Research Using Large Databases Michael J Fisch MD

Research Using Large Databases Michael J. Fisch MD, MPH Professor and Chair, Department of General Oncology The University of Texas MD Anderson Cancer Center

Large Database Sources for Study • • • Large explanatory clinical trials Large pragmatic trials Observational (cohort) studies Administrative databases Cancer Registry data Survey studies

Other Data Sources • Physicians’ Professional Database • AMA/CMS • All practicing US physicians • Medical schools, GME training programs, state licensing agencies, ABMS, physician surveys • Information: demographics (age, gender), education, licensing, specialty training, hospital, location, type of practice • Area Resource File • Health facilities, health professions, measures of resources scarcity, etc
Large Database Research at #ASCOQLTY
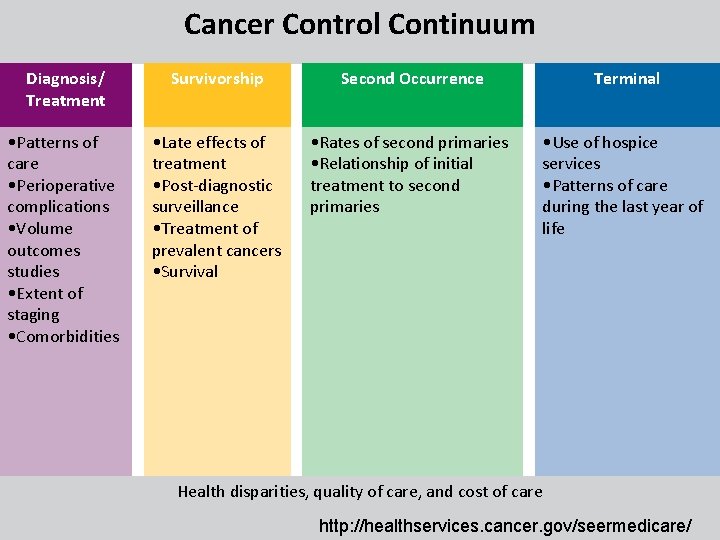
Cancer Control Continuum Diagnosis/ Treatment Survivorship • Patterns of • Late effects of care treatment • Perioperative • Post-diagnostic complications surveillance • Volume • Treatment of outcomes prevalent cancers studies • Survival • Extent of staging • Comorbidities Second Occurrence Terminal • Rates of second primaries • Relationship of initial treatment to second primaries • Use of hospice services • Patterns of care during the last year of life Health disparities, quality of care, and cost of care http: //healthservices. cancer. gov/seermedicare/

Pragmatic vs. Explanatory Trials “Can this intervention work under ideal conditions” Pragmatic Trials “Does this intervention work under usual conditions” Thorpe KE, CMAJ 2009

Patient-Centered Outcomes Research Institute (PCORI)


Selby JV, NEJM 2014

Population Survey Examples • National Health Interview Survey (NHIS, CDC) • National Health and Nutrition Examination Survey (NHANES, CDC) • Medicare Consumer Assessment of Health Plans (CAHPS) • Medicare Health Outcomes Survey (HOS)

Administrative (Claims) Data • Medicare/Medicaid data • All or subset of Medicare/Medicaid patients in US • Cases identified by claims algorithms • Expensive • Marketscan/Private Insurance Data • • • >180 million patients Younger population Prescription drug information Insurance/co-pay information Shorter follow-up

National Cancer Registries • SEER (NCI) • National Program of Cancer Registries (CDC) • National Cancer Data Base (Co. C of the ACOS)

