Renal Pathophysiology Briefly kidney anatomy physiology and function

Renal Pathophysiology

Briefly: kidney anatomy, physiology and function Pathology of filtration Pathology of reabsorption and secretion Classification of kidney’s diseases Clinical Syndromes Glomerular diseases Diseases affecting tubules and interstitium Renal Failure: acute and chronic

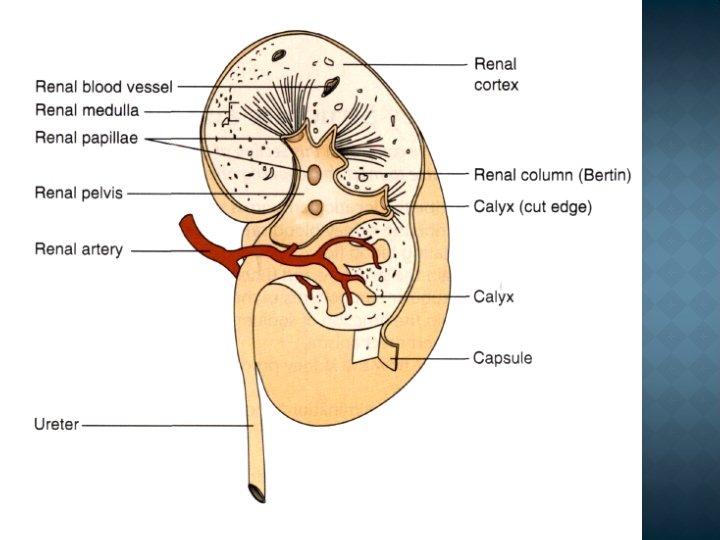
KIDNEY The kidney is a structurally complex organ that has evolved to carry out a number of important functions: excretion of the waste products of metabolism, regulation of body water and salt, maintenance of appropriate acid balance, secretion of a variety of hormones and autacoids.
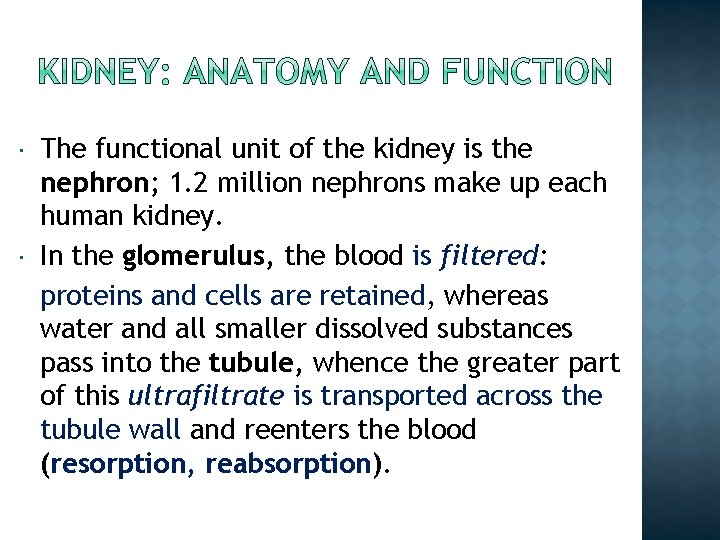
The functional unit of the kidney is the nephron; 1. 2 million nephrons make up each human kidney. In the glomerulus, the blood is filtered: proteins and cells are retained, whereas water and all smaller dissolved substances pass into the tubule, whence the greater part of this ultrafiltrate is transported across the tubule wall and reenters the blood (resorption, reabsorption).

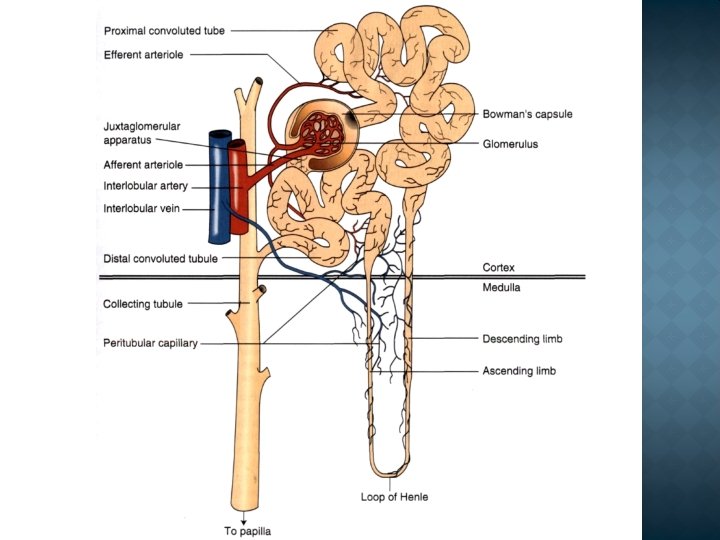
The fraction that is not resorbed remains in the tubule and appears in the terminal urine (excretion). Some urinary solvents enter the nephron lumen from tubule cells by secretion. 1. The renal corpuscle is situated in the renal cortex and is made up of Bowman's capsule and the glomerulus 2. The proximal tubule 3. The loop of Henle 4. The distal tubule

Homeostatic � To maintain constancy of ECF volume and of osmolality by balancing intake and excretion of Na+ and water. � Acid-base balance Excretory � excretes end products of metabolism (e. g. urea, uric acid, creatinine) and xenobiotics Metabolic � metabolic functions (e. g. arginine formation, gluconeogenesis, peptide hydrolysis) � source of hormones (e. g. , angiotensin II, erythropoietin, D-hormone, prostaglandins). Regulatory � BP regulation � Erythropoesis � Coagulation (urokinase)

Filtration Reabsorption Secretion Concentration and Dilution in the Kidney

In kidneys filtrated 150 -180 L /day Glomerular filtration rate (GFR) is the volume/time that is filtered by all glomeruli. An average of one fifth or 20 % of the renal plasma flow (RPF) is filtered at the glomerulus. This ratio, GFR/RPF, is called filtration fraction (FF).

