Reportable Events and Incidents Training SOP KP502 Research

Reportable Events and Incidents Training SOP KP-502 Research Compliance Training 2012 Kaiser Foundation Research Institute KPSC Version 2 Dated 8/15/2012

Required Training The training takes approximately 1 hour. All Research Team Members who contribute to the conduct of research must complete the training, including: ● Principal Investigators (PIs), Sub-investigators, Research Managers/Coordinators, Nurses, and others as determined by the PI (e. g. , Research Assistants, Medical Assistants, Pharmacists, Data Analysts, etc. ) This training focuses on reporting events and incidents to the Institutional Review Board (IRB). It does not cover the PI’s reporting requirements to external sponsors. Note: This training does not apply to Emergency Use or Humanitarian Use Device research. Kaiser Foundation Research Institute 2

Federal Regulations This training supports SOP KP-502, Principal Investigator Reportable Event and Incident Requirements. SOP KP-502 is written to comply with federal reporting regulations including: ● U. S. Department of Health and Human Services 45 CFR 46 ● U. S. Department of Health and Human Services 45 CFR 164 ● U. S. Food and Drug Administration 21 CFR 50, 56, 312, and 812 Kaiser Foundation Research Institute 3

Course Outline Introduction Events and Incidents Summary Table Responsibility for Identifying and Reporting Events and Incidents Reportable Events and Incidents · Unanticipated Problems · · § Adverse Events/Serious Adverse Events § Other Possible Unanticipated Problems Protocol Violations and Deviations Other Reportable Events and Incidents § Breach of Protected Health Information (PHI) § Incarceration of a Study Participant § New Safety Information § Emergent Modifications § Regulatory Visits/Inspections Resources Kaiser Foundation Research Institute 4

Purpose By the end of the training you should be able to: 1. Analyze an event or incident to determine if it is reportable according to SOP KP-502 2. Determine if an event meets the three criteria defining an “unanticipated problem” 3. Understand your responsibility as a PI or Research Team Member to identify, analyze, and report events and incidents 4. Identify resources to help you comply with SOP KP-502 Kaiser Foundation Research Institute 5

Print Summary Table The table on the next slide (slide 7) summarizes reportable events and incidents, reporting time frames, and reporting methods. Print the table to use as a reference during the training. Kaiser Foundation Research Institute 6
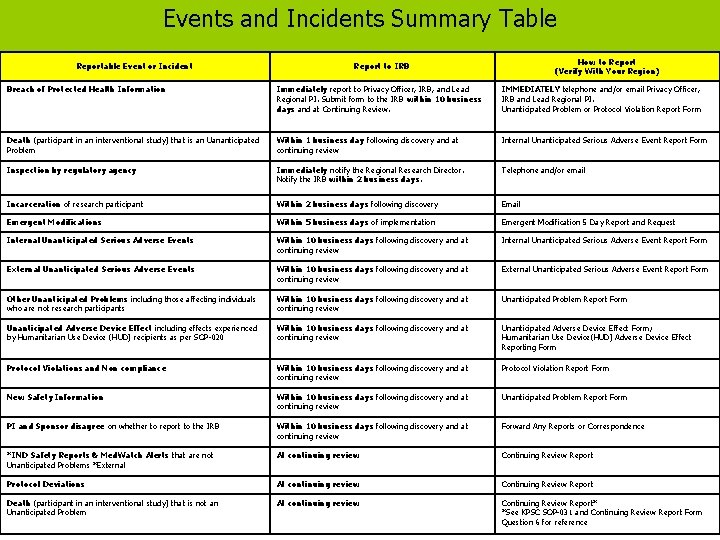
Events and Incidents Summary Table Reportable Event or Incident How to Report (Verify With Your Region) Report to IRB Breach of Protected Health Information Immediately report to Privacy Officer, IRB, and Lead Regional PI. Submit form to the IRB within 10 business days and at Continuing Review. IMMEDIATELY telephone and/or email Privacy Officer, IRB and Lead Regional PI. Unanticipated Problem or Protocol Violation Report Form Death (participant in an interventional study) that is an Uananticipated Problem Within 1 business day following discovery and at continuing review Internal Unanticipated Serious Adverse Event Report Form Inspection by regulatory agency Immediately notify the Regional Research Director. Notify the IRB within 2 business days. Telephone and/or email Incarceration of research participant Within 2 business days following discovery Email Emergent Modifications Within 5 business days of implementation Emergent Modification 5 Day Report and Request Internal Unanticipated Serious Adverse Events Within 10 business days following discovery and at continuing review Internal Unanticipated Serious Adverse Event Report Form External Unanticipated Serious Adverse Events Within 10 business days following discovery and at continuing review External Unanticipated Serious Adverse Event Report Form Other Unanticipated Problems including those affecting individuals who are not research participants Within 10 business days following discovery and at continuing review Unanticipated Problem Report Form Unanticipated Adverse Device Effect including effects experienced by Humanitarian Use Device (HUD) recipients as per SOP-020 Within 10 business days following discovery and at continuing review Unanticipated Adverse Device Effect Form; Humanitarian Use Device(HUD) Adverse Device Effect Reporting Form Protocol Violations and Non compliance Within 10 business days following discovery and at continuing review Protocol Violation Report Form New Safety Information Within 10 business days following discovery and at continuing review Unanticipated Problem Report Form PI and Sponsor disagree on whether to report to the IRB Within 10 business days following discovery and at continuing review Forward Any Reports or Correspondence *IND Safety Reports & Med. Watch Alerts that are not Unanticipated Problems *External At continuing review Continuing Review Report Protocol Deviations At continuing review Continuing Review Report Death (participant in an interventional study) that is not an Unanticipated Problem At continuing review Continuing Review Report* *See KPSC SOP-031 and Continuing Review Report Form Question 6 for reference

