RennerTeller and SpinOrbit Coupling in H 2 S

Renner-Teller and Spin-Orbit Coupling in H 2 S+ and As. H 2 G. Duxbury 1, Christian Jungen 2 and Alex Alijah 3 1 Department of Physics, University of Strathclyde, John Anderson Building, 107 Rottenrow, Glasgow, G 4 0 NG, UK 2 LAC, Laboratoire Aime Cotton du CNRS, Universite de Paris -Sud, 91405 Orsay France 3 UMR CNRS 6089, Universite der Reims, Champagne. Ardenne, BP. 1039, 51687 Reims Cedex 2, France 19/06/2013 MOLSPECT 2013 WG 08 4: 02 pm

Outline of the presentation • As. H 2: • Photographic record of the electronic spectrum of As. H 2 • Comparison of term values derived from ringdown Zhao et al. and from flash photolysis, Dixon et al. • H 2 S+: • Comparison of PES, electron beam induced emission, and recent MATI results 19/06/2013 MOLSPECT 2013 WG 08 4: 02 pm

Analysis of the 2 A 1 to 2 B 1 electronic band system of the As. H 2 Radicals • The absorption spectrum of this band system was first recorded in absorption by Dixon, Duxbury and Lamberton, Proc. Roy. Soc. A 305, 271 (1968). • Apart from one limited series of measurement of its emission spectrum By T Ni and colleagues in 1986 (Chem. Phys. Lett. 126, 417 -420), no detailed studies were carried out until the period from 2000. 19/06/2013 MOLSPECT 2013 WG 08 4: 02 pm

As. H 2 Absorption spectrum recorded using flash photolysis, 1966 -67. The bands analysed were v 2=1 to v 2=3. From v 2=3 to v 2=6 the predissociation increases rapidly, so that above v 2=6 no absorption spectra could be recorded. 19/06/2013 MOLSPECT 2013 WG 08 4: 02 pm

