worldwide 1, 2 Over 600, 000 infections caused by antibiotic-resistant bacteria in Europe in 20153 The burden of infections and infection-related deaths has increased from 20073 1. World Health Organization. Antimicrobial resistance. Available from: https: //www. who. int/en/news-room/fact-sheets/detail/antimicrobial-resistance. Accessed January 2020; 2. World Health Organization, 2015. Worldwide country situation analysis: response to antimicrobial resistance. Available from: https: //www. who. int/drugresistance/documents/situationanalysis/en/. Accessed January 2020; 3. Cassini A, et al. Lancet Infect Dis 2019; 19: 56– 66.

Bacterial pathogens: Gram-positive bacteria Dark blue or violet color by Gram staining 1 Diseases caused: 2 • • • Common pathogens responsible for eye infections: 2 • • Streptococcus pneumoniae (anaerobe) Staphylococcus aureus (facultative anaerobe) 1. Brown RC and Hopps HC. AJCP 1973; 59: 234– 340; 2. Mandell: Principles and Practice of Infectious Diseases, 5 th ed. , 2000 Churchill Livingstone, Inc. Pneumonia Meningitis Otitis media Eye infections Abscess Food poisoning

Bacterial pathogens: Gram-negative bacteria Pink color by Gram staining 1 Common pathogens responsible for eye infections: 2 • • Moraxella catarrhalis (aerobe) Klebsiella pneumoniae (facultative anaerobe) Pseudomonas aeruginosa (obligate anaerobe) Haemophilus influenzae (facultative anaerobe) 1. Brown RC and Hopps HC. AJCP 1973; 59: 234– 340; 2. Mandell: Principles and Practice of Infectious Diseases, 5 th ed. , 2000 Churchill Livingstone, Inc. Diseases caused: 2 • • • Otitis media Respiratory infections Eye infections Endocarditis Meningitis Urinary tract infections Bacteraemia Burn wound infections Nosocomial opportunistic infections
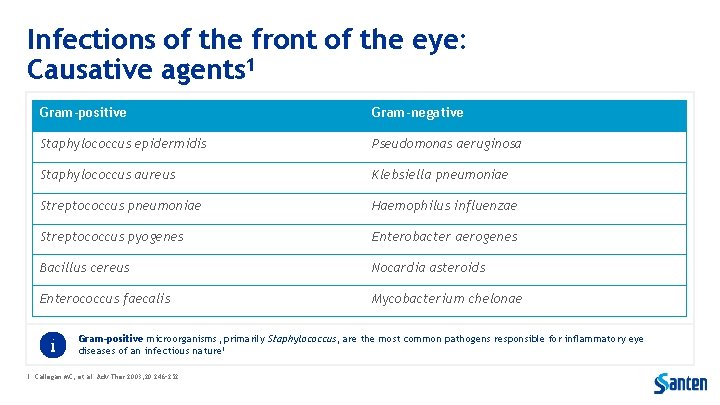
Infections of the front of the eye: Causative agents 1 Gram-positive Gram-negative Staphylococcus epidermidis Pseudomonas aeruginosa Staphylococcus aureus Klebsiella pneumoniae Streptococcus pneumoniae Haemophilus influenzae Streptococcus pyogenes Enterobacter aerogenes Bacillus cereus Nocardia asteroids Enterococcus faecalis Mycobacterium chelonae Gram-positive microorganisms, primarily Staphylococcus, are the most common pathogens responsible for inflammatory eye diseases of an infectious nature 1 1. Callegan MC, et al. Adv Ther 2003; 20: 246– 252.