Filtration depended from: Quantity of functional nephrons Characteristics of renal blood flow Level of filtration pressure (FP) FP = HP – (OP + ITP) = 75 mm. Hg – (25 mm. Hg + 10 mm. Hg) = 40 mm Hg

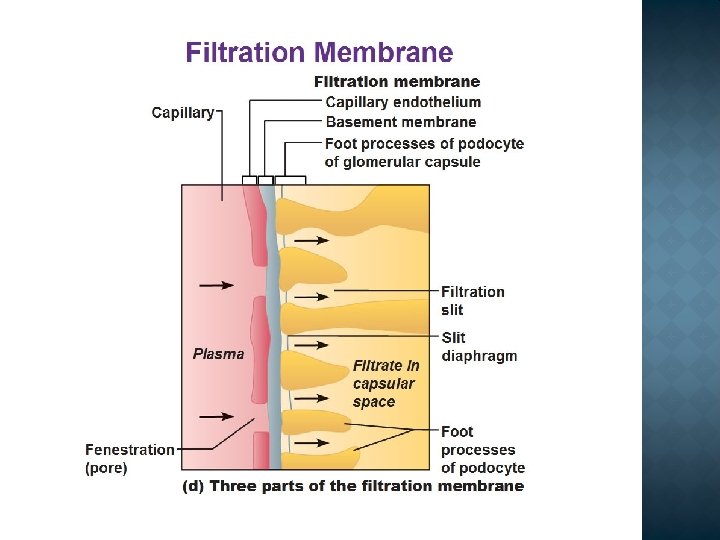
The major characteristics of glomerular filtration are an extraordinarily high permeability to water and small solutes and an almost complete impermeability to molecules of the size and molecular charge of albumin (size: 3. 6 nm radius; 70, 000 k. D). The selective permeability, called glomerular barrier function, discriminates among protein molecules depending on their size (the larger, the less permeable), their charge (the more cationic, the more permeable), and their configuration.

Decrease filtration Hypovolemia Constriction of the arteriole (vas afferens) afferent Decrease BP ( ↓ 70 mm Hg) ↓ Quantity of functional nephrons ↑ OP ↑ ITP
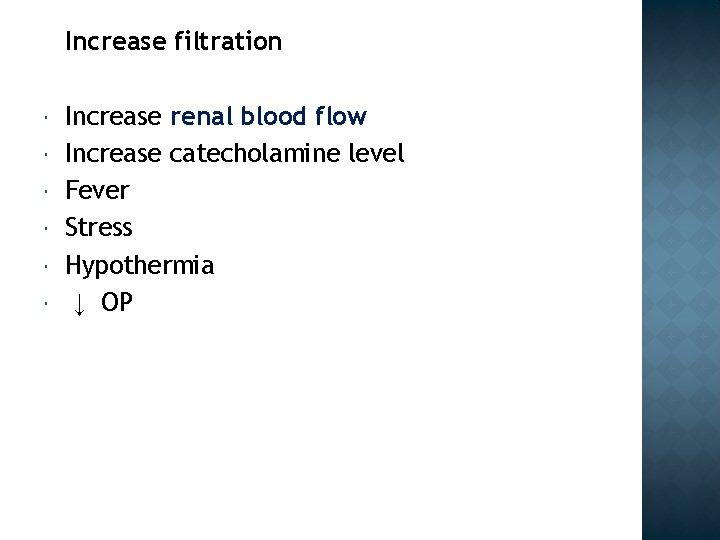
Increase filtration Increase renal blood flow Increase catecholamine level Fever Stress Hypothermia ↓ OP

Epithelium destruction Pathology of regulation Enzymopathias (K – Na pump) Energy deficiency Tubulopathias

Pathology of Na+ metabolism Na+ loss Endocrinopathies Enzymopathias (K+–Na+ pump) Epithelium destruction Activation of aldosterone antagonists (progesteron) Consequences - hypovolemia

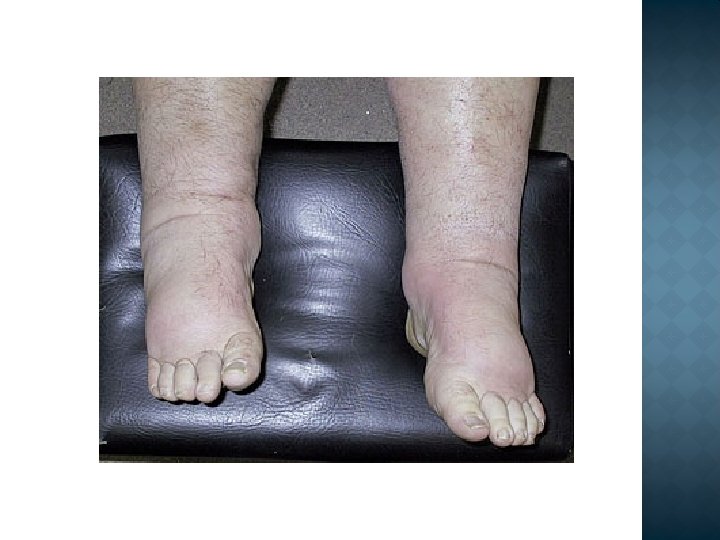
Na accumulation - ↓ filtration - ↑ reabsorption (hyperaldosteronism) Consequences - oligouria - hyperhydration - edema - hypervolemia → BP ↑

In kidneys filtrate near 180 L of H 2 O and 99% reabsorpted Cumulation of water - ↓ filtration - ↑ reabsorption (hyperaldosteronism, ↑ ADH) Consequences Water excess

Loss H 2 O ↑ filtration ↓ reabsorbtion tubulopathias Consequences - polyuria - hypohydration

The kidney is able to concentrate urine because of the creation of a hyperosmotic medullary interstitium and the presence of receptors on the collecting ducts for antidiuretic hormone (ADH). In the presence of ADH, there is an increase in the number of protein water channels expressed on the luminal membrane, and water moves into the cell down its concentration gradient.