Objectives of SEER • Designed for cancer surveillance - Monitoring trends in incidence, stage, survival, case fatality, first course of treatment - Population-based • Large dataset - 170, 000 cases/year, total # > 3 million - Limited variables for any individual • Diagnosis Dates 1973 -2010
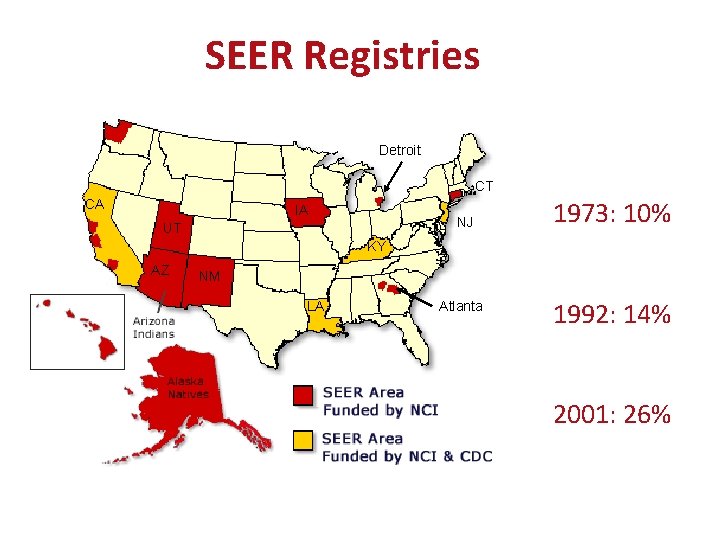
SEER Registries Detroit CT CA IA NJ UT 1973: 10% KY AZ NM LA Atlanta 1992: 14% 2001: 26% **Beware of time changes coincident with expansions**

Collected Data • 4 categories • • Demographic Tumor Treatment Mortality • No information on chemotherapy, oral drugs, recurrence • Data de-identified for public use • Linkages with Medicare Health Outcomes Survey (MHOS), Medicare, and National Longitudinal Mortality Study (NLMS)

The SEER/Medicare Database • Cooperative effort between: - National Cancer Institute - Surveillance, Epidemiology and End Results (SEER) program, and - Centers for Medicare and Medicaid (CMS) • Data linked from SEER and Medicare • 93% of eligible patients linked

Medicare Eligibility/Entitlement • Age: persons 65 years old and older who are eligible for any type of Social Security benefit • Disabled: persons who have received Social Security disability benefits for 24 months • ESRD: persons who end-stage renal disease

SEER-Medicare • Cancer diagnoses 1973 -2009, f/up 2010 • CLAIMS DATA • Includes SEER variables, PLUS • Inpatient bills from 1986+ • Outpatient, hospice, physician, home health, durable medical equipment bills from 1991 • Medicare Part D • Includes a 5% non-cancer control sample

SEER-Medicare • Can use claims to determine • Screening • Treatment, including procedures, diagnostic tests, chemotherapy • Complications of treatment • Comorbidities • Costs of therapy

Obtaining SEER-Medicare data • Not public use data, but are available to researchers • Specific permission required through NCI • These are large datasets and it often costs several thousand dollars to get the data you need • Example: to get the Patient Entitlement and Diagnosis Summary File (PEDSF) and hospice data for lung and colorectal patients over a 10 year period would cost around $7500. http: //appliedresearch. cancer. gov/seermedicare/obtain/