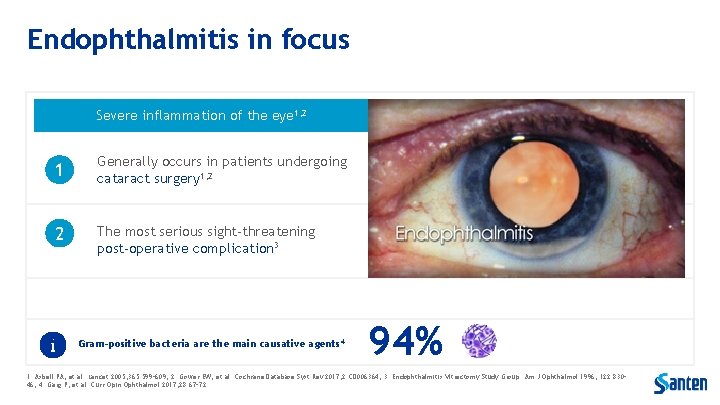
Endophthalmitis in focus Severe inflammation of the eye 1, 2 1 Generally occurs in patients undergoing cataract surgery 1, 2 2 The most serious sight-threatening post-operative complication 3 Gram-positive bacteria are the main causative agents 4 94% 1. Asbell PA, et al. Lancet 2005; 365: 599– 609; 2. Gower EW, et al. Cochrane Database Syst Rev 2017; 2: CD 006364; 3. Endophthalmitis Vitrectomy Study Group. Am J Ophthalmol 1996; 122: 830– 46; 4. Garg P, et al. Curr Opin Ophthalmol 2017; 28: 67– 72.

02 Development of antibiotic resistance

Causes of antibiotic resistance • Antibiotic resistance develops naturally, primarily through gene mutations 1 • However, the modern over-use of antibiotics has accelerated the problem 2, 3 Source: www. who. int/drugresistance 1. D’Costa VM, et al. Nature 2011; 477: 457– 461; 2. Cassini A, et al. Lancet Infect Dis 2019; 19: 56– 66; 3. Barry P, et al. ESCRS Guidelines for Prevention and Treatment of Endophthalmitis Following Cataract Surgery: Data, Dilemmas and Conclusions; 2013. Available from: https: //www. escrs. org/downloads/Endophthalmitis-Guidelines. pdf. Accessed January 2020.
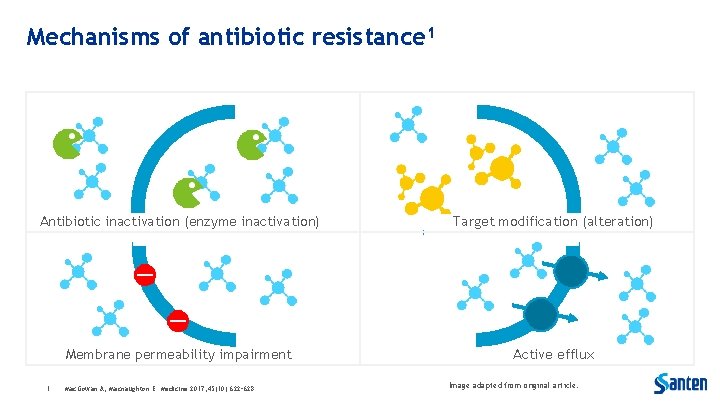
Mechanisms of antibiotic resistance 1 Antibiotic inactivation (enzyme inactivation) Target modification (alteration) Membrane permeability impairment Active efflux 1. Mac. Gowan A, Macnaughton E. Medicine 2017; 45(10): 622– 628. Image adapted from original article.

1. Antibiotic inactivation by enzymes 1 Examples: - hydrolysis of β-lactam antibiotics by β-lactamases - inactivation of aminoglycosides by aminoglycoside modifying enzymes 1. Mac. Gowan A, Macnaughton E. Medicine 2017; 45(10): 622– 628. Image adapted from original article.
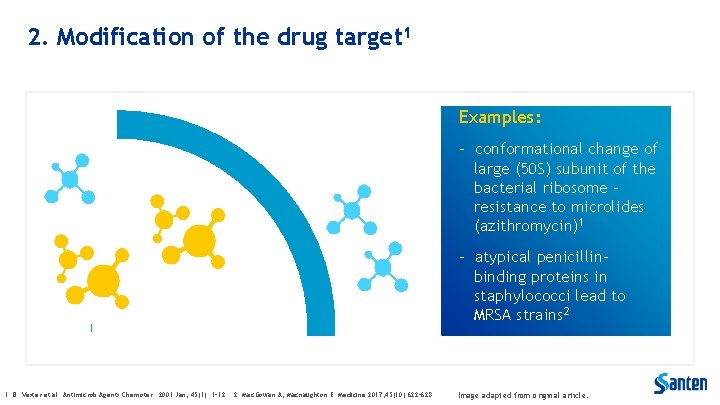
2. Modification of the drug target 1 Examples: - conformational change of large (50 S) subunit of the bacterial ribosome resistance to microlides (azithromycin)1 - atypical penicillinbinding proteins in staphylococci lead to MRSA strains 2 1. B. Vester et al. Antimicrob Agents Chemoter. 2001 Jan; 45(1): 1– 12. 2. Mac. Gowan A, Macnaughton E. Medicine 2017; 45(10): 622– 628. Image adapted from original article.