As. H 2 Spectra 2000 onwards • Hughes, Brown and Evenson, “Rotational spectrum of the As. H 2 radical in its ground state”. J. Mol. Spectrosc, 200, 210 (2000) • He and Cloutier, “Laser spectroscopy and dynamics of the jet-cooled As. H 2 free radical”, J. Chem. Phys, 126, 154312 (2007) • Grimminger and Clouthier, As. D 2 and As. HD, J. Chem. Phys, 137, 224307 (2012) • Zhao, Qin, Ji, Zhang and Chen, “Absorption spectra of the As. H 2 radical in 435 -510 nm by cavity ringdown spectroscopy”, J. Mol. Spectrosc. 256, 192 (2009) 19/06/2013 MOLSPECT 2013 WG 08 4: 02 pm
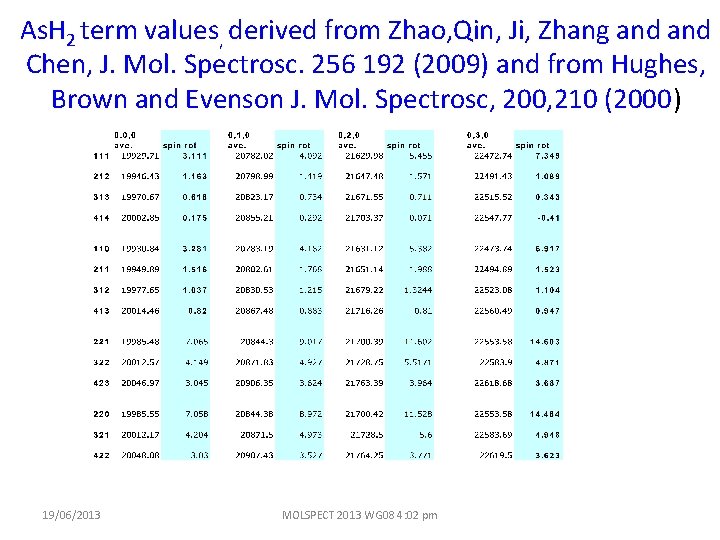
As. H 2 term values, derived from Zhao, Qin, Ji, Zhang and Chen, J. Mol. Spectrosc. 256 192 (2009) and from Hughes, Brown and Evenson J. Mol. Spectrosc, 200, 210 (2000) 19/06/2013 MOLSPECT 2013 WG 08 4: 02 pm
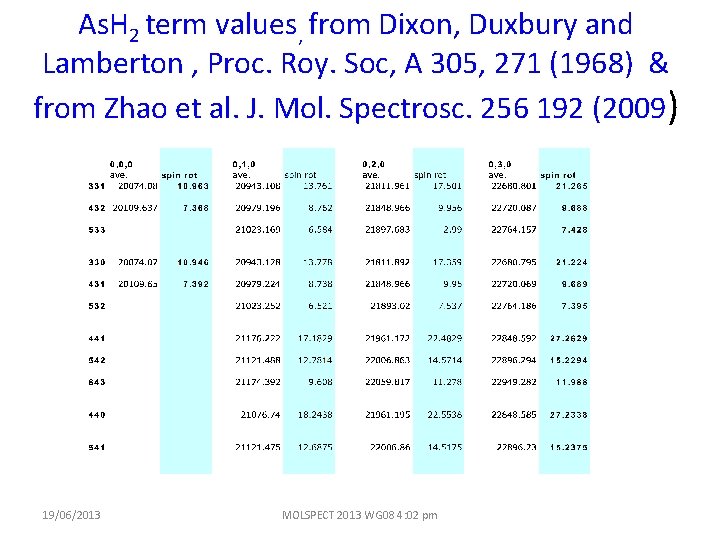
As. H 2 term values, from Dixon, Duxbury and Lamberton , Proc. Roy. Soc, A 305, 271 (1968) & from Zhao et al. J. Mol. Spectrosc. 256 192 (2009) 19/06/2013 MOLSPECT 2013 WG 08 4: 02 pm
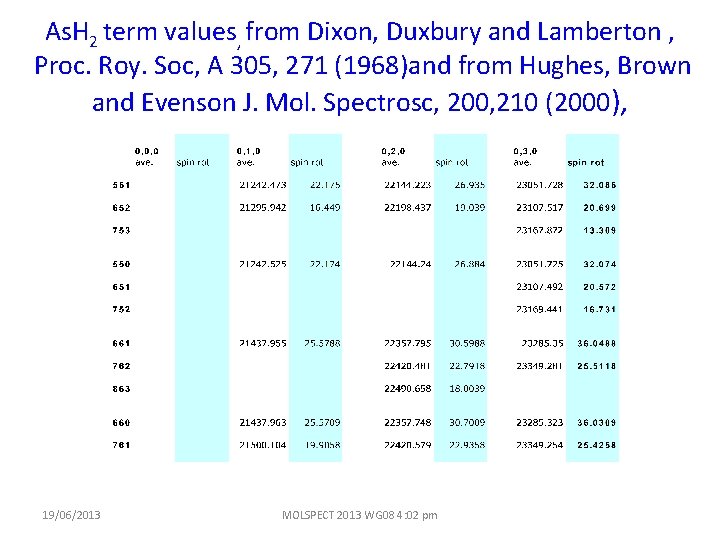
As. H 2 term values, from Dixon, Duxbury and Lamberton , Proc. Roy. Soc, A 305, 271 (1968)and from Hughes, Brown and Evenson J. Mol. Spectrosc, 200, 210 (2000), 19/06/2013 MOLSPECT 2013 WG 08 4: 02 pm
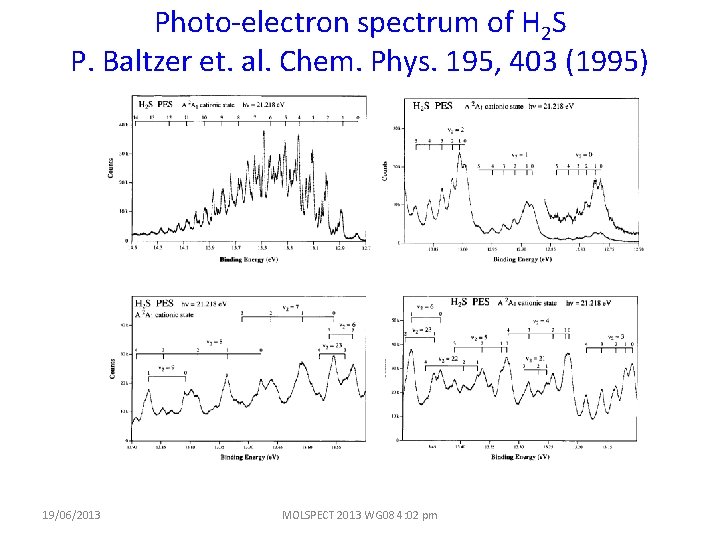
Photo-electron spectrum of H 2 S P. Baltzer et. al. Chem. Phys. 195, 403 (1995) 19/06/2013 MOLSPECT 2013 WG 08 4: 02 pm
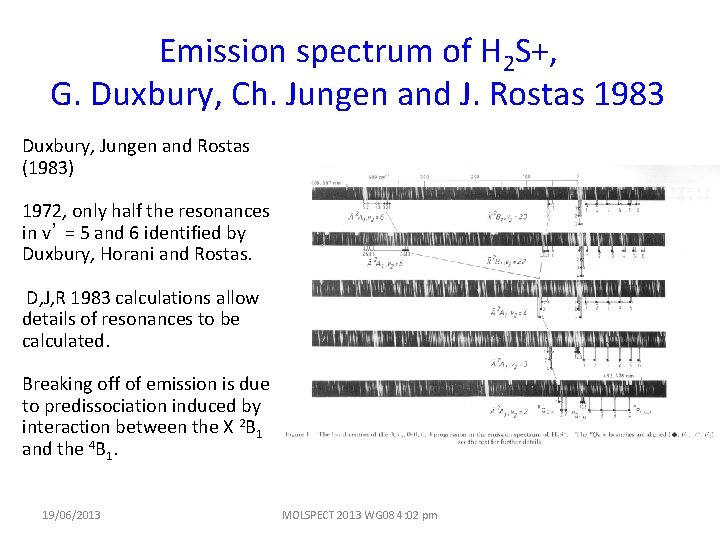
Emission spectrum of H 2 S+, G. Duxbury, Ch. Jungen and J. Rostas 1983 Duxbury, Jungen and Rostas (1983) 1972, only half the resonances in v’ = 5 and 6 identified by Duxbury, Horani and Rostas. D, J, R 1983 calculations allow details of resonances to be calculated. Breaking off of emission is due to predissociation induced by interaction between the X 2 B 1 and the 4 B 1. 19/06/2013 MOLSPECT 2013 WG 08 4: 02 pm
