Research Involving Human Subjects and Conducted by Medical


Research Involving Human Subjects and Conducted by Medical Students Patricia L. Gordon, M. D. Executive Chair, Institutional Review Board (IRB) Executive Director, Human Subjects Protection Office (HSPO) Kathleen Hay, Ph. D, CIP Director, HSPO

Overview of Presentation l l The “why” behind all we do What needs to be done for a study to be approved? How do you get it done? Then what – what do you need to do once a study is approved?

Tips for Success in Navigating the Human Subjects Protection System l l Start early Read and follow directions Understand the process Ask for help early

Research with Human Subjects l l Clinical research requires human volunteers Human participation necessitates public trust Social contract Law

Public Trust Tested by Historical Abuses l Nuremberg Trials – 1946 l Thalidomide – 1961 l The Milgram Study – 1963 l Tuskegee – 1932 - 1972 l Guatemala – 1944 -1948 l Human Radiation Experiments 1944 - 1974

Principles of Human Subject Protection The Belmont Report: Guiding Ethical Principles § Respect for Persons Informed consent § Protection for the vulnerable § § Beneficence Maximize possible benefits § Minimize possible harms § § Justice § Fair distribution of research burdens vs. benefits

Respect for Persons l l captured in the consent process incorporates two ethical convictions: n n individuals should be treated as autonomous agents persons with diminished autonomy are entitled to increased protection

Informed Consent “A process by which a subject voluntarily confirms his or her willingness to participate in a particular trial, after having been informed of all aspects of the trial that are relevant to the subject’s decision to participate. ” www. isotechnika. com/glossary

• Beneficence l l Do not harm Maximize possible benefits and minimize risks Risk: possibility that harm may occur both in the probability of experiencing a harm and the magnitude of the envisioned harm Benefit: something of positive value related to health or welfare

Addressing Beneficence l l Good research design Privacy of confidential medical information Competent investigators Favorable risk-benefit ratio n The fewest number of subjects are placed at the least amount of risk to answer a scientifically valid question

Justice l l Fairness in distribution of risks and benefits. An injustice occurs when: n n benefits to which a person is entitled are denied without good reason or when burdens are imposed unduly


What is the IRB? l l l Ethics review committee Advocates for the rights, safety and welfare of subjects Includes physicians, scientists and non-scientists, and representatives of the community

Penn State Hershey IRB Institutional Review Board n n n Operates under federal regulations, state law and institutional policy Authority to approve, require modification in, or disapprove research IRB approval is needed for all human research at Penn State Hershey

What is Human Research? Department of Health & Human Services (DHHS) l l Research - a systematic investigation. . . , designed to develop or contribute to generalizable knowledge. Human subject - a living individual about whom an investigator conducting research obtains: n data through intervention or interaction with individual, or n identifiable private information – individually identifiable; – reasonably expect not to be observed, recorded or made public (IRB is also the ‘Privacy Board’ at HMC/COM and privacy protection requirements apply to health information of decedents)

What activities require IRB review? l l Research involving existing or prospectively collected human specimens or data (e. g. review of medical records) Educational research Interaction/interventions with individuals n Research involving surveys, interviews or focus groups n Clinical trials of investigational drugs and devices Activities driven by an intent to publish or share beyond the institution

What activities usually do not require IRB review? * l l l Case report or case series Quality improvement projects Quality assessment projects Medical practice and innovative therapy Public health practice (disease monitoring or program evaluation) l *some exceptions and many journals require IRB review

What The IRB Wants To Know l Do you understand ethical research? n l What are you doing? n l Procedures/Protocol To whom are you doing it? n l Training requirement Subject Population Inclusion/Exclusion Criteria What are the risks and benefits? How will you ensure voluntary participation?