Responsibility for Identifying and Reporting Events and Incidents Principal Investigator Research Team Members Institutional Review Board Kaiser Foundation Research Institute 8

Principal Investigator’s Responsibilities for Reporting · Protect study participants’ rights, safety, and welfare · Document and manage potentially reportable events and incidents · Correctly identify, analyze, and report events and incidents to the IRB in the specified timeframe · Ensure that each Research Team Member obtains required training and understands how to identify, analyze, and report events and incidents to the PI Kaiser Foundation Research Institute 9

Research Team Members’ Responsibilities for Reporting · Identify reportable events and incidents · Immediately contact the PI or his/her designee about any event or incident that could be reportable § The PI makes the ultimate determination if an event or incident is reportable to the IRB · If the PI or Sub-investigator is unavailable and you are concerned that an event could pose harm to a research participant, contact your regional IRB Kaiser Foundation Research Institute 10

The IRB Reports to the Following Entities as Required by Law or Policy ● ● US Department of Health and Human Services, Office of Human Research Protections ● Regional Research Leadership n n FDA (for FDA regulated studies) Federalwide Assurance Official at KFRI (Vice President and Director of KFRI) Program Officer or Grant Manager (for federally funded studies) Kaiser Foundation Research Institute n n n Regional Research Director Regional Accountable Official Regional and/or local Research Chair PI’s Department Chair Regional PMG/KFHP/KFH Leaders ● Other Regional or Institutional Officials 11

The IRB May Take the Following Actions* · Suspend or terminate the research · Modify/monitor the research protocol · Modify/monitor the informed consent process and/or require reconsent · Notify present and/or past study participants · Consult with other organizational entities (legal, risk management, compliance, privacy officer, etc. ) · Modify the Continuing Review schedule · Gather information pending a final determination · Other actions as appropriate *SOP KP-003 IRB Reporting for Unanticipated Problems identifies the IRB follow-up process and requirements Kaiser Foundation Research Institute 12

Reportable Events and Incidents Unanticipated Problems Kaiser Foundation Research Institute 13

What is an Unanticipated Problem? When an event, incident or outcome meets ALL three of the following criteria It is an Unanticipated Problem 1. Unexpected in nature, severity, or frequency AND 2. Related or possibly related to participation in the research AND 3. Suggests greater risk of harm to participant(s) or others than previously known Kaiser Foundation Research Institute 14