Administrative Data: National Patterns of Care

Dartmouth Atlas Project www. dartmouthatlas. org
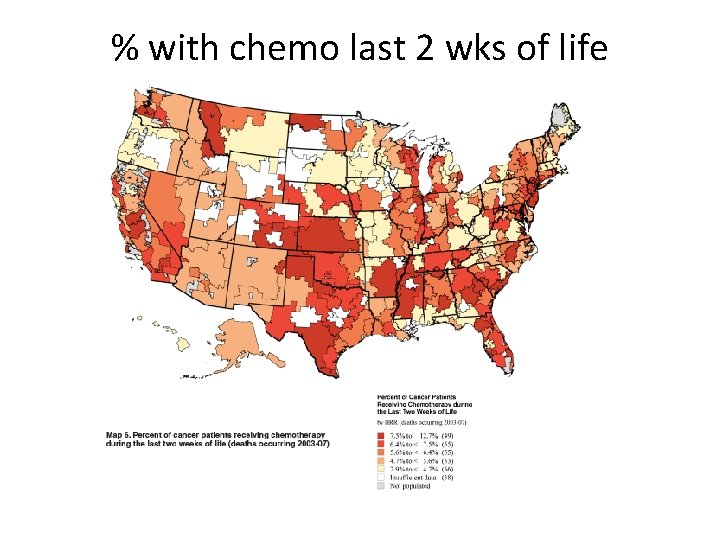
% with chemo last 2 wks of life Goodman DC, Quality of EOL Cancer Care for Medicare Beneficiaries. A Report of the Dartmouth Atlas Project, 11/16/2010. www. dartmouthatlas. org
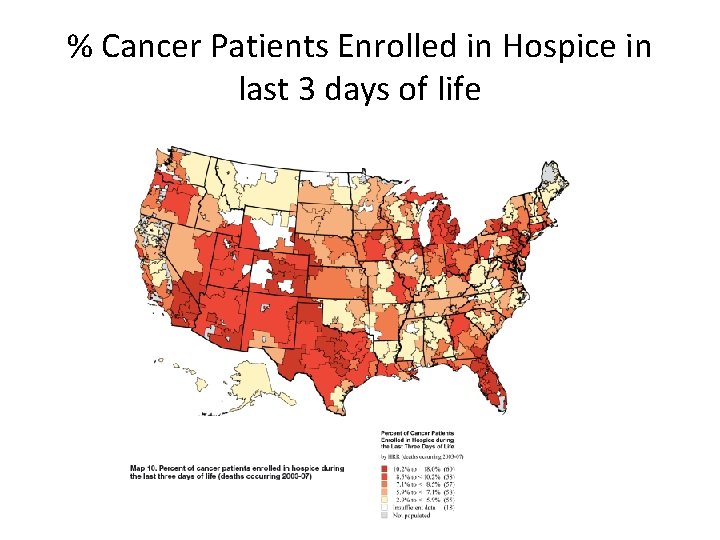
% Cancer Patients Enrolled in Hospice in last 3 days of life

Caveat: Selection Biases • Patients with poorer prognosis more likely to get treatment • Results in implausibly worse survival in more extensively treated patients • Patients with better underlying health more likely to get treatment • Survival better among treated patients • Attempt to correct with statistical methods • matching, multivariate analyses, stratification, restriction, propensity analysis, IVA

Limitations • Overall mortality • Cancer specific mortality • Other cause mortality • Skepticism regarding observational studies that compare effectiveness of different treatments • Studies stronger with other uses of data • Patterns of care, treatment compliance, toxicities of therapy overall and across populations, elderly, minorities, comorbidities

Longitudinal (cohort) study examples

The Study to Understand Prognoses and Preferences for Outcomes and Risks of Treatments ¡SUPPORT Study Provides A Hint w 9105 adults hospitalized with one or more of 9 life threatening conditions; overall 6 -mos mortality 47% w 2 year observational study, then a 2 year controlled trial to evaluate the impact of an intervention • Detailed, validated prognostic models were developed for each patient and shared with the physicians • Specially trained nurses communicated in depth with pt/family • Physicians were provided with detailed written instructions about the patient’s and family’s wishes

SUPPORT STUDY Observational Study Findings ¡MDs did not seem to know what kind of care their patients wanted ¡Severe pain was unacceptably common ¡Too many died in ICU on life support Controlled Trial Findings ¡The experimental group did no better than controls across a broad spectrum of outcomes