IRB Review of Research IRB Review Criteria § Informed Consent ü ü ü § Will be sought prospectively Includes required elements of information Additional safeguards to protect vulnerable subjects Risk are Balanced and Minimized (Beneficence) ü ü ü § (Respect for Persons) Sound research not to unnecessarily expose subjects to risk Risks are minimized (e. g. - use data or proc. already available) Risk to subjects is reasonable vs. potential benefits Safety provisions & monitoring are adequate Privacy & confidentiality provisions are adequate Subject Selection is Equitable (Justice) ü ü Rationale for inclusion/exclusion criteria Involvement of vulnerable populations justified

What are the IRB review categories? l Types of review n n n l Exempt from IRB review Expedited IRB review Full board (convened) IRB review Affected by the level of risk and the type of project

What is minimal risk? Minimal Risk = both probability and magnitude of possible harm or discomfort are not greater than that ordinarily encountered in daily life or during the performance of routine physical or psychological examinations or tests

Access to Private Medical Information l l If no clinical relationship with patient/subject, then no right of access to medical record This access might be granted by the IRB with a waiver of the informed consent requirement for specific research projects under certain circumstances

MSR Project l Confidentiality of Medical Records n n No access to medical records without some clinical access i. e. the patient’s physician (or partner of patient’s physician) Once you get access you are held to confidentiality. Outside of the research you are not at liberty to reveal anything about the patient/subject to anyone, including the fact that he/she has a medical record here

MSR Projects: Exempt vs Expedited l l Exempt from IRB review: “no risk” No identifiers recorded n n n Surveys - anonymous Review of publically available information (public disease registry, blogs) Review of medical records where no identifiers are recorded (no links and no repeat access to medical record)

MSR Projects: Exempt vs Expedited l Expedited: always “not more than minimal risk” n n n Medical record reviews Surveys or educational tools where identifiers are recorded Interventional studies with blood samples, urine samples or innocuous tests (ultrasound, MRI, EKG, exercise on healthy people)

MSR Projects – Example #1 – l Med student designs a study l Study will be conducted at HMC and/or off-site l Study involves human subjects

MSR Projects – Example #1 IRB Requirements for Example #1 l Study must be submitted to the Hershey IRB for review using the standard IRB forms l Student must complete the CITI Course training l Hershey IRB reviews and approves study and issues an approval or exemption determination memo n n After receiving the IRB approval memo the student may start the research Student must provide a copy of the IRB approval memo to the Director, Medical Student Research

MSR Projects – Example #2 l Medical student plans to work on an IRB-approved study here at Hershey l Study has previously been confirmed exempt or received IRB approval

MSR Projects - Example #2 IRB Requirements for Example #2 l Principal Investigator of the study must submit a Modification Request Form to add the student as a co-investigator l Student must complete the CITI Course l PI receives an IRB approval memo for the addition of the student to the study n n After the PI receives this memo the student may commence work on the project Student must provide a copy of this memo to the Director of Medical Student Research

MSR Project – Example #3 l Medical student plans to work as a co-investigator on a study off campus l Study has previously been confirmed exempt or received approval from another IRB

MSR Projects – Example #3 IRB Requirements for Example #3 l Student must submit the following to the Hershey IRB office n n n l MSR proposal Copy of the approval memo from the other IRB Email or letter from the principal investigator at the other institution stating that they agree to supervise the student’s work on the project Student must complete the CITI Course

MSR Projects – Example #3 cont’d l IRB Chair reviews the information to decide if review by the Hershey IRB is required. n If review and approval by the Hershey IRB is required, the study must be submitted to the Hershey IRB using the standard IRB forms as in Example #1.

MSR Projects REMEMBER: l IRB approval must be obtained prospectively l The IRB does not grant retroactive approval for research that has already taken place.