Federal Regulations and KP Policy Require Reporting of Unanticipated Problems · Unanticipated Problems represent a risk of harm that is unexpected and possibly related to the study · The risk of harm may affect study participant(s) and/or others including: family members, Research Team Members, hospital staff, clinical staff, or others as determined by the PI · The PI must take action to protect the study participant(s) or others from the unexpected risk of harm Report Unanticipated Problems to the IRB within 10 Business Days of discovery and at Continuing Review Kaiser Foundation Research Institute 15

Three Steps to Identify an Unanticipated Problem Step 1. Determine if the event is Unexpected Step 2. Determine if the event is Related or possibly related Step 3. Determine if the event Suggests greater risk If an event is: Unexpected + Related or possibly related + Suggests greater risk = Reportable Unanticipated Problem Kaiser Foundation Research Institute 16

Step 1: Determine if the Event is Unexpected The PI determines if the event or incident is unexpected in nature, severity, or frequency, taking into consideration: ● The protocol-related documents: n n IRB-approved research protocol or research application Informed consent documents Investigator brochure Other sources of information such as product labeling and package inserts ● The PI’s knowledge of the characteristics of the study population ● The expected progression of any underlying diseases or conditions of the participants ● The participant’s pre-existing conditions and risk profile for the event Kaiser Foundation Research Institute 17

Step 1: Did Pre-existing Conditions Cause the Event? If the PI determines that a pre-existing condition(s) caused the event, the event is expected and is not an Unanticipated Problem. Does the participant’s clinical history indicate pre-existing conditions or risk factors? For example: ● Clinical conditions such as diabetes, coronary disease, hypertension, pulmonary disease, etc. ● Side-effects or interactions with drugs other than the study medication ● Substance use/abuse ● Environmental influences or risks Do Not Report the Event to the IRB Kaiser Foundation Research Institute 18

Step 2: If Unexpected, Determine if Related or Possibly Related The PI determines if the event is related or possibly related to the research by considering many factors including: ● Did the event occur close in time to the research intervention (drug, device, behavioral)? ● Did more than one participant experience the event? ● Did the event stop or lessen when the intervention was discontinued? If the PI determines the event is related or possibly related to the research, The PI Proceeds to Step Three Kaiser Foundation Research Institute 19

Step 3: If Unexpected and Related to the Research, Determine if the Event Suggests Greater Risk The PI determines if the event or incident could place the participant, family members, Research Team Members, or others at greater risk of harm than was previously known by considering many factors including: ● Is there a greater physical risk of harm? ● Is there a greater psychological risk of harm? ● Is there a greater economic risk of harm? ● Is there a greater social risk of harm? Kaiser Foundation Research Institute 20

Did the Event Meet All Three Criteria for an Unanticipated Problem? Unexpected + Related or possibly related + Suggests greater risk to participants or others = A Reportable Unanticipated Problem Report to the IRB within 10 business days and at Continuing Review Kaiser Foundation Research Institute 21

Unanticipated Problems Questions and Answers The next 12 slides provide examples and walk through the process of determining if an event is a reportable Unanticipated Problem. ● The question appears on the first slide and the answer, with a brief explanation, appears on the second slide ● The first 4 questions address a single event and the final 8 slides offer additional examples Kaiser Foundation Research Institute 22

Unanticipated Problems Question 1 The first four questions address this event. A defective investigational device caused serious harm to the investigator. The PI analyzes this event to determine if it is reportable. Step 1. Is this event unexpected? A) Yes B) No Kaiser Foundation Research Institute 23

Unanticipated Problems Answer 1 Step 1. Is this event unexpected? A) Yes, this event is unexpected. The investigator did not expect the device to be defective. Kaiser Foundation Research Institute 24

Unanticipated Problems Question 2 A defective investigational device caused serious harm to the investigator. The PI determined the event is unexpected. Step 2. Is this event related or possibly related to the research? A) Yes B) No Kaiser Foundation Research Institute 25