Coping with Cancer Study ¡NCI funded, multisite, longitudinal study of advanced cancer patients and their caregivers ¡Recruitment was 2002 -2006 ¡Designed to fill research gaps on racial/ethnic disparities in EOL Care ¡Of participants, 593 self-identified as white, black, or Hispanic
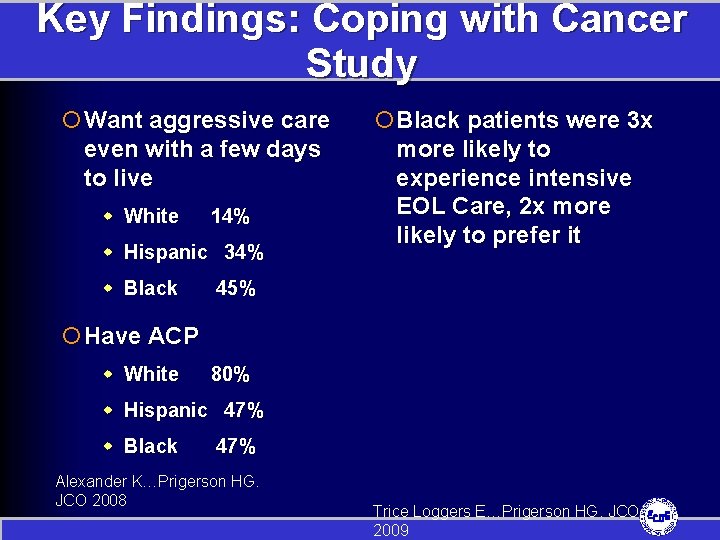
Key Findings: Coping with Cancer Study ¡ Want aggressive care even with a few days to live w White 14% w Hispanic 34% w Black ¡ Black patients were 3 x more likely to experience intensive EOL Care, 2 x more likely to prefer it 45% ¡ Have ACP w White 80% w Hispanic 47% w Black 47% Alexander K…Prigerson HG. JCO 2008 Trice Loggers E…Prigerson HG. JCO 2009

The Disparities: Coping with Cancer Study ¡White patients who prefer intensive EOL care 3 x more likely to receive it than blacks with the same preference ¡Black patients who had EOL discussion and DNR orders still sometimes received intensive EOL care Trice Loggers E…Prigerson HG. JCO 2009

CANCORS Study • Large observational cohort study • 1193 patients with stage IV lung or colorectal cancer who received chemotherapy • Professional interviewers and comprehensive record review

Inaccurate Response to Questions about the Likelihood of Cure with Chemotherapy Odds ratios Hispanic 2. 82 Black 2. 93 Asian 4. 32 Weeks JC et al. N Engl J Med 2012; 367: 1616 -1625

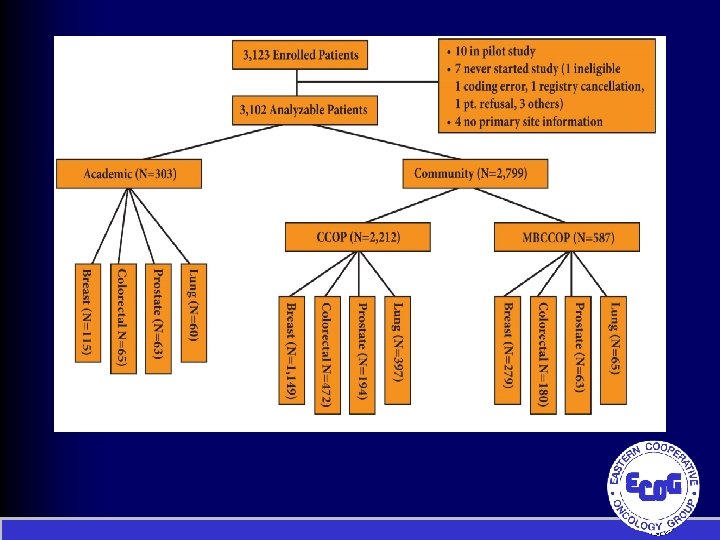
Symptom Outcomes and Practice Patterns (SOAPP) Study Schema R E G I S T E R Initial Assessments • Patient Forms • Clinician Forms • 2310 Breast, colorectal, prostate, and lung patients • 1/3 from academic sites & 2/3 from community sites Follow-up Assessments 28 -35 Days • Patient Forms • Clinician Forms

Cohort study management ¡Centralized data management and stats w No data torturing w Maintain consistency with data coding and analyses ¡Data sharing policies ¡Secondary concepts are hypothesis driven ¡Beware that the work goes beyond the funding
www. ecogsoapp. org
- Slides: 38