Research Development Type 1 Type 2 Diabetes Amrit

Research & Development Type 1 & Type 2 Diabetes Amrit Bhangoo, MD Division of Pediatric Endocrinology Children’s Hospital Orange County May 14 th, 2016

Diabetes Mellitus in USA • Diabetes affects 25. 8 million people • (#1. 25 million type 1 - 1: 4000) • 8. 3 % of US population has diabetes • If current trends continue, 1 of 3 U. S. adults will have diabetes by 2050 • Total estimated diabetes type 1 related cost is $14 billion, 174 billion for all diabetes http: //www. cdc. gov/diabetes/pubs/pdf/ndfs_2011 JDRF website

Monitoring glycemic control: early history From appearance, color, sediment and often taste In 1954 Glucotest/Testape roll 1960’s the “dipstix”

Insulin delivery Novo. Pen Echo® Insulcheck

Quest for Cure of Diabetes • Biological cure • • Islet cell transplantation Stem cell development Immunomodulation BCG • Technological Cure • Hybrid approach
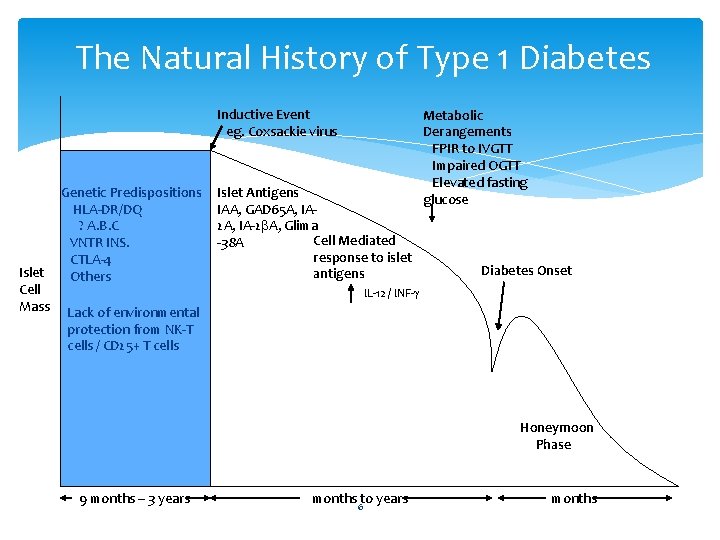
The Natural History of Type 1 Diabetes Inductive Event eg. Coxsackie virus Genetic Predispositions HLA-DR/DQ ? A. B. C VNTR INS. CTLA-4 Islet Others Cell Mass Lack of environmental Islet Antigens IAA, GAD 65 A, IA 2 A, IA-2 b. A, Glima Cell Mediated -38 A response to islet antigens Metabolic Derangements FPIR to IVGTT Impaired OGTT Elevated fasting glucose Diabetes Onset IL-12 / INF-g protection from NK-T cells / CD 25+ T cells Honeymoon Phase 9 months – 3 years months to years 6 months

Immunotherapy Anti. CD 3 monoclonal antibody (m. Ab) Otelixizumab Low doses did not change c-peptide, Hb. A 1 c, insulin dose and glucose Only beneficial in high doses Adverse effects Teplizumab Patients were still insulin dependent Expert Opin Biol Ther. 2016 Jun; 16(6): 841 -6. doi: 10. 1080/14712598. 2016. 1180363. Diabetes Care. 2014 Oct; 37(10): 2746 -54. doi: 10. 2337/dc 13 -0327. Epub 2014 Jul 10. Lancet. 2011 Aug 6; 378(9790): 487 -97. doi: 10. 1016/S 0140 -6736(11)60931 -8. Epub 2011 Jun 28.

Oral Insulin - Prevention Genetically susceptible to type 1 diabetes CD 4+ T-cell proliferative responses Autoantibodies to GAD-65 or IA 2 antigen Diabetes Prevention Trial of Type 1 (DPT-1) -Phase 3 Trialnet Oral Insulin study Pre-Pilot study
Newly diagnosed – Stem cell therapies Multipotent mesenchymal stromal cells (MSCs) Adult stem cells residing in bone marrow, adipose tissue, umbilical cord blood Migrate to injured areas Tissue generation T 1 D-MSCs were able to reverse hyperglycemia in murine diabetes models Lowered ICA, GAD-65 and insulin antibodies of 2 patients in 12 months Stem Cell Res Ther. 2016 Jan 18; 7: 14. doi: 10. 1186/s 13287 -015 -0261 -, Med Sci Monit 19: 852 -857, 2013. .

Stem cell / MSCs Drawbacks Mild therapy “homing” factors toward pancreas Pulmonary first pass effect Contamination Tumorigenicity and immunogenicity Cost-effectiveness

Generation of stem cell-derived βcells from patients with type 1 diabetes Stem cell-derived β-cells from type 1 diabetic patients can be used for treatment of diabetes β cells in vitro Secrete quantities of insulin comparable to adult β cells response to multiple sequential glucose challenges in vitro Nat Commun. 2016 May 10; 7: 11463. doi: 10. 1038/ncomms 11463 Cell. 2014 Oct 9; 159(2): 428 -39. doi: 10. 1016/j. cell. 2014. 09. 040

Islet Cell Technology • Islet cell encapsulation • Natural means, e. g. , seaweed • Artificial membranes • Trying to eliminate the need for chronic immunosuppressant therapy • Implantable cell therapy - UCSD conducting First ever clinical trial of a stem cell-derived therapy

Clinical Islet Transplantation (CIT) Consortium University of Miami, Florida University of Minnesota Minneapolis, Minnesota University of Pennsylvania Philadelphia, Pennsylvania Emory University Atlanta, Georgia Northwestern University Chicago, Illinois University of Alberta Edmonton, Alberta, Canada University of California San Francisco, California University of Illinois at Chicago, Illinois University of Wisconsin Madison Wisconsin Uppsala University Uppsala, Sweden University Hospital Rikshospitalet Oslo, Norway Karolinska University Stockholm, Sweden Massachusetts General Hospital Boston, Massachusetts


Collaborative Islet Transplant Registry Data 2010 571 pancreatic islet allo-transplants 90 with kidney transplant One or two infusions of islets Average number of islets received per infusion was 463, 000. 1 st year - 60 % insulin independence (no insulin for at least 14 consecutive days) 2 nd year - 50 % insulin independence Better outcome age— 35 years or older lower pre-transplant triglyceride, or blood fat, levels lower pre-transplant insulin use Collaborative Islet Transplant Registry seventh annual report. Collaborative Islet Transplant Registry website. Updated December 30, 2011. Accessed July 23, 2013.

