Research Integrity HMS Office for Academic and Research












- Slides: 14

Research Integrity HMS Office for Academic and Research Integrity (ARI)

Factors that Undermine Integrity from Lang, James M. Cheating Lessons: Learning from Academic Dishonesty. 2013. HUP • Low-frequency, high-stakes reviews of performance (vs. high-frequency, low-stakes demonstrations of mastery) • Perception of low probability to succeed • Perception of poor alignment between metrics and value • Perception of bias in evaluations of performance • Lack of positive peer/institutional pressure (to prefer mastery over performance, to refuse to game the system, etc. ) • Perception of low institutional commitment to ethics/training • Hypercompetition Sound familiar? OFFICE FOR Academic and Research Integrity 2

Early Cases Eric T. Poehlman • • Longitudinal Menopause Study (1995) 10 papers, 17 grant applications (1992 -2002) Retractions, $180 K fine, debarment (2005)Eli Kintisch, “Poehlman Sentenced to 1 Year of Prison” (2006) Science Sentenced to prison (2006) Andrew Wakefield • • • Nick Allen, “MMR-autism link doctor Andrew Wakefield defends conduct at GMC hearing” (2008) The Telegraph • Linked MMR vaccine to autism in The Lancet (1998) Conflict of interest and fraud (2004 - 2011) UK General Medical Council (2010) revoked his license and forced full retraction Public health (e. g. , ongoing measles & mumps outbreaks) OFFICE FOR Academic and Research Integrity 3

Recent Events Anil Potti (2010) • Genomics-based personalization of cancer treatment Dong-Pyou Han (2013) • • Faked HIV vaccine data; ORI debarred him for 3 years Grassley-led outcry brought the case to criminal court: 57 -month prison sentence and $7 million fine (2015) Olivier Voinnet (2015) • • • Two institutions: ETH Zurich vs. CNRS Same findings of fact, different conclusions Data as means to an end (integrity of conclusions) vs. data as the end in themselves (integrity of data) OFFICE FOR Academic and Research Integrity 4

The Scale of the Problem Fanelli (2009) in Plo. S ONE • • • Nearly 2% of scientists admitted falsifying data once 1 in 3 reported engaging in other “QRPs” 1 in 7 saw colleagues falsify data; almost ¾ saw QRPs Tavare (2007) in the BMJ • 1 in 7 researchers in U. K. witnessed others falsifying data to increase publishing potential OFFICE FOR Academic and Research Integrity 5

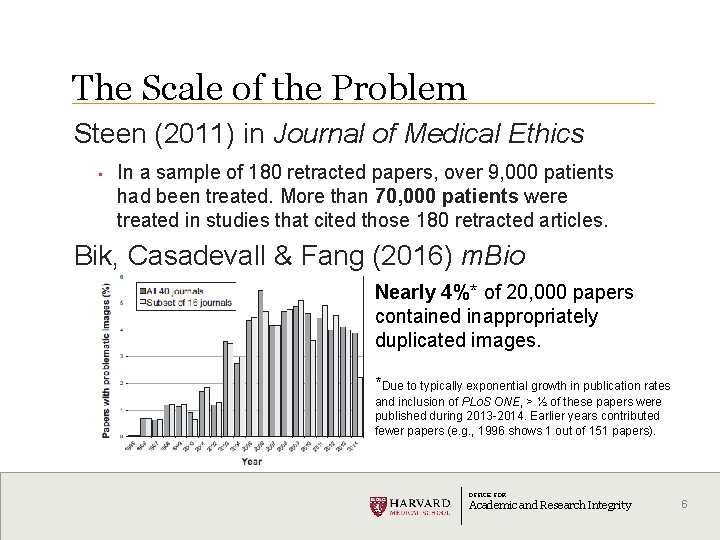
The Scale of the Problem Steen (2011) in Journal of Medical Ethics • In a sample of 180 retracted papers, over 9, 000 patients had been treated. More than 70, 000 patients were treated in studies that cited those 180 retracted articles. Bik, Casadevall & Fang (2016) m. Bio Nearly 4%* of 20, 000 papers contained inappropriately duplicated images. *Due to typically exponential growth in publication rates and inclusion of PLo. S ONE, > ½ of these papers were published during 2013 -2014. Earlier years contributed fewer papers (e. g. , 1996 shows 1 out of 151 papers). OFFICE FOR Academic and Research Integrity 6

