Resin lifetime By Franois de Dardel RESIN LIFETIME

Resin lifetime By François de Dardel

RESIN LIFETIME: HOW LONG WILL MY RESIN LAST ? • Question 1 : How long can I keep my resin before changing it ? • Question 2 : What resin life does R&H guarantee ? • Answer : Many parameters influence resin life © 2009 Dow Water & Process Solutions Lifetime 2

LIFETIME Factors influencing lifetime • • Type of stress Type of process Mechanical • Fluidised • Resin transfers (MBs, CPP) • Continuous ion exchange Suspended solids Mechanical Specific flow rate Mechanical Frequency of regeneration Osmotic Oxidants Chemical Foulants Chemical Temperature Thermal © 2009 Dow Water & Process Solutions Lifetime 3

PHYSICAL STABILITY External forces Internal forces purely mechanical osmotic Osmotic >> mechanical forces (by 10 to 100 times) © 2009 Dow Water & Process Solutions Lifetime 4

MECHANICAL STABILITY Breaking weight test (Chatillon test) Is a simple, straightforward test Measuring the force under which an individual bead breaks (60 beads) But meaningless It does not reflect operating conditions of resin © 2009 Dow Water & Process Solutions Lifetime 5
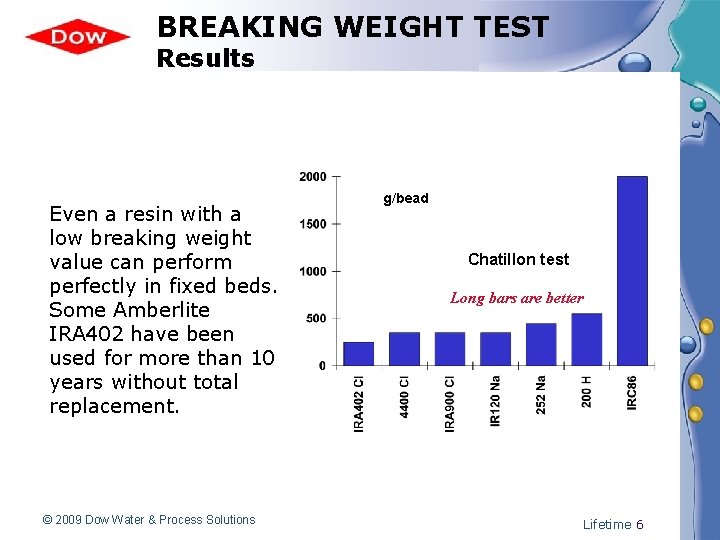
BREAKING WEIGHT TEST Results Even a resin with a low breaking weight value can perform perfectly in fixed beds. Some Amberlite IRA 402 have been used for more than 10 years without total replacement. © 2009 Dow Water & Process Solutions g/bead Chatillon test Long bars are better Lifetime 6
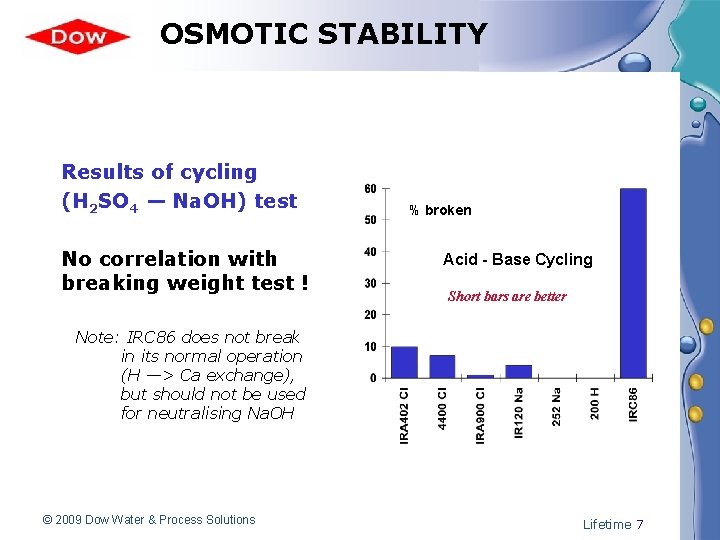