2 variants of pathology ↓ ADH → ↓ reabsorption → ↑ urine ↑ ADH → ↑ reabsorption → ↓ urine Tests for diagnostic 24 hour urine specimen Isoosmotic urine - Hypoosmotic urine(↓ specific gravity of urine) - Nocturia -
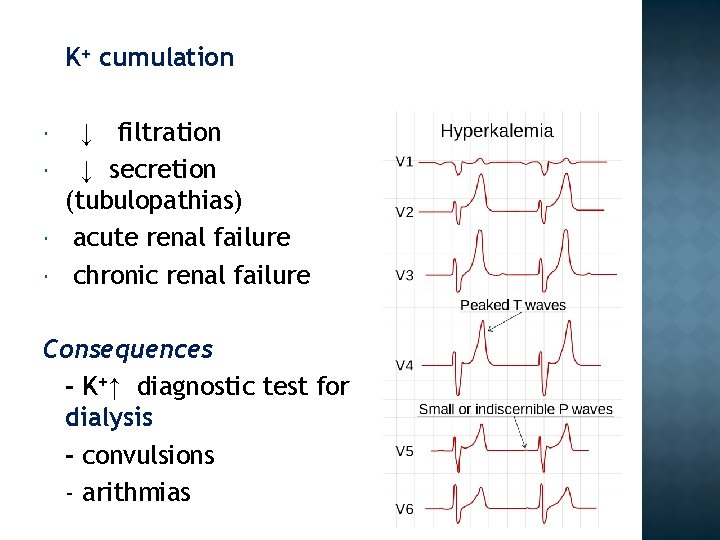
K+ cumulation ↓ filtration ↓ secretion (tubulopathias) acute renal failure chronic renal failure Consequences - K+↑ diagnostic test for dialysis - convulsions - arithmias
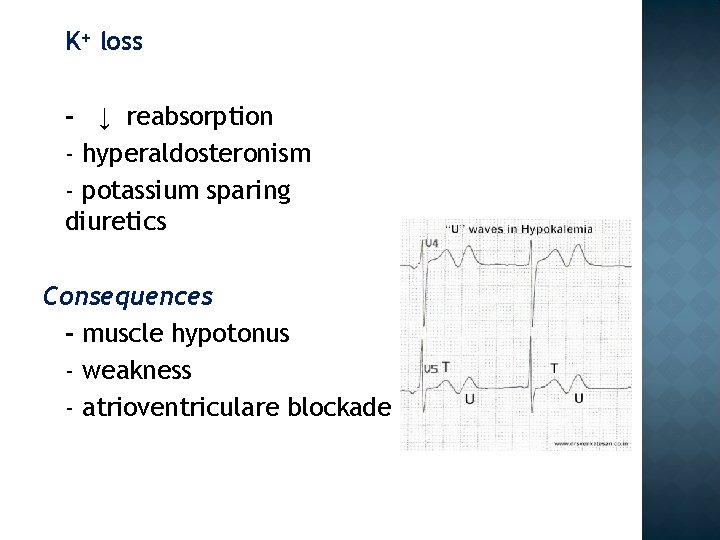
K+ loss - ↓ reabsorption - hyperaldosteronism - potassium sparing diuretics Consequences - muscle hypotonus - weakness - atrioventriculare blockade
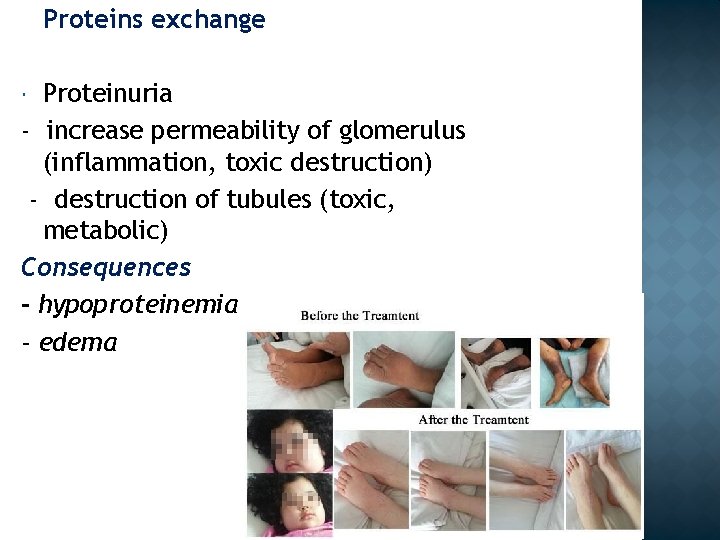
Proteins exchange Proteinuria - increase permeability of glomerulus (inflammation, toxic destruction) - destruction of tubules (toxic, metabolic) Consequences - hypoproteinemia - edema

Ca 2+ metabolism Vit D ↓ → reabsorption ↓ H+ ions Ø acidogenesis ↓ Ø ammoniogenesis ↓ Ø acidosis → Ca ↓

Bone Metabolism ↓GFR leads to ↑ phosphate ↓ calcium + acidosis Hypocalcemia Decreased dihydroxy-vitamin D production Bone acts as a buffer for acidosis, leading to chronic bone loss in renal failure Low vitamin D causes poor calcium absorbtion and hyperparathyroidism (high PTH) Increased PTH maintains normal serum Ca 2+ and PO 42 - until GFR <30 m. L/min Chronic hyperparathyroidism and bone buffering of acids leads to severe osteoporosis

DISEASES OF THE KIDNEY: v v v Diseases of of glomeruli, tubules, interstitium, blood vessels. Glomerular diseases are often immunologically mediated, whereas tubular and interstitial disorders are more likely to be caused by toxic or infectious agents.