How do I determine if my project needs IRB review? n n n Decision charts & FAQs on IRB website Call the Human Subjects Protection Office for a consultation Request for a formal determination u Submit an ‘IRB Determination’ form
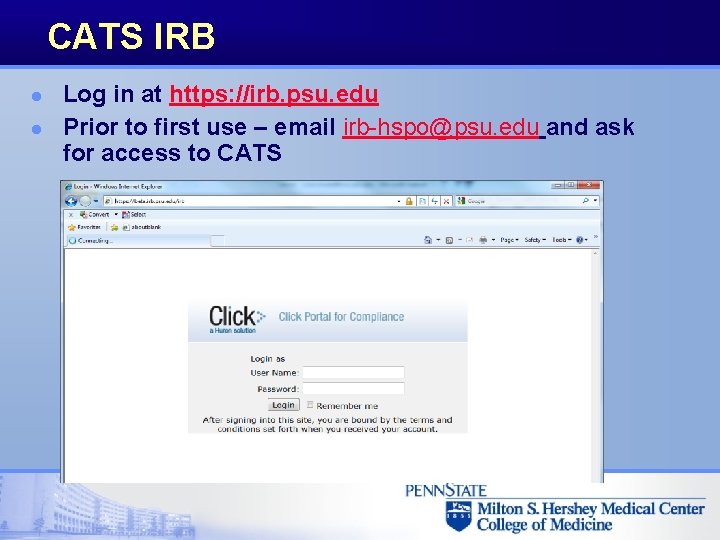
CATS IRB l l Log in at https: //irb. psu. edu Prior to first use – email irb-hspo@psu. edu and ask for access to CATS
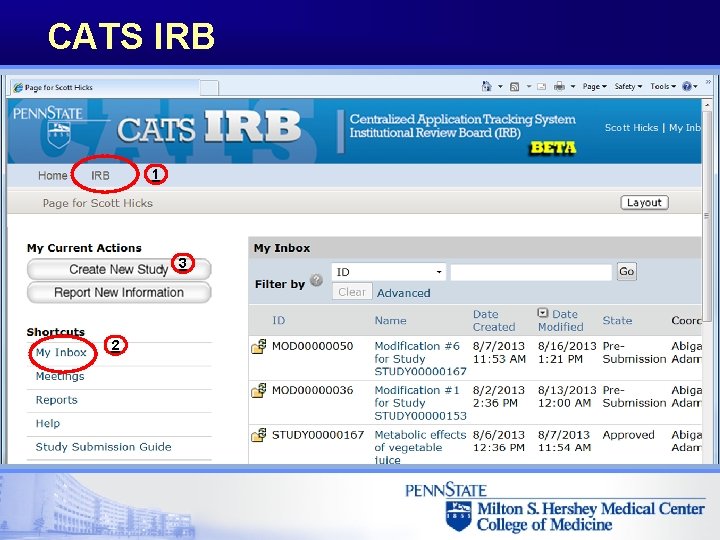
CATS IRB 1 3 2
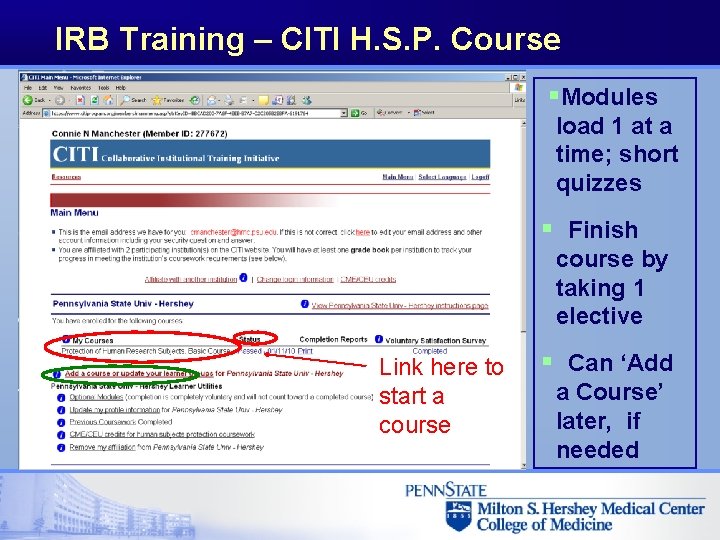
IRB Training – CITI H. S. P. Course §Modules load 1 at a time; short quizzes § Finish course by taking 1 elective Link here to start a course § Can ‘Add a Course’ later, if needed