Via. Cyte Inc - Pancreas in a Capsule Encapsulation Research Phase 1/2 trial called STEP ONE, or Safety, Tolerability, and Efficacy of VC-01 Combination Product in Type One Diabetes. UCSDSchool of Medicine, in partnership with Via. Cyte, Inc

Technological “Cure” Self Monitoring & Self Management of Diabetes
Blood Glucose Meters i. BG Star meter/app (Sanofi Aventis, available) One. Touch® Verio®Sync Accu-Chek ® Aviva Expert Bolus Advisor System LG KP 8400 cell phone (Korea, 2002); Infopia LG Glucophone/JVAGO 5965 (US, 2008) Freestyle Insu. Linx (Abbott)

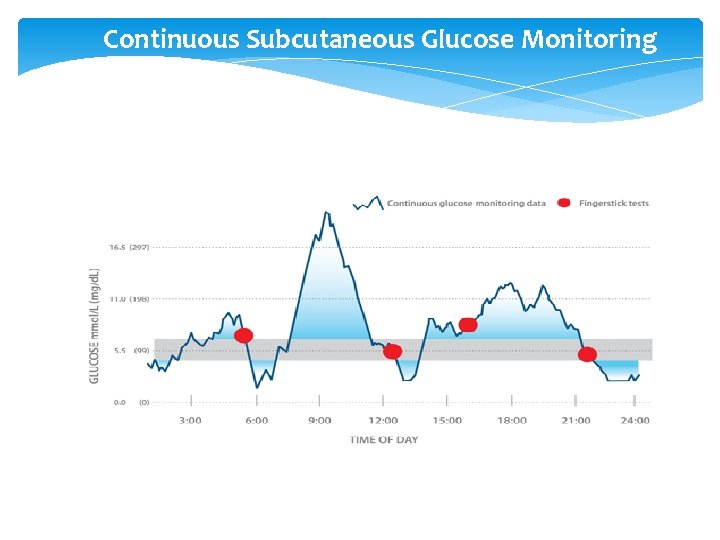
Continuous Subcutaneous Glucose Monitoring
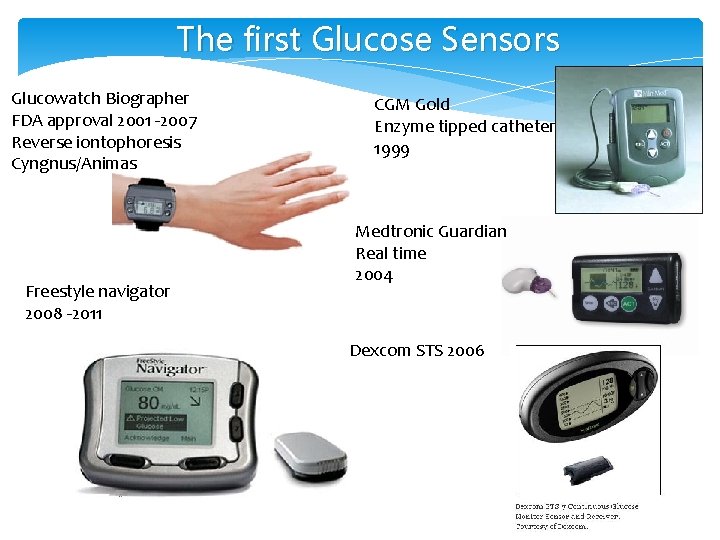
The first Glucose Sensors Glucowatch Biographer FDA approval 2001 -2007 Reverse iontophoresis Cyngnus/Animas Freestyle navigator 2008 -2011 CGM Gold Enzyme tipped catheter 1999 Medtronic Guardian Real time 2004 Dexcom STS 2006

How Does CGM Work? Glucose in the interstitial fluid hits the sensor causing an glucose-oxidation reaction to occur
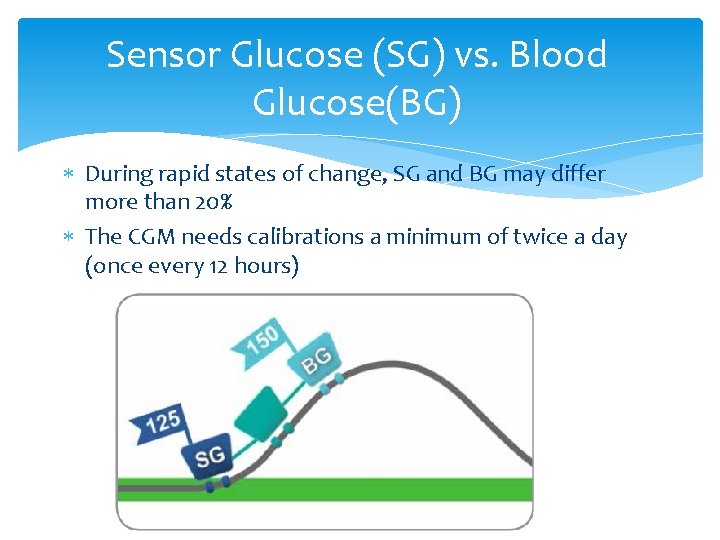
Sensor Glucose (SG) vs. Blood Glucose(BG) During rapid states of change, SG and BG may differ more than 20% The CGM needs calibrations a minimum of twice a day (once every 12 hours)
Todays Glucose Sensors – U. S.