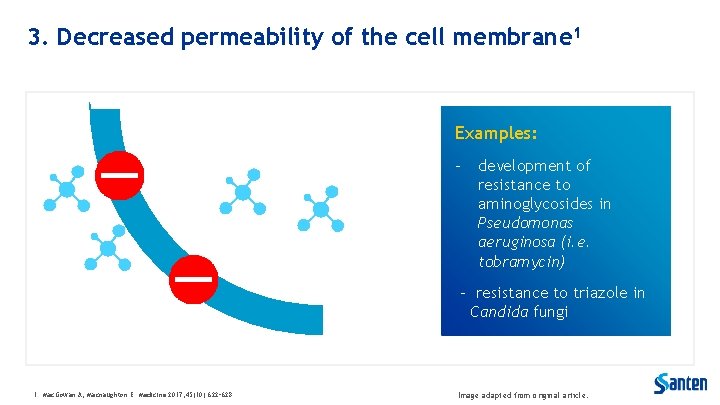
3. Decreased permeability of the cell membrane 1 Examples: - development of resistance to aminoglycosides in Pseudomonas aeruginosa (i. e. tobramycin) - resistance to triazole in Candida fungi 1. Mac. Gowan A, Macnaughton E. Medicine 2017; 45(10): 622– 628. Image adapted from original article.
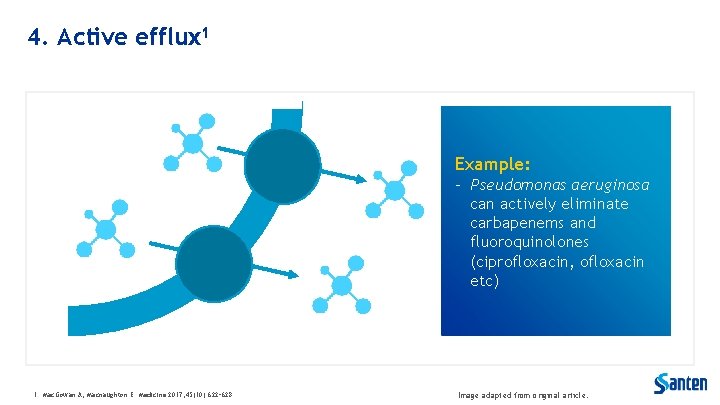
4. Active efflux 1 Example: - Pseudomonas aeruginosa can actively eliminate carbapenems and fluoroquinolones (ciprofloxacin, ofloxacin etc) 1. Mac. Gowan A, Macnaughton E. Medicine 2017; 45(10): 622– 628. Image adapted from original article.
03 Classification and pharmacology of antibiotics
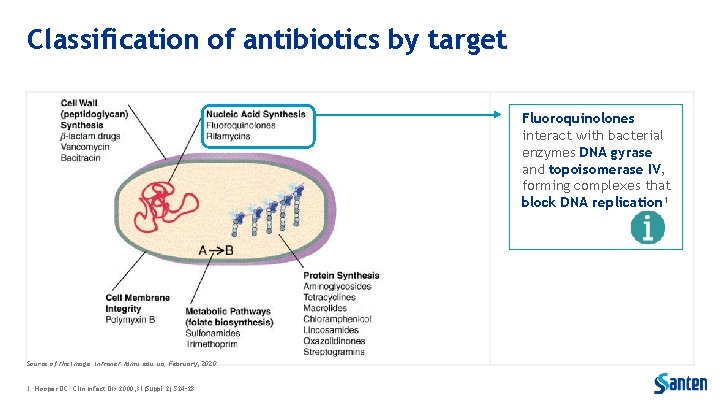
Classification of antibiotics by target Fluoroquinolones interact with bacterial enzymes DNA gyrase and topoisomerase IV, forming complexes that block DNA replication 1 Source of the image: intranet. tdmu. edu. ua; February, 2020. 1. Hooper DC. Clin Infect Dis 2000; 31(Suppl 2): S 24– 28.