IRB Submission Process Submit 8 weeks in advance • • Best to consult with HSPO staff first if unclear Instructions & online process at www. pennstatehershey. org/web/irb IRB Submissions include: l l Completing an online submission form Uploading all supporting documents n n Research protocol Informed consent process and documents Recruitment methods/materials, if applicable Supporting materials, if applicable

International Research l l Student must contact Global Health Center and HSPO 6 months prior to departure date Student must submit all documents to HSPO by 3 months prior to departure date

Other committees that review research Other reviews needed, when applicable: l l l Conflict of Interest Review Committee (CIRC) Human Use of Radioisotopes Committee Institutional Biosafety Committee Anatomic Pathology Departmental scientific review committees Links and info. on IRB web site

After Approval is Received l l l Do what you said you were going to do. If you need to change anything, you need IRB approval first. If something doesn’t go according to plan, the IRB needs to know asap. The IRB needs to review the study’s progress. Respond to all communication from the IRB in a timely manner.

Key Responsibilities for Investigators l The principal investigator has the ultimate responsibility for the conduct of the study u The ethical performance of the project u The protection of the rights and welfare of human participants u Strict adherence to the study protocol and any stipulations imposed by the IRB u Compliance with applicable federal, state, and local regulations and PSU policies u Assurance that key personnel are trained/qualified u Acquisition of legally effective informed consent

Key Responsibilities for Investigators l Obtaining legally effective informed consent from participants or their LAR n n n Using only the currently approved, date-stamped informed consent documents Ensuring that only IRB-approved investigators obtain informed consent Informing subject of any new information

Ongoing IRB Review Responsibilities Continuing Review l Continuing review approval must be obtained within the stipulated timeframe n l l Approval effective for a maximum of 1 year Failure to do so results in automatic expiration of IRB approval PI receives e-mail reminder and progress report form to complete

Ongoing IRB Review Requirements Changes to Research l Changes in the research plan must be approved by IRB prior to implementation IRB must review all changes u Obtain IRB approval before implementing changes, unless necessary to avoid immediate hazard u n Types of changes u Protocol amendments - permanent intentional action exceptions - one-time, intentional action

Ongoing IRB Review Requirements Reportable New Information n Investigators must notify the IRB of any new information that falls into one or more of the categories listed on next slide Within 5 business days Process: u Researchers report information to the IRB and u IRB determines if the information represents unanticipated problems involving risks to participants or others or non-compliance

Ongoing IRB Review Requirements Information that needs to be reported promptly to the IRB l l l Information that indicates a new or increased risk, or new safety issue: IB, interim analysis, safety monitoring reports, Harm experienced by subject or others 1) unexpected 2) probably related as determined by the HMC PI Non-compliance w/ federal regs or determinations of IRB Audit, inspection or inquiry by fed. agency & resulting reports Reports by RQA and study monitors l l l l Failure to follow the protocol Breach of confidentiality Changes to protocol without prior IRB review to eliminate immediate hazard to subject Incarceration of a subject in a study not approved to involve prisoners Complaint of a subject that cannot be resolved by the research team Premature suspensions or termination of a study by the sponsor, investigator or institution Unanticipated adverse device effect

Tips for Success in Navigating the Human Subjects Protection System l l Start early Read and follow directions Understand the process Ask for help early

Resources for Investigators l Human Subjects Protection Office Phone (717) 531 -5687, Academic Support Bldg. #1140 u Individual Consultations l IRB/HSPO web site: www. pennstatehershey. org/web/irb n Resources/Investigator Resources u Educational requirements – CITI course access u Making an IRB Submission – instructions and templates l IRB Seminars n n Dates posted on IRB web site Select presentations available at http: //media 1. hmc. psu. edu/mediasite/Catalog/
- Slides: 50