CGM Parts Enlite Sensors Transmitters Receivers Inserters Dexocm
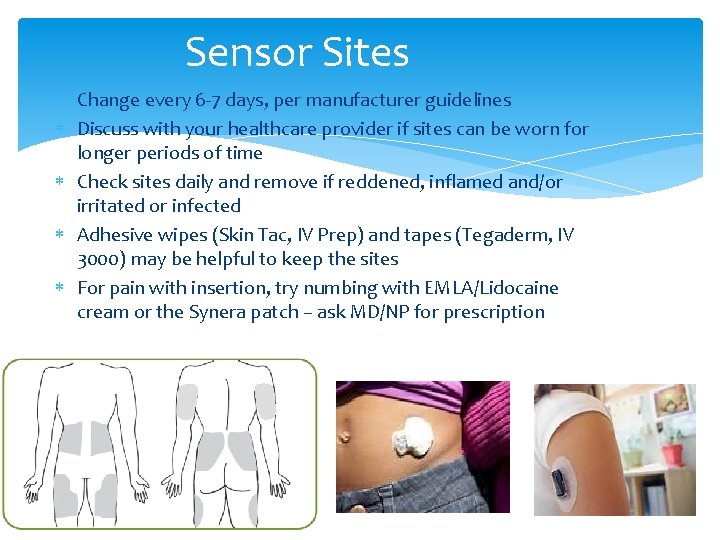
Sensor Sites Change every 6 -7 days, per manufacturer guidelines Discuss with your healthcare provider if sites can be worn for longer periods of time Check sites daily and remove if reddened, inflamed and/or irritated or infected Adhesive wipes (Skin Tac, IV Prep) and tapes (Tegaderm, IV 3000) may be helpful to keep the sites For pain with insertion, try numbing with EMLA/Lidocaine cream or the Synera patch – ask MD/NP for prescription

Dexcom Share and Dexcom G 5 Dexcom receiver that has Share blue-tooth capability builtin so you will not have to have a cable and phone attached to receiver Must have i. Phone, i. Pod touch, or i. Pad with internet connection close to Dexcom receiver Dexcom Share app Real-time data available to 5 users on their i. Phone, i. Pad or i. Pod touch

Sensor Glucose vs. Meter Glucose mean absolute relative difference (MARD) Meter MARD Free. Style Lite 4. 9% Free. Style Freedom Lite 5. 5% Accu-Chek Aviva 6. 8% Contour 9. 0% One. Touch Ultra. Easy 9. 7% Sensor MARD Dexcom <10% Enlite 2 13 -17% J Diabetes Sci Technol. A comparative effectiveness analysis of three continuous glucose monitors: the Navigator, G 4 Platinum, and Enlite. 2014 Jul; 8(4): 699 -708 Accuracy Evaluation of Five Blood Glucose Monitoring Systems Obtained from the Pharmacy: A European Multicenter Study with 453 Subjects. 2012 Apr; 14(4): 330– 337

Mini. Med Connect is available for i. Phone® or i. Pod touch®. If you have a Mini. Med® 530 G or a Mini. Med Paradigm® Revel. TM insulin pump Friends and family can view online with Care. Link®

Glucose Sensors (contd. ) Navigator Libre – Approved in Europe Glucose Tattoo Medtronic & Ford Motor Company glucose monitoring on the go
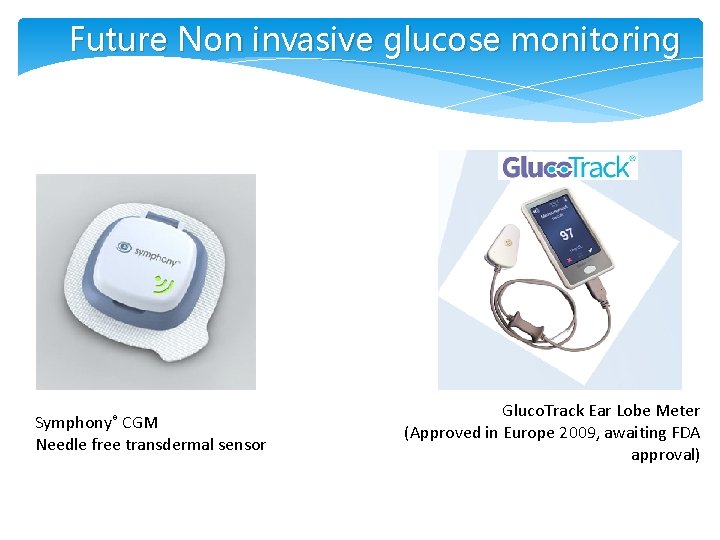
Future Non invasive glucose monitoring Symphony® CGM Needle free transdermal sensor Gluco. Track Ear Lobe Meter (Approved in Europe 2009, awaiting FDA approval)