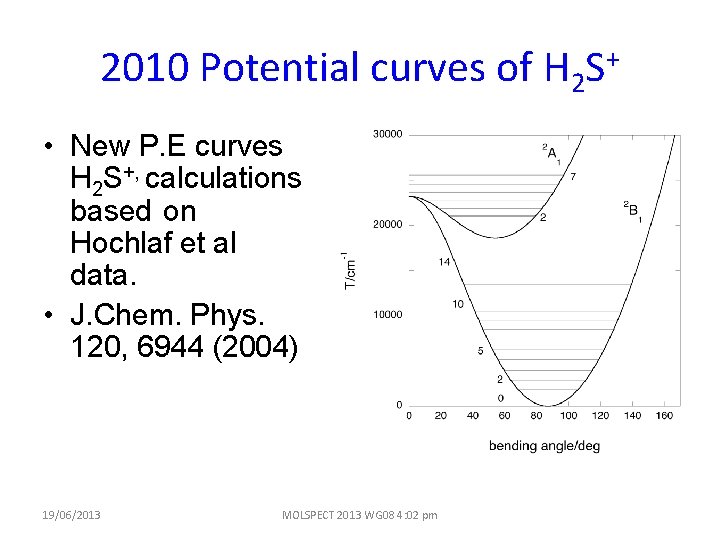
2010 Potential curves of H 2 S+ • New P. E curves H 2 S+, calculations based on Hochlaf et al data. • J. Chem. Phys. 120, 6944 (2004) 19/06/2013 MOLSPECT 2013 WG 08 4: 02 pm
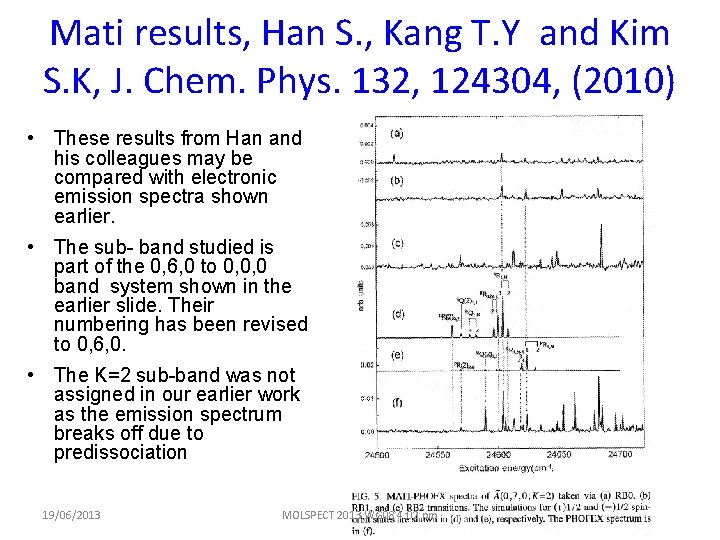
Mati results, Han S. , Kang T. Y and Kim S. K, J. Chem. Phys. 132, 124304, (2010) • These results from Han and his colleagues may be compared with electronic emission spectra shown earlier. • The sub- band studied is part of the 0, 6, 0 to 0, 0, 0 band system shown in the earlier slide. Their numbering has been revised to 0, 6, 0. • The K=2 sub-band was not assigned in our earlier work as the emission spectrum breaks off due to predissociation 19/06/2013 MOLSPECT 2013 WG 08 4: 02 pm

A comparison of the analysis of the fluorescence (DJR) and MATI (HKK)spectra 19/06/2013 MOLSPECT 2013 WG 08 4: 02 pm

Conclusion • The main points of this presentation are: • The comparison between the different experimental methods of investigating the spectrum of As. H 2 and of the hydrogen sulphide ion. • Different barrier heights give rise to very different spectra • The need to use an appropriate partitioning of the large amplitude Hamiltonian. The rapid increase of the splitting of the K’=0 and K’=1 subbands of H 2 S+ after the barrier to linearity is reached demonstrates the changeover to quasilinear behaviour for v 2’=5 and above. • The high resolution MATI spectra of Han, Kang and Kim allow higher ro-vibronic levels to be seen above the predissociation threshold, in particular for v 2’= 7 and above. 19/06/2013 MOLSPECT 2013 WG 08 4: 02 pm
- Slides: 14