OSMOTIC STABILITY Results of cycling (H 2 SO 4 — Na. OH) test No correlation with breaking weight test ! % broken Acid - Base Cycling Short bars are better Note: IRC 86 does not break in its normal operation (H —> Ca exchange), but should not be used for neutralising Na. OH © 2009 Dow Water & Process Solutions Lifetime 7
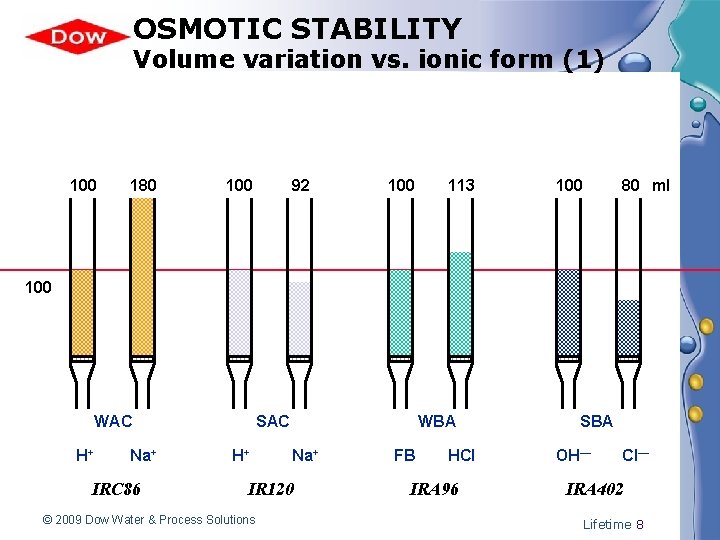
OSMOTIC STABILITY Volume variation vs. ionic form (1) 100 180 100 92 100 113 100 80 ml 100 WAC H+ Na+ IRC 86 SAC H+ WBA Na+ IR 120 © 2009 Dow Water & Process Solutions FB HCl IRA 96 SBA OH— Cl— IRA 402 Lifetime 8
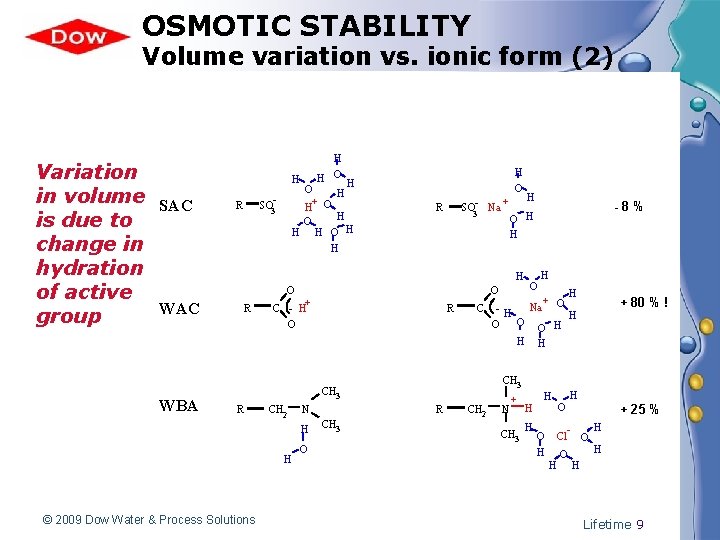
OSMOTIC STABILITY Volume variation vs. ionic form (2) Variation in volume SAC is due to change in hydration of active WAC group WBA H H O H H+ O H H SO 3 - R H O + SO 3 - Na R R O C - H+ O R C CH 2 H H © 2009 Dow Water & Process Solutions R N O CH 3 O H H + Na O -H H O O H H CH 3 R -8% O H H H O H CH 2 N + CH 3 H H H O + 80 % ! H + 25 % H Cl O H H Lifetime 9
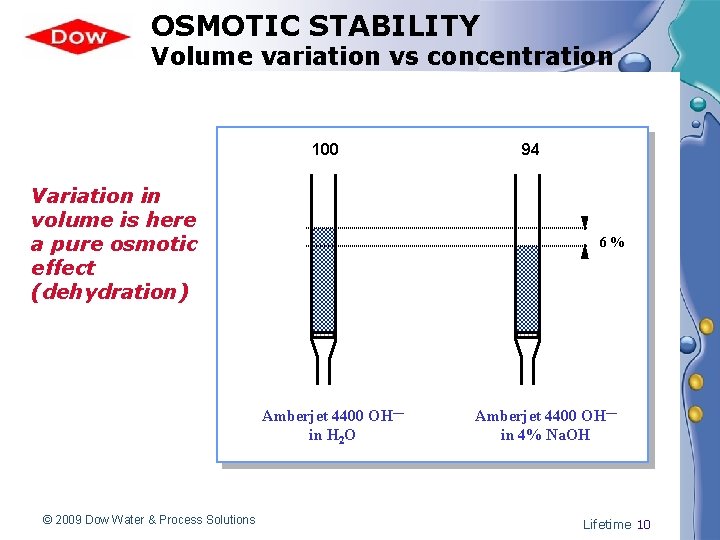
OSMOTIC STABILITY Volume variation vs concentration 100 Variation in volume is here a pure osmotic effect (dehydration) 6% Amberjet 4400 OH— in H 2 O © 2009 Dow Water & Process Solutions 94 Amberjet 4400 OH— in 4% Na. OH Lifetime 10

CHEMICAL STABILITY OXIDATION Cl 2, O 3, O 2 + worsening factors such as • metals ( catalyse oxidation ) • temperature ( accelerates reaction ) Matrix breaks down resin swells and gets soft More DVB = better resistance (macroporous resins are better) © 2009 Dow Water & Process Solutions Maximum acceptable chlorine ( in conventional applications ) IRC 86 1 mg/L HP 336 0. 1 mg/L IR 120 0. 2 mg/L Amberjet 1200 0. 2 mg/L Amberjet 1500 0. 3 mg/L Ambersep 200 0. 5 mg/L IRA 402 0. 1 mg/L Amberjet 4400 0. 15 mg/L IRA 96 0. 05 mg/L Lifetime 11