Types of antibiotic activity • Bacteriostatic: 1 antibiotics inhibit the growth and reproduction of microbial cells, but do not cause cell death o Tetracyclines, lincomycin, chloramphenicol • Bactericidal: 2 antibiotics bind irreversibly to the cellular target, killing the microorganism o Cephalosporins, aminoglycosides, fluoroquinolones 1. Gower EW, et al. Cochrane Database Syst Rev 2017; 2: CD 006364; 2. Van Bambeke F, et al. Clin Microbiol Infect 2005; 11: 256– 280.

Antibiotics and spectrum of action 1 • Broad spectrum: fluoroquinolones, levomycetin, tetracyclines and cephalosporins (2 nd and 3 rd generations) • Narrow spectrum: Colistin (Gr-) etc TB, tuberculosis 1. Coleman, JP and Smith, CJ. Microbial Classification. 2007 Elsevier Inc. • Anti-TB: streptomycin, rifampicin, florimycin • Anti-fungal: nystatin, levorin, griseofulvin, amphotericin B
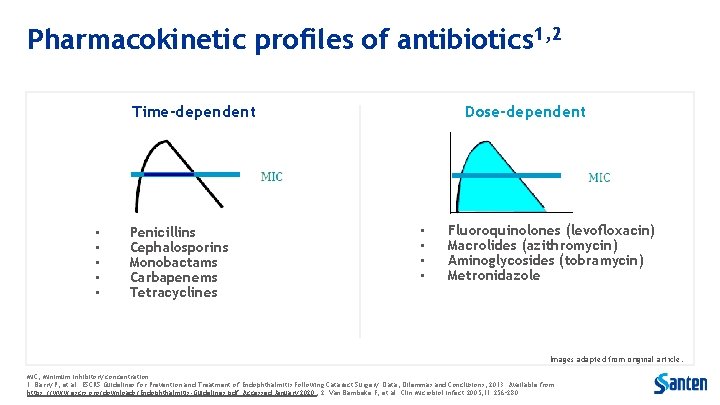
Pharmacokinetic profiles of antibiotics 1, 2 Time-dependent • • • Penicillins Cephalosporins Monobactams Carbapenems Tetracyclines Dose-dependent • • Fluoroquinolones (levofloxacin) Macrolides (azithromycin) Aminoglycosides (tobramycin) Metronidazole Images adapted from original article. MIC, Minimum inhibitory concentration. 1. Barry P, et al. ESCRS Guidelines for Prevention and Treatment of Endophthalmitis Following Cataract Surgery: Data, Dilemmas and Conclusions; 2013. Available from: https: //www. escrs. org/downloads/Endophthalmitis-Guidelines. pdf. Accessed January 2020 ; 2. Van Bambeke F, et al. Clin Microbiol Infect 2005; 11: 256– 280.