Non invasive glucose monitoring Or. Sense: Occlusion near infrared spectroscopy
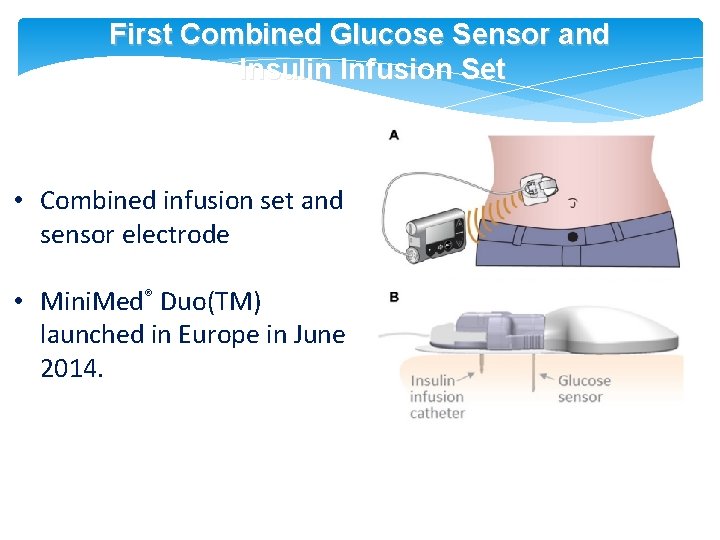
First Combined Glucose Sensor and Insulin Infusion Set • Combined infusion set and sensor electrode • Mini. Med® Duo(TM) launched in Europe in June 2014.
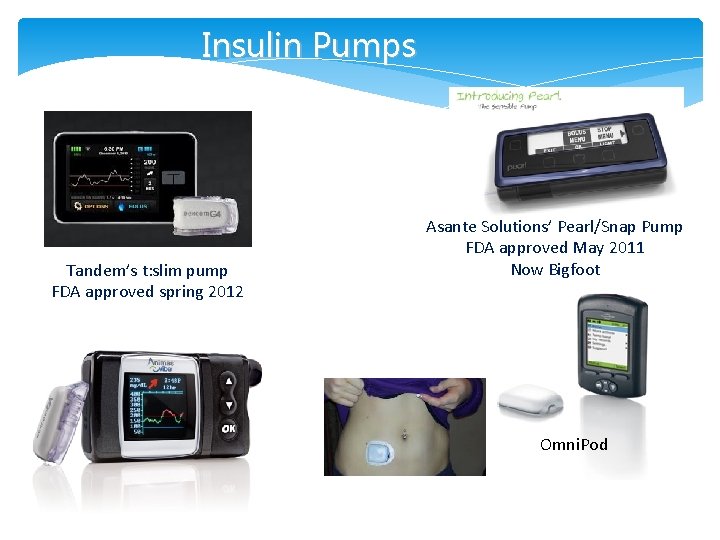
Insulin Pumps Tandem’s t: slim pump FDA approved spring 2012 Asante Solutions’ Pearl/Snap Pump FDA approved May 2011 Now Bigfoot Omni. Pod
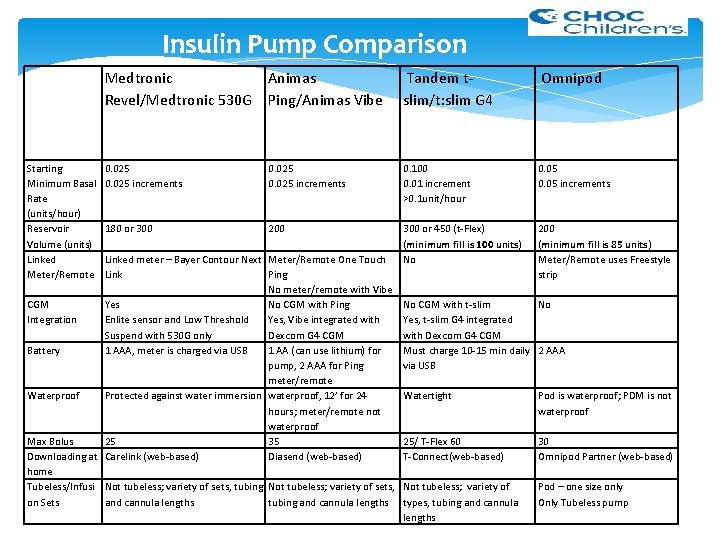
Insulin Pump Comparison Medtronic Animas Revel/Medtronic 530 G Ping/Animas Vibe Tandem tslim/t: slim G 4 Omnipod 0. 025 increments 0. 100 0. 01 increment >0. 1 unit/hour 0. 05 increments 180 or 300 200 300 or 450 (t-Flex) (minimum fill is 100 units) No 200 (minimum fill is 85 units) Meter/Remote uses Freestyle strip Starting Minimum Basal Rate (units/hour) Reservoir Volume (units) Linked Meter/Remote Linked meter – Bayer Contour Next Link Yes Enlite sensor and Low Threshold Suspend with 530 G only 1 AAA, meter is charged via USB Meter/Remote One Touch Ping No meter/remote with Vibe CGM No CGM with Ping Integration Yes, Vibe integrated with Dexcom G 4 CGM Battery 1 AA (can use lithium) for pump, 2 AAA for Ping meter/remote Waterproof Protected against water immersion waterproof, 12’ for 24 hours; meter/remote not waterproof Max Bolus 25 35 Downloading at Carelink (web-based) Diasend (web-based) home Tubeless/Infusi Not tubeless; variety of sets, tubing Not tubeless; variety of sets, on Sets and cannula lengths tubing and cannula lengths No CGM with t-slim No Yes, t-slim G 4 integrated with Dexcom G 4 CGM Must charge 10 -15 min daily 2 AAA via USB Watertight Pod is waterproof; PDM is not waterproof 25/ T-Flex 60 T-Connect(web-based) 30 Omnipod Partner (web-based) Not tubeless; variety of types, tubing and cannula lengths Pod – one size only Only Tubeless pump
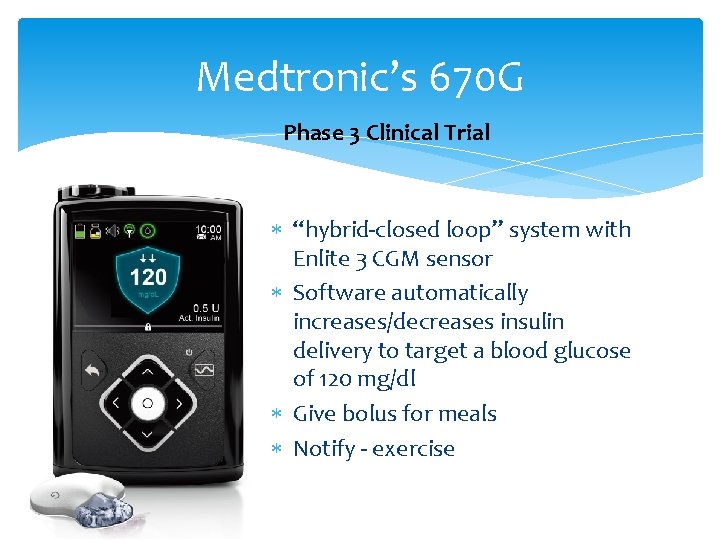
Medtronic’s 670 G Phase 3 Clinical Trial “hybrid-closed loop” system with Enlite 3 CGM sensor Software automatically increases/decreases insulin delivery to target a blood glucose of 120 mg/dl Give bolus for meals Notify - exercise