CHEMICAL CONTAMINATION FOULING • All resins • Oil, grease • Bacteria, algae • Cation resins • Na exchange: Fe, Mn • H exchange: Ca. SO 4 in case of H 2 SO 4 regeneration • Anion resins • Organics, silica © 2009 Dow Water & Process Solutions Lifetime 12

ORGANIC FOULING of Anion Resins Resistance to fouling • Type 1 poor • high affinity for organics • difficult to regenerate • thus high tendency to fouling • Type 2 better Natural organics foul Anion resins • better regenerability • thus less easily fouled • Acrylic good • aliphatic structure • no Van der Waals attraction • good elution of organics © 2009 Dow Water & Process Solutions Lifetime 13

FOULING INDEX N = Organics (ppm of KMn. O 4) Total anions ( meq/L) Resin N max A’Jet 4200 A’Jet 4600 IRA 402 IRA 410 IRA 458 IRA 478 IRA 900 IRA 910 IRA 96 IRA 67 4 6 15 15 6 10 12 20 © 2009 Dow Water & Process Solutions N measures the fouling risk of the water. The table shows the value under which a given resin can be used safely Lifetime 14

MAXIMUM ORGANIC LOAD Recommended maximum load per cycle in g/LR as KMn. O 4 Amberjet Amberlite 4200 4400 4600 9000 IRA 402 IRA 410 IRA 900 IRA 458 IRA 478 IRA 92 IRA 96 IRA 67 © 2009 Dow Water & Process Solutions max. g/LR 2 1 4 3 8 10 10 12 25 Organic load is the quantity of organics passing through the resin bed Lifetime 15

REMOVAL OF ORGANICS Resins do remove organics from water; however, resins are not designed for that purpose. No guarantee can be given for % removal. Approximate figures : Organic removal WBA 40 to 70 % SBA • Styrenic gel 20 to 60 % • Styrenic MR 50 to 100 % • Acrylic gel 40 to 80 % • Acrylic MR 60 to 100 % © 2009 Dow Water & Process Solutions Lifetime 16

ELUTION OF ORGANICS Approximate figures : Elution • WBA 50 to • SBA • Type 1 • Type 2 • Acrylic 100 % 30 to 70 % 60 to 95 % 90 to 100 % Fouling = Accumulation of organics when elution (during regeneration) is less than 100% Treatment for fouled resins: • Alkaline brine • 10 % Na. Cl + 2 % Na. OH • Hydrochloric acid • 10 % hot HCl © 2009 Dow Water & Process Solutions Lifetime 17

TEMPERATURE STABILITY • Frost: no measurable effect down to - 40°C • Allow resin to thaw at 10 to 30°C • Heat: cation OK up to 100°C • Beware of oxidants / metals can catalyse reaction • Heat: anion resins are sensitive • • • WBA styrenic up to 60 °C WBA acrylic up to 35 °C SBA type 1 up to 60 °C SBA type 2 up to 30 °C SBA acrylic up to 35 °C (higher temperatures possible in certain special applications) © 2009 Dow Water & Process Solutions Lifetime 18
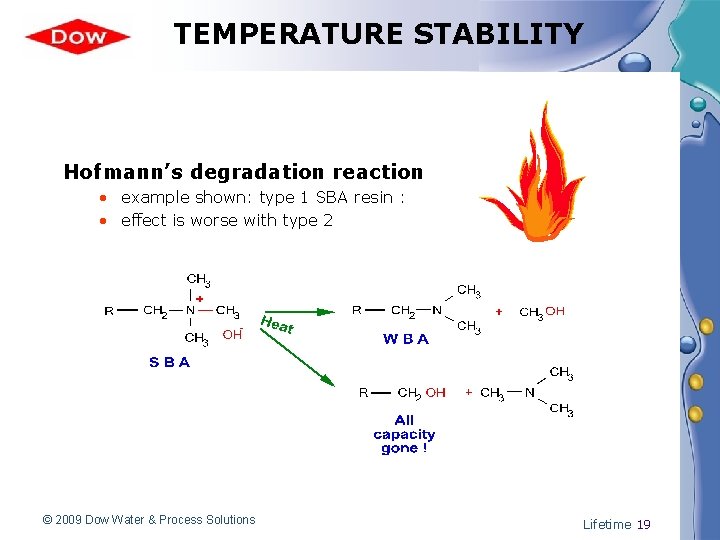
TEMPERATURE STABILITY Hofmann’s degradation reaction • example shown: type 1 SBA resin : • effect is worse with type 2 © 2009 Dow Water & Process Solutions Lifetime 19

CONCLUSION Nobody knows the date of resin death. . but Follow all recommendations, this will guarantee long life to your resins. © 2009 Dow Water & Process Solutions Lifetime 20
- Slides: 20