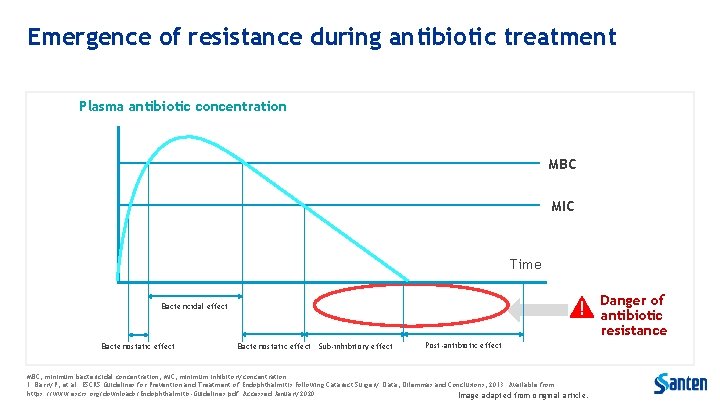
Emergence of resistance during antibiotic treatment Plasma antibiotic concentration MBC MIC Time Danger of antibiotic resistance Bactericidal effect Bacteriostatic effect Sub-inhibitory effect Post-antibiotic effect MBC, minimum bactericidal concentration; MIC, minimum inhibitory concentration. 1. Barry P, et al. ESCRS Guidelines for Prevention and Treatment of Endophthalmitis Following Cataract Surgery: Data, Dilemmas and Conclusions; 2013. Available from: https: //www. escrs. org/downloads/Endophthalmitis-Guidelines. pdf. Accessed January 2020. Image adapted from original article.

04 Principles and challenges of modern antibiotic therapy

Perspectives on modern antibiotic therapy Treatment challenges 1, 2 Clinical guidelines 2, 3 • Increased spread of resistant strains • Changes in the spectrum of pathogens • Heterogeneous approach to the prophylaxis and treatment of microbial infections • Increase in the number of generic antibiotic drugs with unconfirmed efficacy • The latest clinical recommendations acknowledge the growing problem of antibiotic resistance • A rational use of antibiotics is suggested, with reduced treatment duration to prevent the development of resistance There is currently a need for antibiotics with proven efficacy against a range of pathogens, which allow for shorter treatment duration to avoid the development of antimicrobial resistance 1. World Health Organization. Antimicrobial resistance. Available from: https: //www. who. int/en/news-room/fact-sheets/detail/antimicrobial-resistance. Accessed January 2020; 2. World Health Organization, 2015. Worldwide country situation analysis: response to antimicrobial resistance. Available at: Global Guidelines for the Prevention of Surgical Site Infections. WHO 2018. Available from: https: //apps. who. int/iris/bitstream/handle/10665/277399/9789241550475 -eng. pdf. Accessed January 2020; 3. Barry P, et al. ESCRS Guidelines for Prevention and Treatment of Endophthalmitis Following Cataract Surgery: Data, Dilemmas and Conclusions; 2013. Available from: https: //www. escrs. org/downloads/Endophthalmitis-Guidelines. pdf. Accessed January 2020.
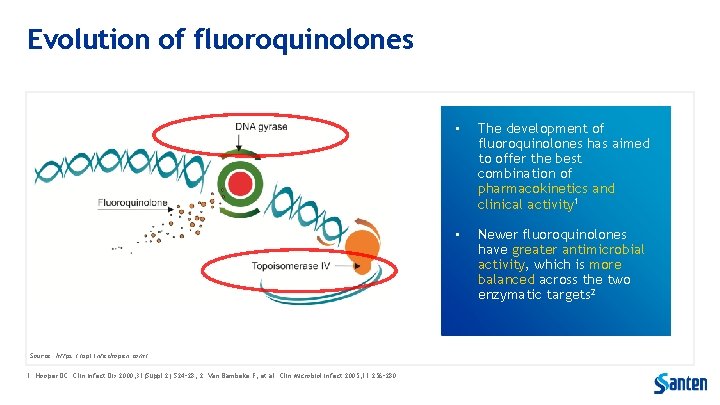
Evolution of fluoroquinolones Source: https: //api. intechopen. com/ 1. Hooper DC. Clin Infect Dis 2000; 31(Suppl 2): S 24– 28; 2. Van Bambeke F, et al. Clin Microbiol Infect 2005; 11: 256– 280. • The development of fluoroquinolones has aimed to offer the best combination of pharmacokinetics and clinical activity 1 • Newer fluoroquinolones have greater antimicrobial activity, which is more balanced across the two enzymatic targets 2
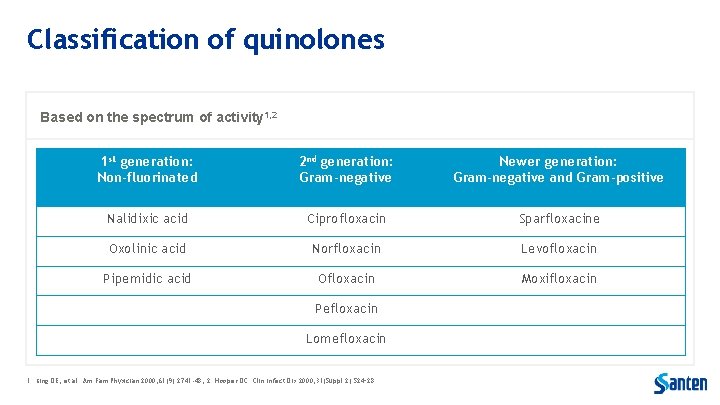
Classification of quinolones Based on the spectrum of activity 1, 2 1 st generation: Non-fluorinated 2 nd generation: Gram-negative Newer generation: Gram-negative and Gram-positive Nalidixic acid Ciprofloxacin Sparfloxacine Oxolinic acid Norfloxacin Levofloxacin Pipemidic acid Ofloxacin Moxifloxacin Pefloxacin Lomefloxacin 1. King DE, et al. Am Fam Physician 2000; 61(9): 2741 -48; 2. Hooper DC. Clin Infect Dis 2000; 31(Suppl 2): S 24– 28.