Future Patch Pumps Jewel. PUMPTM Debiotech Jewel Micro-chip 7 day wear 500 IU of insulin Clinical trials in France (FDA application filed 2014? )

Patch Pumps Cellnovo France & UK Medingo Solo Roche (FDA approved in July 2009) Valeritas V-Go (FDA approved in Dec. 2010) Preset basal rate to deliver 20, 30, or 40 Bolus dosing in 2 Unit increments up to 36 Units Calibra Finesse (FDA approved in Jan. 2010) Launch in 2016 200 units of insulin Bolus only pump 1 -2 units of Insulin / 0. 5 -5 unit boluses )
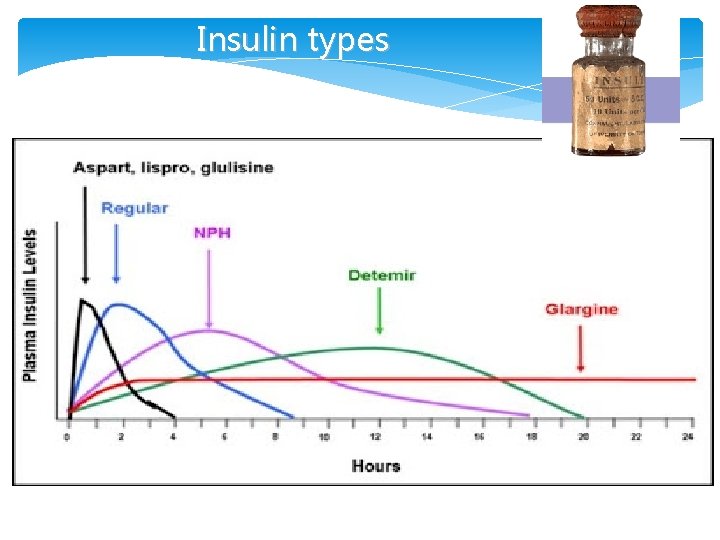
Insulin types

New Insulins/ Alternate Routes of Insulin Delivery • Viaject (in Phase III development) – works faster than all existing insulins, mimics first-phase insulin response • Afrezza Technosphere inhaled insulin (Mann. Kind) – works like first-phase insulin response (FDA approved – Adults) • Oral-lyn buccal spray insulin • Smart. Insulin (Smart. Cells, Inc. ) • Insulin Patch (Pass. Port)
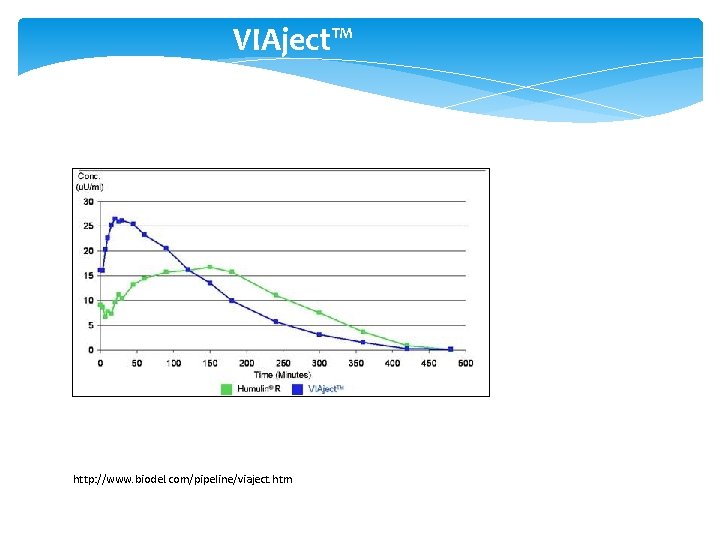
VIAject™ http: //www. biodel. com/pipeline/viaject. htm
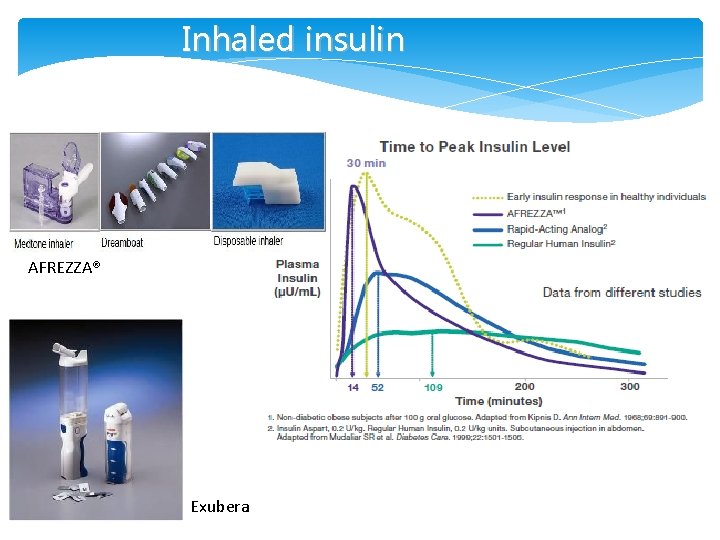
Inhaled insulin AFREZZA® Exubera
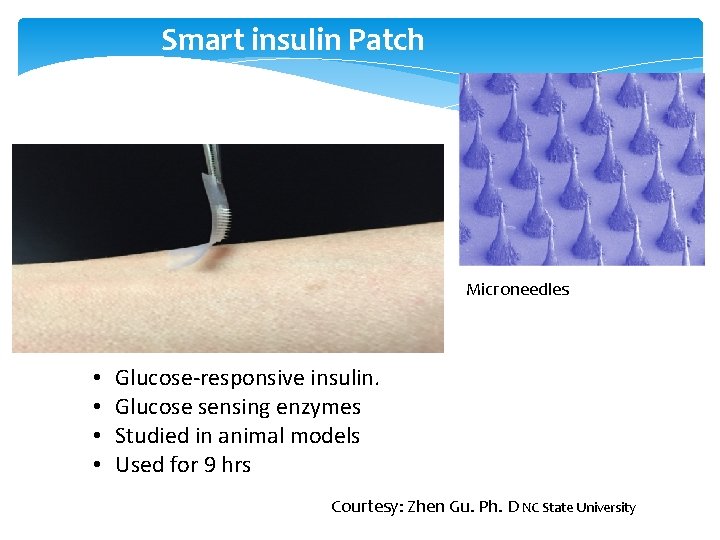
Smart insulin Patch Microneedles • • Glucose-responsive insulin. Glucose sensing enzymes Studied in animal models Used for 9 hrs Courtesy: Zhen Gu. Ph. D NC State University