RENAL PATHOLOGY Glomerular diseases: Glomerulonephritis Tubular diseases: Acute tubular necrosis Interstitial diseases: Pyelonephritis Diseases involving blood vessels: Nephrosclerosis Cystic diseases Tumors

CLINICAL SYNDROMES

CLINICAL SYNDROMES: The nephrotic syndrome Acute nephritic syndrome Asymptomatic hematuria or proteinuria Rapidly progressive glomerulonephritis Acute renal failure Chronic renal failure Urinary tract infection Nephrolithiasis (renal stones)

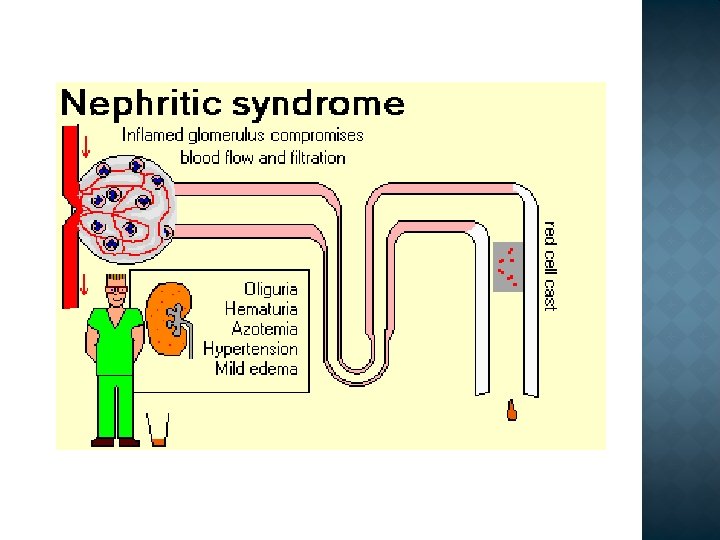
Differential diagnosis Nephrotic syndrome Ø Ø Massive proteinuria Hypoalbuminemia Edema Hyperlipidemia/-uria Nephritic syndrome Ø Ø Hematuria Oliguria Azotemia Hypertension
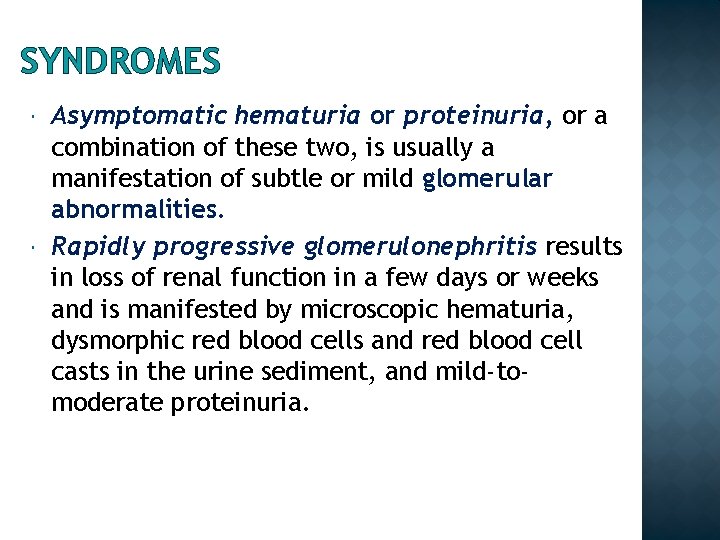
SYNDROMES Asymptomatic hematuria or proteinuria, or a combination of these two, is usually a manifestation of subtle or mild glomerular abnormalities. Rapidly progressive glomerulonephritis results in loss of renal function in a few days or weeks and is manifested by microscopic hematuria, dysmorphic red blood cells and red blood cell casts in the urine sediment, and mild-tomoderate proteinuria.

SYNDROMES Acute renal failure is dominated by oliguria or anuria (no urine flow), with recent onset of azotemia. It can result from glomerular injury (such as crescentic glomerulonephritis), interstitial injury, vascular injury (such as thrombotic microangiopathy), or acute tubular necrosis. Chronic renal failure, characterized by prolonged symptoms and signs of uremia, is the end result of all chronic renal diseases.
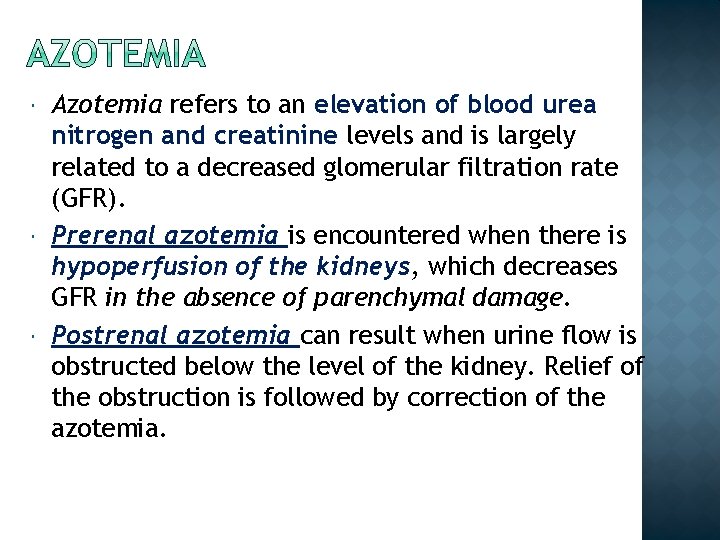
Azotemia refers to an elevation of blood urea nitrogen and creatinine levels and is largely related to a decreased glomerular filtration rate (GFR). Prerenal azotemia is encountered when there is hypoperfusion of the kidneys, which decreases GFR in the absence of parenchymal damage. Postrenal azotemia can result when urine flow is obstructed below the level of the kidney. Relief of the obstruction is followed by correction of the azotemia.