Levofloxacin: A newer generation fluoroquinolone 1 Levofloxacin is a L-enantiomer of ofloxacin 2 Greater activity than older -generation fluoroquinolones 1– 3 Levofloxacin can address the requirements of a modern antibiotic Broad spectrum of action against Gram-positive and Gram-negative bacteria 1, 2, 4 1. Liu HH. Drug Saf 2010; 33: 353– 69; 2. Schwab IR, et al. Ophthalmology 2003; 110: 457– 65; 3. Norrby SR. Expert Opin Pharmacother 1999; 1: 109– 19; 4. Kanda Y, et al. Drugs R D 2012; 12: 177– 85. High water solubility at a neutral p. H, allowing for high concentrations 4
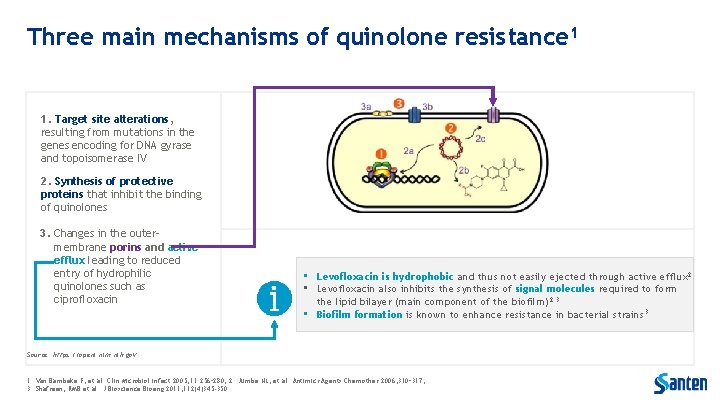
Three main mechanisms of quinolone resistance 1 1. Target site alterations, resulting from mutations in the genes encoding for DNA gyrase and topoisomerase IV 2. Synthesis of protective proteins that inhibit the binding of quinolones 3. Changes in the outermembrane porins and active efflux leading to reduced entry of hydrophilic quinolones such as ciprofloxacin • Levofloxacin is hydrophobic and thus not easily ejected through active efflux 2 • Levofloxacin also inhibits the synthesis of signal molecules required to form the lipid bilayer (main component of the biofilm)2. 3 • Biofilm formation is known to enhance resistance in bacterial strains 3 Source: https: //openi. nlm. nih. gov 1. Van Bambeke F, et al. Clin Microbiol Infect 2005; 11: 256– 280; 2. Jumbe NL, et al. Antimicr Agents Chemother 2006; 310– 317; 3. Shafreen, RMB et al. J Bioscience Bioeng 2011; 112(4)345 -350.