Unanticipated Problems Answer 2 Step 2. Is this event related or possibly related to the research? A) Yes, this event is related to the research. The investigational device is being used for the research study. Kaiser Foundation Research Institute 26

Unanticipated Problems Question 3 A defective investigational device caused serious harm to the investigator. The PI determined the event is unexpected and related to the research. Step 3. Does this event suggest greater risk of harm to participants or others than was previously known? A) Yes B) No Kaiser Foundation Research Institute 27

Unanticipated Problems Answer 3 Step 3. Does this event suggest greater risk of harm to participants or others than was previously known? A) Yes, this event suggests greater risk to participants or others – in this case the investigator. Kaiser Foundation Research Institute 28

Unanticipated Problems Question 4 A defective investigational device caused serious harm to the investigator. Is this event a reportable Unanticipated Problem? A) Yes B) No Kaiser Foundation Research Institute 29

Unanticipated Problems Answer 4 Is this event a reportable Unanticipated Problem? A) Yes, this event is an Unanticipated Problem. It is unexpected, related or possibly related to the research, and suggests greater risk of harm to participants or others than previously known. The PI reports an Unanticipated Problem within 10 business days and at Continuing Review. Kaiser Foundation Research Institute 30

Unanticipated Problems Question 5 A study participant’s 3 year-old took the study medication and became listless and unresponsive. Does the PI report this event to the IRB as an Unanticipated Problem? A) Yes B) No Kaiser Foundation Research Institute 31

Unanticipated Problems Answer 5 Does the PI report this event to the IRB as an Unanticipated Problem? A) Yes, this is an Unanticipated Problem because it is unexpected, related to study participation, and suggests greater risk to participants or others – in this case the 3 year-old. The PI reports Unanticipated Problems to the IRB within 10 days and at Continuing Review. Kaiser Foundation Research Institute 32

Unanticipated Problems Question 6 A Research Team Member experiences a study related needle stick. The study participant has hepatitis C. Does the PI report this event to the IRB as an Unanticipated Problem? A) Yes B) No Kaiser Foundation Research Institute 33

Unanticipated Problems Answer 6 Does the PI report this event to the IRB as an Unanticipated Problem? A) Yes, this is an Unanticipated Problem because it is unexpected, related to study participation, and suggests greater risk to participants or others – in this case the Research Team Member. The PI reports Unanticipated Problems to the IRB within 10 days and at Continuing Review. Kaiser Foundation Research Institute 34

Reportable Events and Incidents Adverse Events Serious Adverse Events Kaiser Foundation Research Institute 35

What is an Adverse Event? Adverse Events are unfavorable medical or psychological events experienced by a study participant during clinical research: ● ● ● A new symptom Worsening of an existing condition A clinically significant abnormal lab finding Kaiser Foundation Research Institute 36

What is a Serious Adverse Event? Serious Adverse Events include: ● Death ● Life threatening condition/situation ● New or prolonged hospitalization ● An enduring or significant incapacity or substantial disruption of the ability to conduct normal life functions ● The delivery of a child with congenital anomaly or birth defect ● Other medical events that the PI determines require intervention to prevent the above outcomes Kaiser Foundation Research Institute 37

Report Only Adverse Events that are Unanticipated Problems Adverse Events are not reportable to the IRB unless they meet all three criteria for an Unanticipated Problem. Unexpected + Related or possibly related + Suggests greater risk to participants or others = A Reportable Unanticipated Problem Report to the IRB within 10 business days and at Continuing Review Kaiser Foundation Research Institute 38

Report Adverse Events Requiring Significant Modification If the PI determines that an Adverse Event or Serious Adverse Event requires significant modification to the brochure, study protocol, or informed consent, to ensure patient safety – it is an Unanticipated Problem. Report to the IRB within 10 business days and at Continuing Review Kaiser Foundation Research Institute 39