When azotemia progresses to clinical manifestations and systemic biochemical abnormalities, it is termed uremia. Uremia is characterized not only by failure of renal excretory function but also by a host of metabolic and endocrine alterations incident to renal damage. There is, in addition, secondary gastrointestinal (e. g. , uremic gastroenteritis), neuromuscular (e. g. , peripheral neuropathy), and cardiovascular (e. g. , uremic fibrinous pericarditis) involvement.

SYNDROMES Urinary tract infection is characterized by bacteriuria and pyuria (bacteria and leukocytes in the urine). The infection may be symptomatic or asymptomatic, and it may affect the kidney (pyelonephritis) or the bladder (cystitis) only. Nephrolithiasis (renal stones) is manifested by renal colic, hematuria, and recurrent stone formation.


Consists of the following structures: A thin layer of fenestrated endothelial cells A glomerular basement membrane (GBM) The visceral epithelial cells (podocytes), The entire glomerular tuft is supported by mesangial cells lying between the capillaries (secretion a number of BAS).

Glomerulonephritis is a general term for a group of disorders in which: there is immunologically mediated injury to glomeruli the kidneys are involved symmetrically secondary mechanisms of glomerular injury come into play following an initial immune insult the renal lesion may be part of a generalized disease (e. g. systemic lupus erythematosus (SLE).

“primary” (idiopathic) Ø pathology is confined to the kidney Ø any systemic features as direct consequence of glomerular dysfunction “secondary” Ø kidney abnormality as a part of multisystem disorder

Primary Glomerular Diseases Minimal-change disease. Focal and segmental glomerulosclerosis Membranous nephropathy Acute postinfectious GN. Membranoproliferative GN Ig. A nephropathy. Chronic GN Glomerulopathies Secondary to Systemic Diseases Lupus nephritis (systemic lupus erythematosus) Diabetic nephropathy. Amyloidosis GN secondary to lymphoplasmacytic disorders Goodpasture syndrome. Wegener's granulomatosis Microscopic polyangiitis. Henoch-Schönlein purpura Bacterial endocarditis-related GN. Thrombotic microangiopathy GN secondary to extrarenal infection Hereditary Disorders Alport syndrome (nephritis is accompanied by nerve deafness, eye disorders, including lens dislocation, posterior cataracts, and corneal dystrophy). Fabry disease Podocyte/slit-diaphragm protein mutations

Glomerular diseases Nephrotic syndrome Minimal change disease Focal segmental glomerulosclerosis Membranous nephropathy Nephritic syndrome Post-infectious GN Ig. A (immune) nephropathy

Two chief pathogenetic mechanisms are recognized: deposition or in situ formation of immune complexes (most human glomerulonephritides) deposition of antiglomerular basement membrane antibody (fewer than 5% of glomerulonephritides). Both of these pathogenetic mechanisms activate secondary mechanisms that produce glomerular damage.

2 forms of antibody-associated injury have been established: 1 injury resulting from deposition of soluble circulating antigen-antibody complexes in the glomerulus, and 2 injury by antibodies reacting in situ within the glomerulus, either with insoluble fixed (intrinsic) glomerular antigens or with molecules planted within the glomerulus The antigen may be exogenous or endogenous: � exogenous (e. g. bacterial) - for example, a nephritogenic haemolytic Streptococcus � endogenous —patients with SLE may form antibodies to host DNA, leading to a glomerulonephritis.
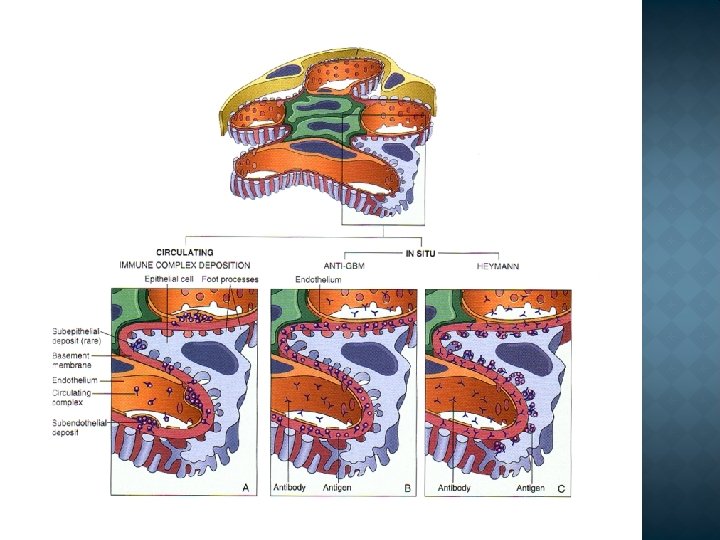

Several events can be triggered by the above immunological insults (type III): complement activation Ø fibrin deposition Ø platelet aggregation Ø glomerular lesions usually consist of leukocytic infiltration (exudation) into glomeruli and variable proliferation of endothelial, mesangial, and parietal epithelial cells Ø activation of kinin system Ø q Resulting in an increase in capillary permeability and glomerular damage.