2011 U. S. Senators, Clinicians and People with Type 1 Diabetes Urge FDA to Issue Clear and Reasonable Guidance on Artificial Pancreas In Just 23 Days, Over 100, 000 Individuals Sign Petition for the FDA to Advance, Not Delay, the Development of an Artificial Pancreas

Closed-Loop Systems – Hypoglycemia Prevention - Most of the severe hypoglycemic events occur at night 1 - 75% of hypoglycemic seizures in children 2 - 6% of the deaths (T 1 DM aged <40 yr) were due to “dead-inbed” syndrome 3 - Hypoglycemia occurred 8. 5% of nights on open CGM 4 - 71% of youth didn’t respond to glucose sensor alarms during the night 5 • Insulin + glucagon • Bolus and then suspend basal • Medtronic Mini. Med 530 G pump – auto-shutoff for lows (2 hour suspend) FDA approved 1 The DCCT , NEJM; 329(14): 977 -86, 1993 2 Davis et al, Diabetes Care; 20(1): 22 -5, 1997 3 Sovik and Thordarson, Diabetes Care; 22(l 2): B 40 -2, 1999 4 JDRF CGM Study group , Diabetes Care; 33: 1004 -8, 2010 5 Buckingham et al, Diabetes Technol Ther; 7: 440 -7, 2005
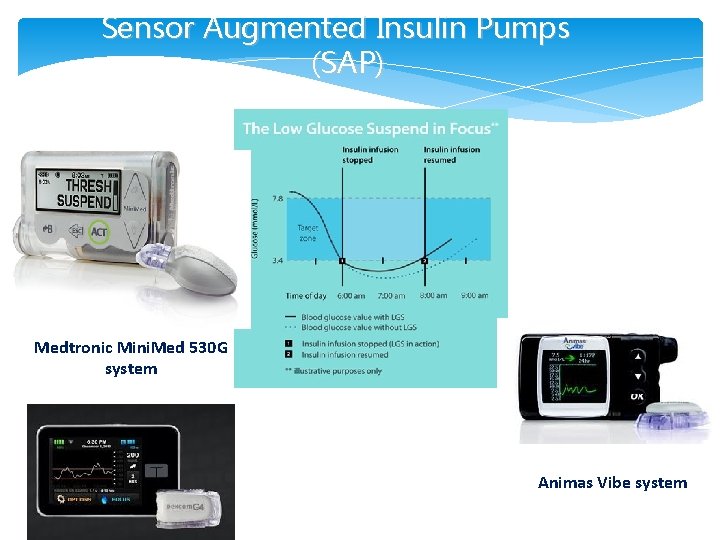
Sensor Augmented Insulin Pumps (SAP) Medtronic Mini. Med 530 G system Animas Vibe system )
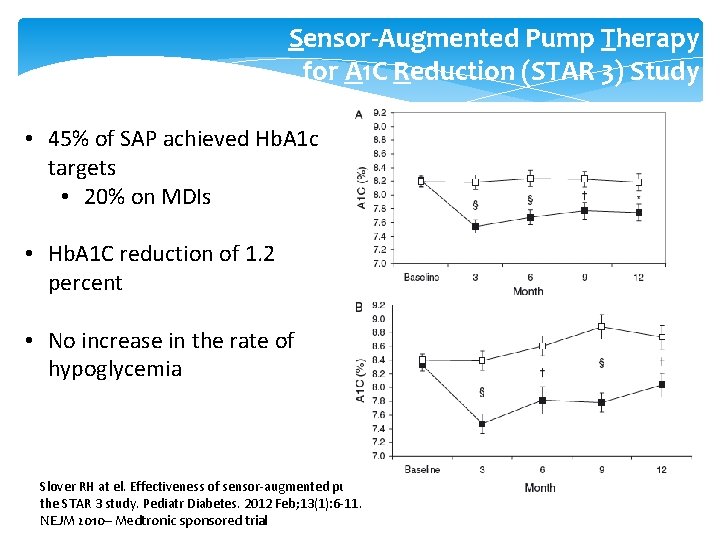
Sensor-Augmented Pump Therapy for A 1 C Reduction (STAR 3) Study • 45% of SAP achieved Hb. A 1 c targets • 20% on MDIs • Hb. A 1 C reduction of 1. 2 percent • No increase in the rate of hypoglycemia Slover RH at el. Effectiveness of sensor-augmented pump therapy in children and adolescents with type 1 diabetes in the STAR 3 study. Pediatr Diabetes. 2012 Feb; 13(1): 6 -11. NEJM 2010– Medtronic sponsored trial

Counter regulatory delivery

Ilet –”Bionic Pancreas”

