Representing Compounds Molecular Molecular formula shows the types














- Slides: 17

Representing Compounds - Molecular • Molecular formula – shows the types and numbers of atoms combined in a single molecule of a compound • Ex. CO, H 2 O 2, SO 3, O 2

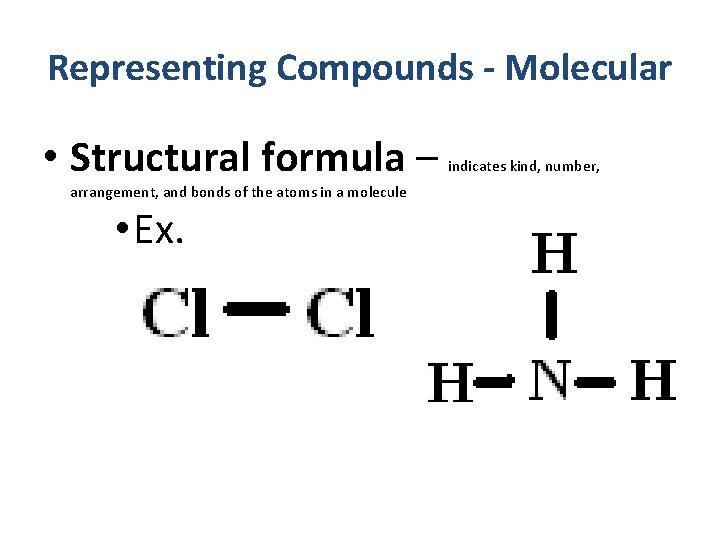
Representing Compounds - Molecular • Structural formula – shows kind, number, arrangement, and bonds (single, double, or triple) of the atoms in a molecule (or polyatomic ion)

Representing Compounds - Molecular • Structural formula – arrangement, and bonds of the atoms in a molecule • Ex. indicates kind, number,

Representing Compounds - Molecular • Lewis structures - diagrams that show valence e- as dots, the inner e- and nucleus are included in the letter symbol for the element being represented

Representing Compounds - Molecular • Lewis structures Reminder… Group 1

Representing Compounds - Molecular • Lewis structures Reminder… Group 2

Representing Compounds - Molecular • Lewis structures Reminder… Group 13

Representing Compounds - Molecular • Lewis structures Reminder… Group 14

Representing Compounds - Molecular • Lewis structures Reminder… Group 15

Representing Compounds - Molecular • Lewis structures Reminder… Group 16

Representing Compounds - Molecular • Lewis structures Reminder… Group 17

Representing Compounds - Molecular • Lewis structures Reminder… Group 18

Representing Compounds - Molecular • Lewis structures – Structures of individual elements may be joined to form compounds – pairs of dots (or a dash) between symbols represent bonds (or electron pairs), dots adjacent only to one symbol are unshared e-

Representing Compounds - Molecular • Lewis structures… – Lone (unshared) pair of e- – pair of ethat is not involved in bonding, but instead belongs exclusively to one atom

Representing Compounds - Molecular • Lewis structures Ex. F 2, NH 3, H 2 O, CH 4, O 2, CO 2, N 2, CH 2 O, C 2 H 2, PI 3 (must draw structures)

Representing Compounds - Molecular • Lewis structures Octet exceptions: Less than full octet • Ex: Boron compounds such as BF 3

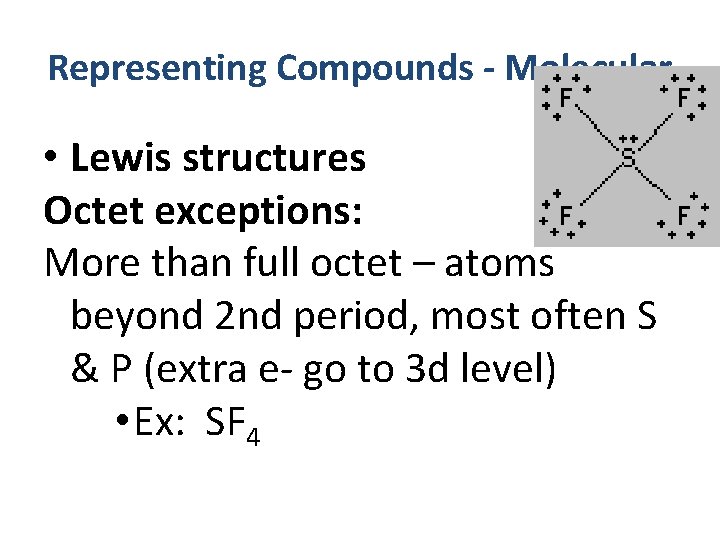
Representing Compounds - Molecular • Lewis structures Octet exceptions: More than full octet – atoms beyond 2 nd period, most often S & P (extra e- go to 3 d level) • Ex: SF 4