Glomerular damage, reflected by loss of glomerular barrier function, is manifested by proteinuria and, in some instances, by reductions in GFR. A major pathway of antibody-initiated injury is complement-leukocyte-mediated. Activation of complement →generation of chemotactic agents (C 5 a) →recruitment of neutrophils and monocytes. Neutrophils → proteases →GBM degradation; free radicals →cell damage; and arachidonic acid metabolites→reduction in GFR.

Other mediators of glomerular damage include: monocytes and macrophages, platelets, which aggregate in the glomerulus during immune-mediated injury and release prostaglandins and growth factors; resident glomerular cells (epithelial, mesangial, and endothelial), which can be stimulated to secrete mediators such as IL-1, arachidonic acid metabolites, growth factors, NO, and fibrin-related products, which cause glomerular cell proliferation.
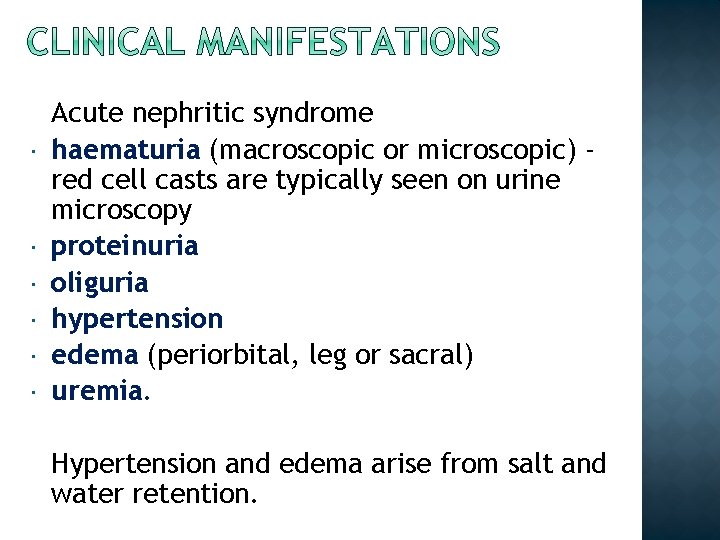
Acute nephritic syndrome haematuria (macroscopic or microscopic) red cell casts are typically seen on urine microscopy proteinuria oliguria hypertension edema (periorbital, leg or sacral) uremia. Hypertension and edema arise from salt and water retention.

reduction the GFR to 30% to 50% of normal, Ø progression to end-stage renal failure Ø glomerulosclerosis. Remaining glomeruli undergo hypertrophy to maintain renal function. Hemodynamic changes, including ↑ in singlenephron GFR, blood flow, and transcapillary pressure (capillary hypertension). Endothelial and epithelial cell injury →hyaline entrapment, ↑ deposition of mesangial matrix, and sclerosis of glomeruli. Ø

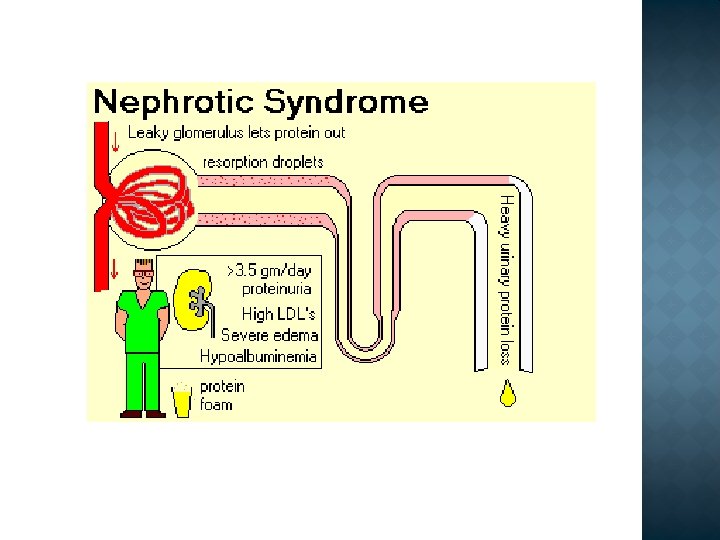
ØProteinuria: Nephrotic range proteinuria – Protein >3. 5 g/day ØHypoalbuminemia (< 3. 0 g/d. L) ØEdema ØHypercholesterolemia – Fasting level >200 mg/d. L – d/t increased production from liver
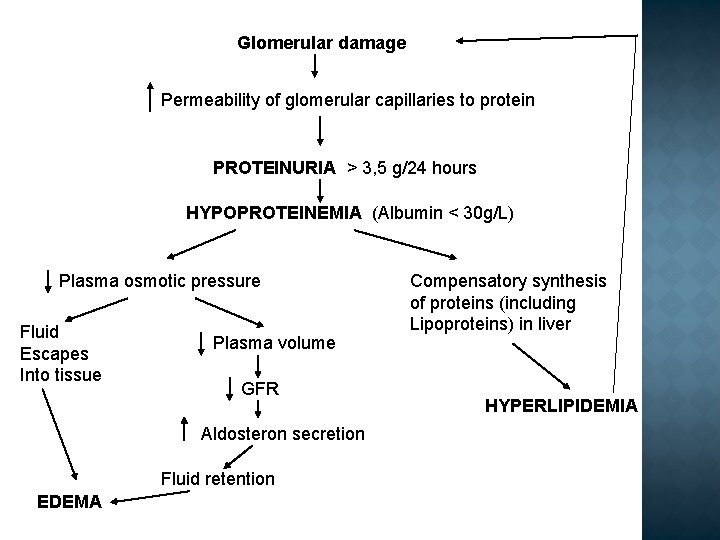
Glomerular damage Permeability of glomerular capillaries to protein PROTEINURIA > 3, 5 g/24 hours HYPOPROTEINEMIA (Albumin < 30 g/L) Plasma osmotic pressure Fluid Escapes Into tissue Plasma volume GFR Aldosteron secretion Fluid retention EDEMA Compensatory synthesis of proteins (including Lipoproteins) in liver HYPERLIPIDEMIA