Bionic Pancreas – the i. Let Dual Chamber pump Glucagon and Insulin Reservoir http: //sites. bu. edu/bionicpancreas/about-us/the-bionic-pancreas-ilet/
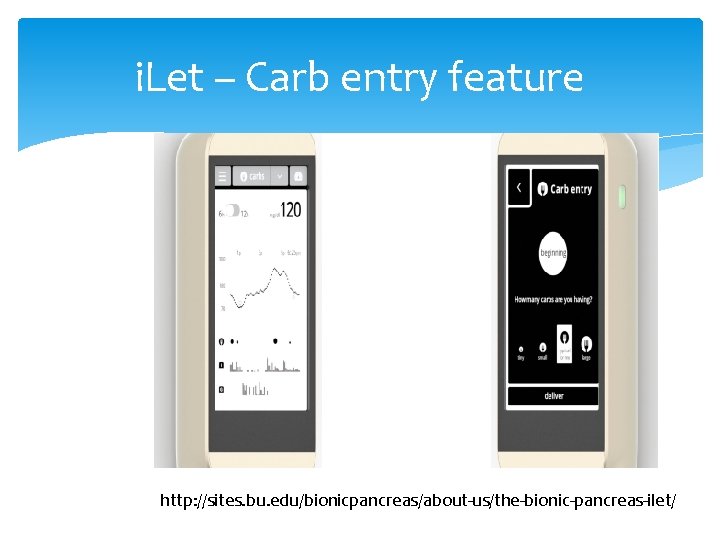
i. Let – Carb entry feature http: //sites. bu. edu/bionicpancreas/about-us/the-bionic-pancreas-ilet/
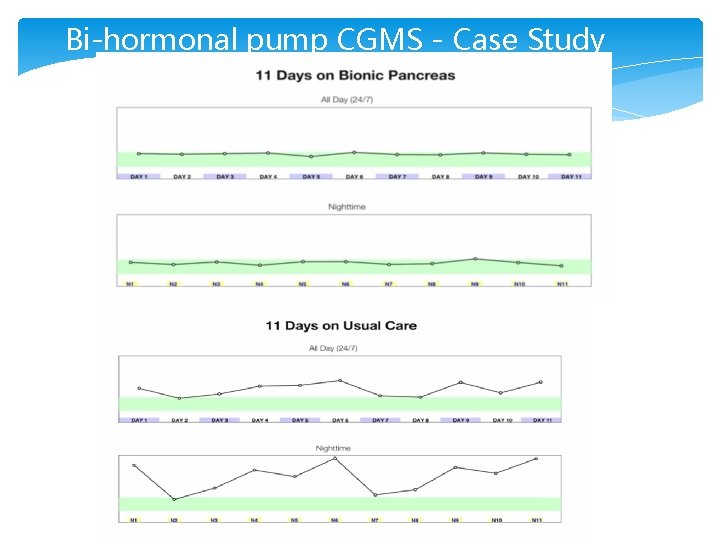
Bi-hormonal pump CGMS - Case Study
The burden of diabetes: so much to consider What affects my diabetes and what I need to know: – Blood glucose --Hb. A 1 c – Food – Activity – Insulin – Illness – Stress level What I need to carry: – Insulin pump – Blood glucose meter – CGM – Computer – Internet access – Mobile phone – Instruction manuals --Too much information! -- Too many devices!

The burden of diabetes Who needs to know about my diabetes? – The hospital doctors – PCP – CDE/ diabetes nurses – The dietitian – The family - School – The pharmacy – The pump supplier Too many people!

Nightscout – CGM in the Cloud Allows real time access to a Dexcom G 4 CGM from web browsers via smartphones, computers, tablets, and the Pebble smartwatch You can see your child’s real-time sensor readings while they are away from you Requires cable, dedicated Android phone i. Pad, i. Phone, Pebblewatch set-up info

OPEN APS The Open Artificial Pancreas System project is an open and transparent effort to make safe and effective basic Artificial Pancreas System (APS) technology widely available to more quickly improve and save as many lives as possible and reduce the burden of Type 1 diabetes.

OPEN APS Insulin pump and CGM Centralized controller - Raspberry Pi minicomputer USB or Bluetooth connection capability Translator device such as a Carelink USB Wireless Internet connection

OPEN APS /Do. It. Yourself Parents/Friends/Patients – Coders/programmer Algorithms based on CGM Insulin on board Carbohydrate decay Calculated on active insulin Real-time predictive alerts for future high or low BG states (hours in advance) Continually updated recommendations for required insulin or carbs http: //diyps. org/
Have a app for that • • Glooko Logbook - Free d. Life Diabetes Companion My. Net. Diary™ Moves Ontrack Glucose buddy Insulincalc Diasend
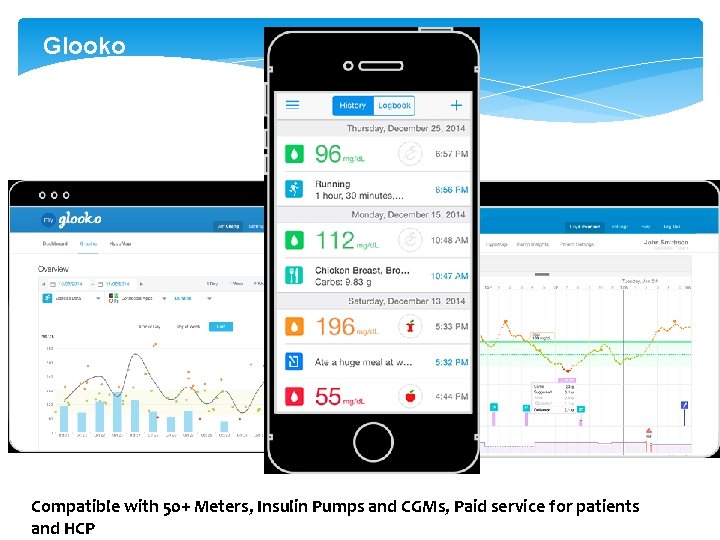
Glooko Compatible with 50+ Meters, Insulin Pumps and CGMs, Paid service for patients and HCP