Analyzing Adverse Events Questions and Answers The next 10 slides include five examples of Adverse Events or Serious Adverse Events. · Using what you know about Unanticipated Problems, analyze each event to determine if it should be reported to the IRB Kaiser Foundation Research Institute 40

Analyzing Adverse Events Question 1 Five interventional study participants develop severe anemia, requiring hospitalization. ● ● ● The consent form does not identify anemia as a study risk The participants have no risk factors for anemia None of the control group participants have shown evidence of anemia Conduct your analysis of this event. Would the PI report to the IRB? A) No. The PI would not report because the event was expected. It does not meet all three criteria for an Unanticipated Problem. B) Yes. The PI reports the event because it meets all three criteria of an Unanticipated Problem. Kaiser Foundation Research Institute 41

Analyzing Adverse Events Answer 1 Would the PI report to the IRB? B) Yes. The PI reports the event because it meets all three criteria of an Unanticipated Problem and should be reported to the IRB within 10 business days and at Continuing Review. The consent form does not mention anemia, and the participants do not have risk factors for anemia. The event is unexpected. Multiple participants experienced the same event. There is no anemia in the control group. The event is possibly related to the research. Hospitalization and severe anemia in five participants suggests greater risk to participants. Kaiser Foundation Research Institute 42

Analyzing Adverse Events Question 2 In a psychology study, participants are in a small, windowless, soundproof booth. One participant experiences claustrophobia while in the booth. ● The consent form describes claustrophobic reactions as one of the study risks Conduct your analysis of this event. Would the PI report to the IRB? A) Yes. The adverse event is expected + related + suggests greater risk. B) Yes. The adverse event is unexpected + related + suggests greater risk. C) No. The adverse event is expected + related + does not suggest greater risk. Kaiser Foundation Research Institute 43

Analyzing Adverse Events Answer 2 Would the PI report to the IRB? C) No. The adverse event is expected + related + does not suggest greater risk. The PI does not report as an Unanticipated Problem. The event is expected because the informed consent describes claustrophobic reactions as one of the study risks. The proximity in time indicates that the event is related to study participation. The event does not suggest greater risk than previously known. Claustrophobia was an expected risk identified in the consent form. Kaiser Foundation Research Institute 44

Analyzing Adverse Events Question 3 Three of the first ten study participants develop severe gastroesophageal reflex disease (GERD) within one week of beginning the study. ● ● The consent form does not identify GERD as a study risk After the drug is discontinued, the GERD symptoms resolve Conduct your analysis of this event. Would the PI report to the IRB? A) Yes. The event is expected + related + suggests greater risk. B) Yes. The event is unexpected + related + suggests greater risk. C) No. The event is expected + related + suggests no greater risk. Kaiser Foundation Research Institute 45

Analyzing Adverse Events Answer 3 Would the PI report to the IRB? B) Yes. The event is unexpected + related + suggests greater risk. The PI reports an Unanticipated Problem within 10 days and at Continuing Review. The consent form does not mention GERD. The event is unexpected. Several factors suggest that event is related to the research: proximity in time, 3 participants experienced the event, and the symptoms resolved after the drug was discontinued. The GERD symptoms are severe, this suggests greater risk to study participants. Kaiser Foundation Research Institute 46

Analyzing Adverse Events Question 4 One of the study participants required hospitalization from kidney failure after beginning the study. ● The consent form mentions possible kidney problems, but not the level of severity the participant is experiencing ● The participant did not have any risk factors for kidney failure Conduct your analysis of this event. Would the PI report to the IRB? A) Yes. The event is expected + related + suggests greater risk. B) Yes. The event is unexpected + related + suggests greater risk. C) No. The event is expected + related + suggests no greater risk. Kaiser Foundation Research Institute 47

Analyzing Adverse Events Answer 4 Would the PI report to the IRB? B) Yes. The event is unexpected + related + suggests greater risk. The PI reports an Unanticipated Problem within 10 days and at Continuing Review. The event is unexpected because it is more frequent or severe than the description in the consent form. The event is possibly related because the participant had no risk factors for kidney failure. The severity of the event suggests greater risk to study participants. Kaiser Foundation Research Institute 48