All glomerulonephritides and minimal-change glomerular lesions Systemic vasculitides, mainly SLE Diabetic glomerulosclerosis Amyloidosis Drugs Allergies
FSGS is a lesion characterized histologically by sclerosis affecting some but not all glomeruli (focal involvement). 1. 2. 3. 4. Associated with the nephrotic syndrome and occur: in association with HIV or heroin abuse as a secondary event in other forms of GN (e. g. , Ig. A nephropathy); as a maladaptation after nephron loss in inherited or congenital forms resulting from mutations affecting cytoskeletal or related proteins

Membranous nephropathy is idiopathic in about 85% of cases. It may be secondary to other disorders: 1. infections (chronic hepatitis B, syphilis, schistosomiasis, malaria); 2. malignant tumors (carcinoma of the lung and colon and melanoma) 3. SLE and other autoimmune conditions; 4. exposure to inorganic salts (gold, mercury); 5. drugs (penicillamine , captopril, nonsteroidal anti-inflammatory).

Autoimmune reaction against unknown renal antigen Immune complexes (MAC causes activation of glomerular mesangial cells and podocytes, inducing them to liberate proteases and oxidants that can damage capillary walls) Thickened GBM Subepithelial deposits
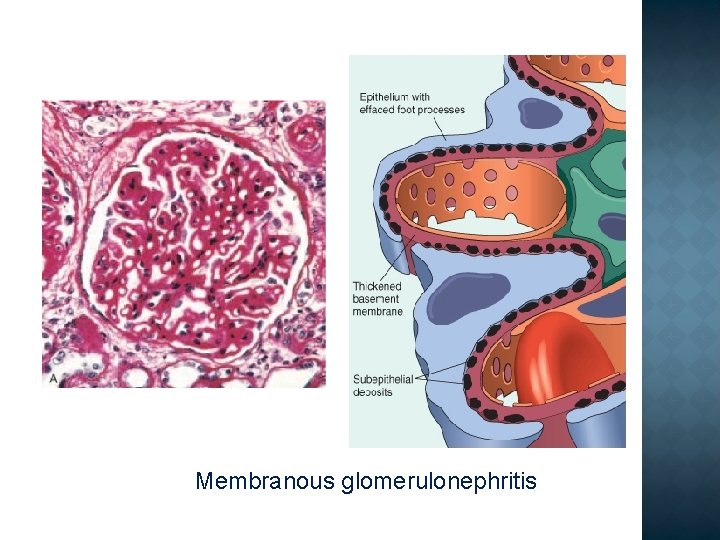
Membranous glomerulonephritis

One of the more frequently occurring glomerular disorders, Caused by glomerular deposition of immune complexes resulting in diffuse proliferation and swelling of resident glomerular cells and frequent infiltration of leukocytes, especially neutrophils. Postinfectious (streptococcal, pneumococcal and staphylococcal inf. , mumps, measles, chickenpox, and hepatitis B and C. )
Immune complexes Hypercellular glomeruli Granular deposits of Ig. G and complement within the capillary walls and some mesangial areas, “Subepithelial humps” Child after throat infection
IG A NEPHROPATHY Common Child with hematuria after upper pespiratory infection The pathogenic hallmark is the deposition of Ig. A in the mesangium. Variable prognosis (slow progression to chronic renal failure occurs in 25% to 50% of cases during a period of 20 years).

“Acute”: days to weeks “Subacute/Rapidly progressive”: over weeks to few months “Chronic”: many months to years

CHRONIC GN Among all individuals who require chronic hemodialysis or renal transplantation, 30% to 50% have the diagnosis of chronic GN. Develops insidiously and is discovered only late in its course, after the onset of renal insufficiency. Proteinuria, hypertension, or azotemia on routine medical examination. Transient episodes of either the nephritic or the nephrotic syndrome. Microscopic hematuria is usually present


Acute tubular necrosis Acute pyelonephritis Chronic Pyelonephritis Drug-induced interstitial nephritis
The most common cause of acute renal failure Reversible lesion Destruction of tubular epithelium Acute suppression of renal function (urine >400 ml/day)

1. Ischemic (state of hypoperfusion: trauma, septicemia, acute pancreatitis, hypotension, shock 2. Nephrotoxic (heavy metals: mercury, Ca. Cl 4, antibiotics: Gentamicin)

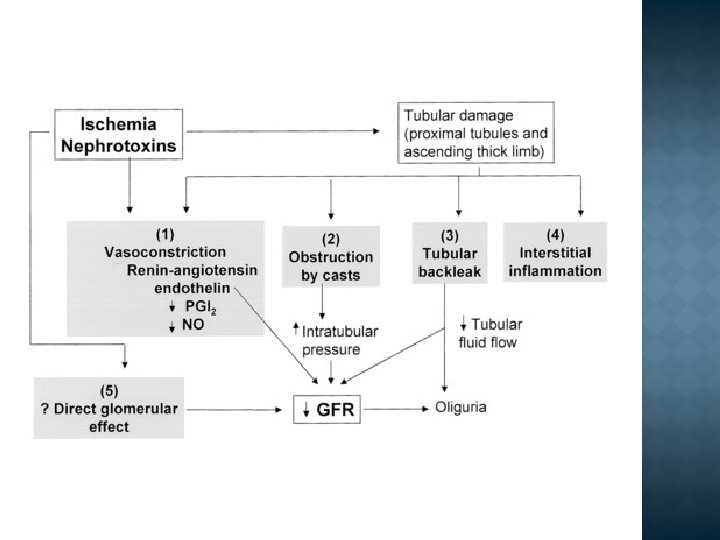
1. Tubular injury Sensitive to ischemia and toxins Injury Functional defect: increase Na+ delivery to distal tubules – vasoconstriction Cytokines – vasoconstriction Tubular debris – block urine outflow – increase the pressure Fluid leak in interstitium – collapse of tubules