Downloading and Interpreting Data Medtronic Carelink Personal Web-based PC and MAC compatible Uses Carelink USB or Bayer Contour Link USB Sensor and Pump data combined Can have data linked to MD Office database Dexcom Clarity Software downloaded from website Iphone PC compatible Uses USB cable Sensor data Can email reports Diasend, Gloo. Ko, Tidepool Web-based PC and MAC compatible Use USB cable Sensor Data Can have linked to CHOC database
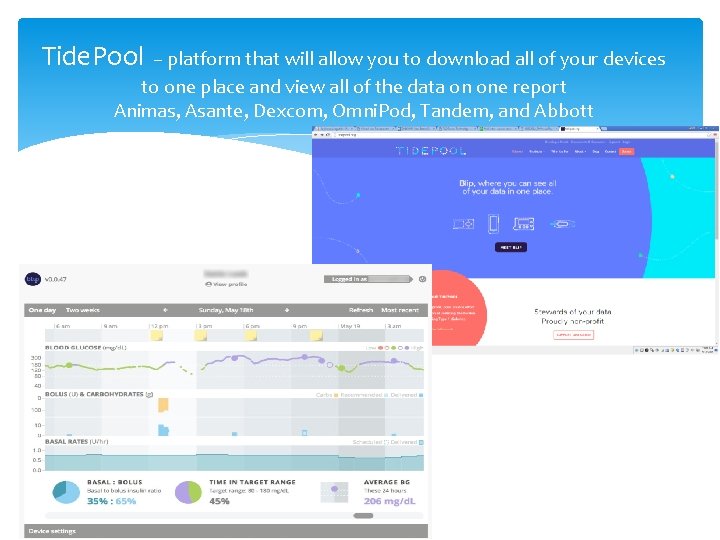
Tide. Pool – platform that will allow you to download all of your devices to one place and view all of the data on one report Animas, Asante, Dexcom, Omni. Pod, Tandem, and Abbott

Developments in Type 2 Diabetes Mellitus

Traditional Oral Medications Metformin – Insulin Sensitizing agent Before starting metformin, obtain the patient’s e. GFR. Contraindicated in patients with an e. GFR below 30 e. GFR between 30 and 45 m. L/minute/1. 73 m 2 is not recommended. Obtain an e. GFR at least annually Sulfonylureas - Stimulate insulin release Cardiovascular mortality was lower in those taking metformin vs sulfonylureas. Metformin-containing drugs: Drug safety communication - revised warnings for certain patients with reduced kidney function [news release]. Silver Spring, MD: FDA; April 8,
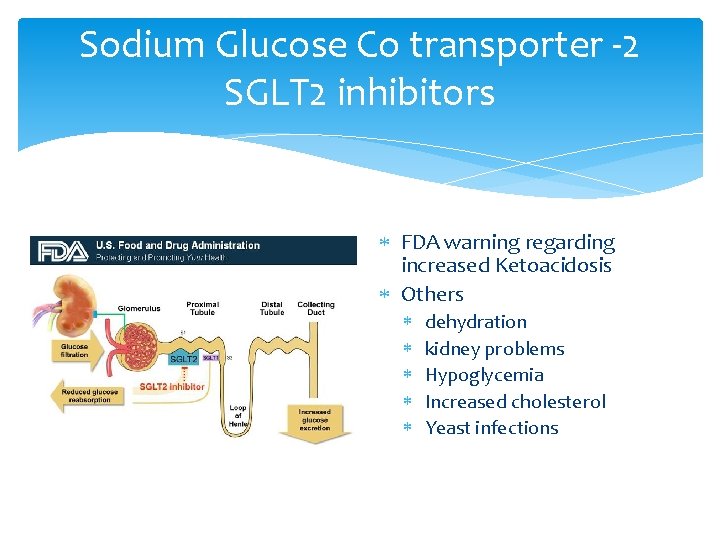
Sodium Glucose Co transporter -2 SGLT 2 inhibitors FDA warning regarding increased Ketoacidosis Others dehydration kidney problems Hypoglycemia Increased cholesterol Yeast infections

SGLT 2 inhibitors lower blood sugar by causing the kidneys to remove sugar from the body through the urine. Generic Brand name Canagliflozin Invokana Canagliflozin and metformin Invokamet Dapagliflozin Farxiga Dapagliflozin and metformin extendedrelease Xigduo XR Empagliflozin Jardiance Empagliflozin and linagliptin Glyxambi

Glucagon like peptides GLP-1 agonists Generic Brand name exenatide Byetta/ Bydureon (ER) liraglutide Victoza, Saxenda (Obesity) albiglutide Tanzeum dulaglutide Trulicity Stimulates insulin secretion Inhibits glucagon secretion Lowering blood sugar Shorter-acting agonists lowering post-meal glucose spikes Long-acting Effect both post-meal and fasting glucose levels

GLP-1 agonists CI: MEN 2 SYNDROME Medullary thyroid carcinoma personal or family history Pancreatitis Gallbladder disease Renal impairment Suicidal thoughts
DPP-4 Inhibitors Inhibits enzyme DPP-4 Increases GLP-1 Stimulates insulin production Lowers glucagon Generic Brand Name Alogliptin Nesina Linagliptin Tradjenta Saxagliptin Ongglyza Sitagliptin Januvia Increased risk of hospitalization for heart failure

TZDs Generic Name Brand Name pioglitazone Actos rosiglitazone Avandia activate a protein PPAR g decreasing insulin resistance. Lower BG levels Needs to monitor LFTs


Summary Immune therapies to slow or alter the autoimmune process are Evolving Pump and Sensor technologies are evolving rapidly Closed loop systems will be leading the future Insulin only algorithms Insulin + Glucagon (bi-hormonal) Diabetes providers would need more exposure to newer technologies Tech savvy parents or patients will be expecting more from providers, drug & device manufacturers and FDA Type 2 Diabetes Mellitus – Much more treatment options In Pediatrics – Insulin & Metformin

Questions?
- Slides: 74