Analyzing Adverse Events Question 5 Three experimental group participants develop a rash that lasts for 3 hours. ● The consent form does not mention a rash as a study risk ● The participants have no risk factors for a rash ● Three control group participants also develop a rash Conduct your analysis of this event. Would the PI report to the IRB? A) Yes. The event is expected + related + suggests greater risk. B) No. The event is unexpected + not related + suggests no greater risk. C) Yes. The event is unexpected + related + suggests greater risk. Kaiser Foundation Research Institute 49

Analyzing Adverse Events Answer 5 Would the PI report to the IRB? B) No. The event is unexpected + not related + suggests no greater risk. The PI does not report as an Unanticipated Problem. The event is unexpected because the consent form does not identify a rash as a risk and the participants do not have risk factors for a rash. The PI’s analysis indicates that the event is not related to study participation because three control group members also experienced a rash. The rashes are not serious and do not suggests greater risk to study participants. Kaiser Foundation Research Institute 50

Other Reportable Unanticipated Problems When PIs and Sponsors Disagree IND Safety Report (External Adverse Event) Med. Watch Alert (External Adverse Event) Death of a Study Participant Kaiser Foundation Research Institute 51

When PIs and Sponsors Disagree The PI determines that an Adverse Event is not an Unanticipated Problem. The sponsor disagrees and notifies the PI, in writing, that the event is an Unanticipated Problem. The PI sends all reports and correspondence related to the Adverse Event to the IRB. Send Report to the IRB within 10 business days The IRB determines if follow-up action is required. Kaiser Foundation Research Institute 52

Example: When PIs and Sponsors Disagree A study participant experiences a nose bleed that lasts 2 hours. The PI reports this to the sponsor as an Adverse Event, but states that it does not place participants at greater risk and is not reportable as an Unanticipated Problem. The sponsor disagrees and wants the Adverse Event reported to the IRB. The PI sends all reports and correspondence related to the Adverse Event to the IRB within 10 business days The IRB determines if follow-up action is required. Kaiser Foundation Research Institute 53

Reporting IND Safety Reports or Med. Watch Alerts (External Adverse Events) Sponsors release an Investigational New Drug (IND) Safety Report to notify PIs and the FDA of an Adverse Event related to an investigational agent. The FDA releases Med. Watch Alerts to provide new safety information for FDA approved drugs, medical devices, vaccines and other biologics. The PI conducts an independent analysis of the IND Safety Report and the Med. Watch Alert. ● If the PI determines that the Adverse Event is an Unanticipated Problem, the PI reports to the IRB within 10 business days and at Continuing Review ● If the PI determines that the Adverse Event is not an Unanticipated Problem, the PI reports to the IRB at Continuing Review Kaiser Foundation Research Institute 54

Reporting the Death of a Participant Depends on Study Type and Unanticipated Problem Analysis 1. Death of an Interventional Study Participant + Unanticipated Problem = Report to the IRB within 1 business day of discovery and at Continuing Review 2. Death of an Interventional Study Participant + Not an Unanticipated Problem = Report at Continuing Review 3. Death of a Non-interventional Study Participant = Not Reportable Kaiser Foundation Research Institute 55

Reporting Time Frames Questions and Answers The next 4 slides provide 2 examples. To determine the IRB reporting time frame consider: ● The type of study ● Whether the event is an Unanticipated Problem Kaiser Foundation Research Institute 56

Reporting Time Frames Question 1 An interventional study participant is a passenger in a car accident. She dies from her injuries. When does the PI report the death to the IRB? A) At the time of Continuing Review B) Within one business day of discovery and at Continuing Review C) The PI does not report the death Kaiser Foundation Research Institute 57

Reporting Time Frames Answer 1 When does the PI report the death to the IRB? A) At the time of Continuing Review The death occurred in an Interventional study and is not an Unanticipated Problem. The death does not meet all three criteria for an Unanticipated Problem. The death is not related to study participation. The death does not suggest greater risk to other study participants. Kaiser Foundation Research Institute 58