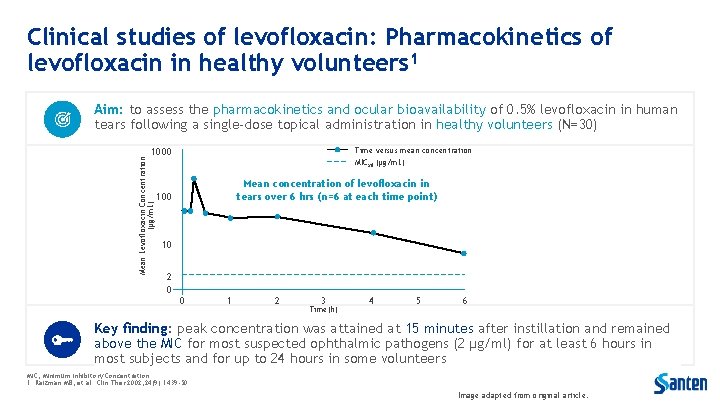
Clinical studies of levofloxacin: Pharmacokinetics of levofloxacin in healthy volunteers 1 Aim: to assess the pharmacokinetics and ocular bioavailability of 0. 5% levofloxacin in human tears following a single-dose topical administration in healthy volunteers (N=30) Time versus mean concentration Mean Levofloxacin Concentration (μg/m. L) 1000 MICx 0 (μg/m. L) Mean concentration of levofloxacin in tears over 6 hrs (n=6 at each time point) 100 10 2 0 0 1 2 3 Time(h) 4 5 6 Key finding: peak concentration was attained at 15 minutes after instillation and remained above the MIC for most suspected ophthalmic pathogens (2 μg/ml) for at least 6 hours in most subjects and for up to 24 hours in some volunteers MIC, Minimum Inhibitory Concentration 1. Raizman MB, et al. Clin Ther 2002; 24(9): 1439 -50. Image adapted from original article.
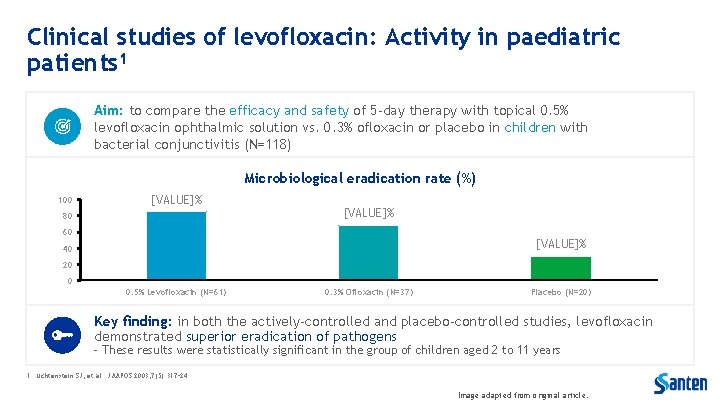
Clinical studies of levofloxacin: Activity in paediatric patients 1 Aim: to compare the efficacy and safety of 5 -day therapy with topical 0. 5% levofloxacin ophthalmic solution vs. 0. 3% ofloxacin or placebo in children with bacterial conjunctivitis (N=118) Microbiological eradication rate (%) 100 [VALUE]% 80 [VALUE]% 60 [VALUE]% 40 20 0 0. 5% Levofloxacin (N=61) 0. 3% Ofloxacin (N=37) Placebo (N=20) Key finding: in both the actively-controlled and placebo-controlled studies, levofloxacin demonstrated superior eradication of pathogens - These results were statistically significant in the group of children aged 2 to 11 years 1. Lichtenstein SJ, et al. J AAPOS 2003; 7(5): 317– 24. Image adapted from original article.