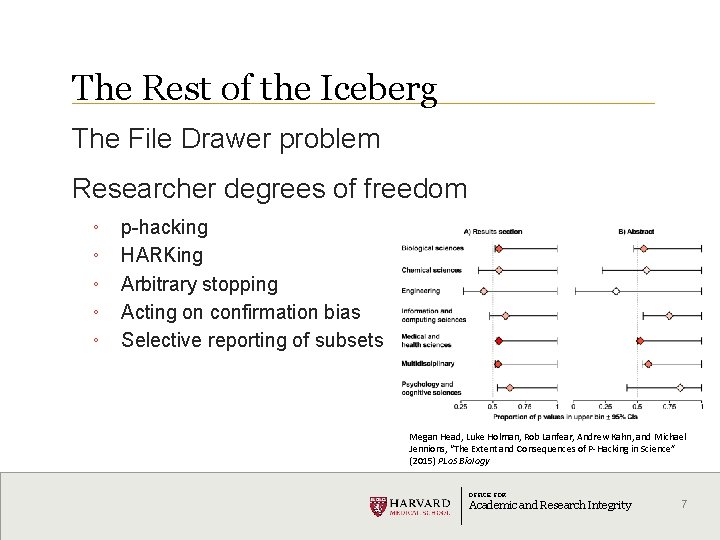
The Rest of the Iceberg The File Drawer problem Researcher degrees of freedom º º º p-hacking HARKing Arbitrary stopping Acting on confirmation bias Selective reporting of subsets Megan Head, Luke Holman, Rob Lanfear, Andrew Kahn, and Michael Jennions, “The Extent and Consequences of P-Hacking in Science” (2015) PLo. S Biology OFFICE FOR Academic and Research Integrity 7

Under USA Public Health Service 1) Fabrication = making up data and recording them 2) Falsification = manipulating materials/processes or changing/omitting data, such that the research is not accurately represented in the research record 3) Plagiarism = appropriating ideas, processes, results, or words without giving appropriate credit Research misconduct = any one of these three… while proposing, performing, reviewing, or reporting research. It is NOT honest error or differences of scientific opinion. Code of Federal Regulations title 42 (PHS), part 93 OFFICE FOR Academic and Research Integrity 8

The Federal Standard Federal regulations dictate procedures that must be followed during investigations of possible misconduct, and enforce the confidentiality of those proceedings (with rare exceptions). Under 42 CFR 93, a formal finding of research misconduct requires that: • There is a significant departure from accepted practices of the relevant research community; and • The misconduct is committed intentionally, or knowingly, or recklessly; and • The allegation is proven by a preponderance of evidence. (There is also a statute of limitations. Sort of. ) OFFICE FOR Academic and Research Integrity 9

Research Misconduct Process • Sources of Allegations • • • In-person complaint Anonymous evaluation of public data (e. g. , Clare Francis) Funders (e. g. , OASH ORI or NSF OIG) Journals Preliminary Assessment Confidentiality (and reputation) Sequestration of data & Notification Inquiry Investigation OFFICE FOR Academic and Research Integrity 10

Process Outcomes • Panel reports to Standing Committee • Committee to HMS Dean and affiliate President/CEO (Deciding Officials) • Reporting (suggested or required) to • • • Current employer Letters of Reference Board of Registration in Medicine NIH, NSF, Do. D, FDA, and/or other authorities & funders Journals OFFICE FOR Academic and Research Integrity 11

Risk Factors • Record keeping Data management strategies: Established systems support human memory and organize electronic files • • Training and mentorship on standards • • Explicit guidance on lab practice (from authorship criteria, to RCR, to protocols and analytical pipelines) International collaboration Communication barriers and untested assumptions • Reluctance to appear to be “the weak link” • Dependence on research success for visa status • • Faculty & trainee stress levels OFFICE FOR Academic and Research Integrity 12

And Most Importantly… • Careful Review of Original Primary Data • • • Write once, read many times (WORM): read-only raw data repositories with metadata, q. v. OMERO Review raw data regularly Discuss and challenge analytical pipeline, interpretations, and confounds • Redundant/independent replication where feasible • The value of data as an end in itself OFFICE FOR Academic and Research Integrity 13

Questions? • Dan Wainstock, Ph. D. º º º HMS Associate Director for Research Integrity Daniel_Wainstock@hms. harvard. edu +1 -617 -432 -8219 OFFICE FOR Academic and Research Integrity 14