Reporting Time Frames Question 2 An interventional study participant dies from kidney failure after taking the investigational drug. ● Kidney failure is not a known study risk ● The participant had no risk factors for kidney failure When does the PI report the death to the IRB? A) At the time of Continuing Review B) Within one business day of discovery and at Continuing Review C) The PI does not report the death Kaiser Foundation Research Institute 59

Reporting Time Frames Answer 2 When does the PI report the death to the IRB? B) Within one business day of discovery and at Continuing Review The death occurred in an Interventional study and is an Unanticipated Problem. The death meets all three criteria for an Unanticipated Problem. The event is unexpected, related to study participation and suggests greater risk to other study participants. Kaiser Foundation Research Institute 60

Reportable Events and Incidents Protocol Violations Protocol Deviations Kaiser Foundation Research Institute 61

Report Protocol Violations A Protocol Violation is a departure from the IRB approved research plan that: ● Places the safety, rights, or welfare of one or more study participants at risk And/Or ● Could impact the integrity of the study Report Protocol Violations within 10 business days of discovery and at Continuing Review Kaiser Foundation Research Institute 62

Examples: Protocol Violations 1. The inclusion/exclusion criteria were not used correctly to include or exclude participants ● If study participants do not meet the inclusion criteria it could result in harm to the participants and/or impact the integrity of the study 2. The PI used an old version of the consent form ● An old version of the consent form does not represent the current IRB approved form and may be missing important information. This could place the safety, rights, or welfare of study participants at risk and impact the integrity of the study Report Protocol Violations within 10 business days of discovery and at Continuing Review Kaiser Foundation Research Institute 63

Additional Examples of Protocol Violations A Protocol Violation places the safety, rights, or welfare of one or more study participants at risk and/or impacts the integrity of the study. ● A participant received the incorrect study intervention ● A researcher did not obtain the participant’s signature and date on the consent form before initiating screening activities ● A research staff improperly dispensed an investigational agent ● Multiple incidents of protocol deviations reflect a pattern that constitute a Protocol Violation Kaiser Foundation Research Institute 64

Report Protocol Deviations The PI determines if a protocol departure is a Violation or a Deviation. An incident that is a violation in one situation may be a deviation in another. A Protocol Deviation is a departure from the IRB approved research plan that: ● Does not place the safety, rights, or welfare of one or more study participants at risk And ● Does not impact the integrity of the study Report Protocol Deviations at Continuing Review Kaiser Foundation Research Institute 65

Examples: Protocol Deviations 1. The investigator conducts the procedure in the wrong sequence. The PI determines that: ● The change did not place the safety, rights, or welfare of study participants at risk And ● Did not impact the integrity of the study 2. The investigator did not perform a required lab test. The PI determines that: ● The missing lab test did not place the safety, rights, or welfare of study participants at risk And ● Did not impact the integrity of the study Report Protocol Deviations at Continuing Review Kaiser Foundation Research Institute 66

Other Reportable Events and Incidents Breach of Protected Health Information Incarceration of a Study Participant New Safety Information Emergent Modifications Regulatory Visits/Inspections Kaiser Foundation Research Institute 67

Required Reporting of Breaches A Breach is the unauthorized acquisition, access, use, or disclosure of PHI that compromises privacy or security. There are numerous federal regulations that may apply to a breach, including the Common Rule, FDA regulations, Privacy Rule, Security Rule, CMS regulations, and others. Some of these regulations require KP to notify the appropriate regulatory agency within as soon as 24 hours following the discovery of the breach. Failure to report within the required time frame could result in significant fines for KP and possible civil and criminal penalties. Kaiser Foundation Research Institute 68

How to Report a Breach of PHI The PI reports a Breach: ● IMMEDIATELY by phone or email to the Regional Privacy and Security Officer, the IRB, and the Lead Regional PI for multiregion studies ● To the IRB as a Protocol Violation or an Unanticipated Problem within 10 business days and at Continuing Review Note: Any breach of a Limited Data Set must be reported because it contains PHI. Kaiser Foundation Research Institute 69