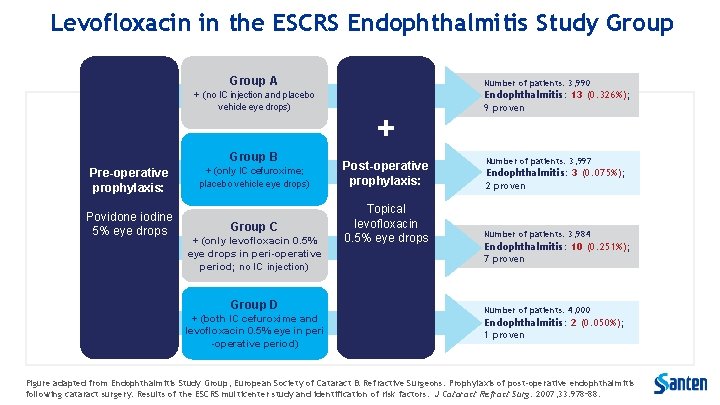
Levofloxacin in the ESCRS Endophthalmitis Study Group A + (no IC injection and placebo vehicle eye drops) Post-operative prophylaxis: Group B Pre-operative prophylaxis: Povidone iodine 5% eye drops Number of patients: 3, 990 + + (only IC cefuroxime; placebo vehicle eye drops) Post-operative prophylaxis: Group C Topical levofloxacin 0. 5% eye drops + (only levofloxacin 0. 5% eye drops in peri-operative period; no IC injection) Group D + (both IC cefuroxime and levofloxacin 0. 5% eye in peri -operative period) Endophthalmitis: 13 (0. 326%); 9 proven Number of patients: 3, 997 Endophthalmitis: 3 (0. 075%); 2 proven Number of patients: 3, 984 Endophthalmitis: 10 (0. 251%); 7 proven Number of patients: 4, 000 Endophthalmitis: 2 (0. 050%); 1 proven Figure adapted from Endophthalmitis Study Group, European Society of Cataract & Refractive Surgeons. Prophylaxis of post-operative endophthalmitis following cataract surgery: Results of the ESCRS multicenter study and identification of risk factors. J Cataract Refract Surg. 2007; 33: 978– 88.

Levofloxacin in the ESCRS Endophthalmitis Study Group 1 Aim: to investigate the role of antibiotics in preventing endophthalmitis in patients undergoing phacoemulsification cataract surgery Key finding: in this large, multi-centre, controlled study, patients who received levofloxacin eye drops perioperatively had a lower risk of developing endophthalmitis than those who received placebo* • Use of intracameral cefuroxime at the end of surgery reduced the occurrence of post-operative endophthalmitis • Incidence rates of endophthalmitis were 0. 049% (presumed) and 0. 025% (proven) in the group receiving intra-cameral cefuroxime and peri-operative topical levofloxacin • These rates were the lowest of the 4 treatment groups ESCRS, European Society of Cataract and Refractive Surgeons 1. ESCRS Endophthalmitis Study Group. J Cataract Refract Surg 2007; 33: 978 -988.

Summary: Key learning points Antimicrobial resistance is a major threat to public health worldwide, 1, 2 and its burden is increasing 3 Gram-positive bacteria, mainly Staphylococcus, are the most common pathogens responsible for eye infections, 4 including endophthalmitis 5 While resistance to antibiotics develops naturally, 6 the modern over-use of antibiotic drugs has contributed to the problem 3 The development of quinolones has optimised their pharmacokinetics and clinical activity, while reducing toxicity and the risk of resistance 7, 8 Levofloxacin is a newer generation fluoroquinolone that can address the requirements of a modern antibiotic for broad spectrum of action, high efficacy and short treatment duration 9, 10 1. World Health Organization. Antimicrobial resistance. Available from: https: //www. who. int/en/news-room/fact-sheets/detail/antimicrobial-resistance. Accessed January 2020; 2. World Health Organization, 2015. Worldwide country situation analysis: response to antimicrobial resistance. Available from: https: //www. who. int/drugresistance/documents/situationanalysis/en/. Accessed January 2020; 3. Cassini A, et al. Lancet Infect Dis 2019; 19: 56– 66; 4. Callegan MC, et al. Adv Ther 2003; 20: 246– 252; 5. Prashant G. Curr Opin Ophthalmol 2017, 28: 67 72; 6. D’Costa VM, et al. Nature 2011; 477: 457– 461; 7. Hooper DC. Clin Infect Dis 2000; 31(Suppl 2): S 24– 28; 8. Van Bambeke F, et al. Clin Microbiol Infect 2005; 11: 256– 280; 9. Schwab IR, et al. Ophthalmology 2003; 110: 457– 65; 10. Schwab IR, et al. Ophthalmology 2003; 110: 457– 65.
- Slides: 35