Reporting a Breach to the IRB The PI reports a Breach to the IRB as a Protocol Violation or an Unanticipated Problem within 10 business days and at Continuing Review. ● A Protocol Violation places the safety, rights, or welfare of one or more study participants at risk and/or impacts the integrity of the study ● An Unanticipated Problem is unexpected, related or possibly related to participation in the research and suggests greater risk of harm to participants or others Kaiser Foundation Research Institute 70

Report Participant Incarceration When a study participant is incarcerated, the participant’s status changes to “vulnerable population. ” • Additional federal regulations apply to vulnerable populations • Stop all study interactions, interventions, and data collection Exception: The PI and IRB Chair may determine that it is in the participant’s best interest to continue in the study until other arrangements can be made. Report participant incarceration to the IRB within 2 business days of discovery Kaiser Foundation Research Institute 71

Report New Safety Information that is an Unanticipated Problem The PI assesses New Safety Information to determine if it meets the three criteria of an Unanticipated Problem. Usually, New Safety Information reveals increased risk or reduced benefit to participants discovered during the course of the study. Report New Safety Information that is an Unanticipated Problem to the IRB within 10 business days and at Continuing Review Kaiser Foundation Research Institute 72

Report Emergent Modifications are changes made to the protocol or conduct of a study to protect the safety, rights, or welfare of research participants and/or others. When an immediate hazard exists, the PI may modify the research plan to protect the participants prior to receiving approval from the IRB. The PI takes the necessary action to protect the study participants and others. Report to the IRB within 5 business days of implementation Emergent Modifications are RARE! Kaiser Foundation Research Institute 73

Report Regulatory Audits or Inspections Unannounced or scheduled Audits or Inspections may occur at any time. Immediately notify the Regional Research Director Report to the IRB within 2 business days of notification Follow KP Policy when meeting auditors or inspectors. 1. 2. 3. 4. Immediately ask for identification Provide a place to wait until a department manager is available Do not provide access to PHI or confidential business information, until an authorized individual has verified that the auditor or inspector has the right to access information Always treat the individual with courtesy and respect. If you are concerned that his/her behavior is inappropriate, contact building security. Kaiser Foundation Research Institute 74

Resources Copy and paste the URLs below into your browser. · SOP KP-502, Version 1, Principal Investigator Reportable Event · · and Incident Requirements http: //kpnet. kp. org/kfri/sops/all. htm Kaiser Foundation Research Institute and KP Regional Research Links http: //kpnet. kp. org/kfri/about/index. htm Reviewing and Reporting Unanticipated Problems and Adverse Events: On HHS You. Tube Channel: http: //www. youtube. com/watch? v=hs. US 0 k 3 Ie_g Note: If your computer does not have the necessary KP intranet or internet access you will not be able to access the links above. Kaiser Foundation Research Institute 75
Congratulations! You have completed the course. Notify your Research Training Coordinator that you have completed the course. Click the X button in the upper right corner to close this window. Kaiser Foundation Research Institute 76

Kaiser Permanente Southern California Documentation of Completion n In order to receive credit for completing this training, you must complete this page with your handwritten signature, indicating that you have read and understood the content of this Reportable Events and Incidents Research Compliance Training Step 1: Email a copy of this complete and signed Documentation of Completion Page in PDF format from your KP. ORG email to KPSC. IRB@kp. org. Step 2: After two weeks, you must upload in i. RIS the complete and signed Documentation of Completion Page in PDF format to your i. RIS My Account Information, under Education History. v If you are a new user, go to http: //irissupport. kp-scalresearch. org/ to get a username and password. Click on the orange “new users” button located on the right hand side of your screen. v If you already have an i. RIS username and password, log on to the i. RIS home page http: //iris. kp-scalresearch. org/. ___________ Printed Name ____________ Signature (in blue or black ink only) ______________________ KP Location and Department Kaiser Foundation Research Institute KPSC Version 2 Dated 8/15/2012 _______ Date _________ Phone number 77
- Slides: 77