2. Blood flow disturbance (persistent, severe), (endothelial cell injury) Vasoconstriction Endothelial injury: release of endothelin, decrease in nitric oxide Others: reninangiotensin, norepinephrine)

Causes Infectious: bacterial, viral, fungal, parasitic Non-infectious: physical (radiation) chemical (toxins, drugs), metabolic, Ischemic, Immune Acute pyelonephritis Chronic pylelonephritis

Commonly bacterial Gram negative: E. coli, Proteus, Klebsiella, Pseudomonas, Enterobacter Risk factors Anomalies Obstruction, bladder dysfunction, reflux Pregnancy Immunosuppression

Routes of infections Hematogenous Ascending infection: Adhesion to mucosa – colonization of urethra – ascending of infection Female>male Symptoms Pain at the costovertebral angle, fever, chills, malaise Urine: pyuria, bacteria Dysuria, frequencey, urgency

Interstitial inflammation and scarring of the renal parenchyma is associated with grossly visible scarring and deformity of the pelvicalyceal system. It is an important cause of chronic renal failure. It can be divided into two forms: 1. Chronic obstructive pyelonephritis Recurrent infections 2. Chronic reflux-associated pyelonephritis Vesico-ureteral reflux

Clinical Late presentation: renal insufficiency, hypertension Contracted kidneys Tubular dysfunction: polyruia/nocturia

Acute drug-induced interstitial nephritis: Antibiotics NSAIDs Diuretics Begin 15 days after exposure Fever, eosinophilia, rash acute renal failure, hematuria, proteinuria

Acute drug-induced interstitial nephritis: Pathogenesis Immune mechanism, hypersensitivity Drug is trapped in the kidney during secretion Results in injury ? Type I, high Ig. E ? Type IV, granuloma

Acute drug-induced interstitial nephritis: Morphology: Edema Inflammatory infiltrate: lymphocytes, macrophages, eosinophils Sometimes: granulomas Analgesic Nephropathy: Pathogenesis Unclear, papillary necrosis, inflammation Oxidative damage Aspirin inhibits Pg synthesis (vasoconstriction)
Benign nephrosclerosis - renal changes in benign hypertension. The kidneys are symmetrically atrophic. Hyaline thickening of the walls of arteries and arterioles, known as hyaline arteriolosclerosis. Malignant Hypertension and Malignant Nephrosclerosis 5% of persons with elevated BP ↑ permeability of the small vessels to fibrinogen and platelet deposition. Intravascular thrombosis. The kidneys become markedly ischemic (RAAS ↑).

Adult polycystic kidney disease is an autosomal dominant disease; 10% of cases of chronic renal failure; kidneys may be very large and contain many cysts. Autosomal recessive (childhood) polycystic kidney disease is caused by mutations in the gene encoding fibrocystin; it is less common and strongly associated with liver abnormalities; kidneys contain numerous small cysts. Medullary cystic disease is being increasingly recognized as a cause of chronic renal failure in children and young adults; complex inheritance, and is associated with mutations in several genes.


Renal failure is a clinical condition, where the glomerular filtration rate is inadequate to clear the blood of nitrogenous substances (urea, uric acid, creatinine, and creatine). The retention of nonprotein nitrogen in the plasma water is called azotemia, and the clinical syndrome is called uremia. The number of filtrating nephrons falls below 1/3 of normal, as determined by measurement of a GFR below 40 ml/min.

Acute renal failure is a syndrome defined by a sudden loss of renal function over several hours to several days The causes of acute renal failure may classified: Prerenal (55 -60%) Intrinsic (renal) (35 -40%) Postrenal ( ~ 5%)
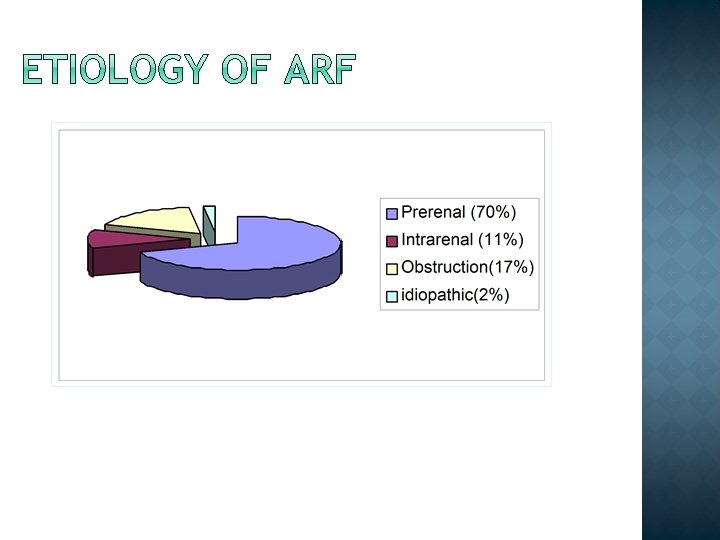
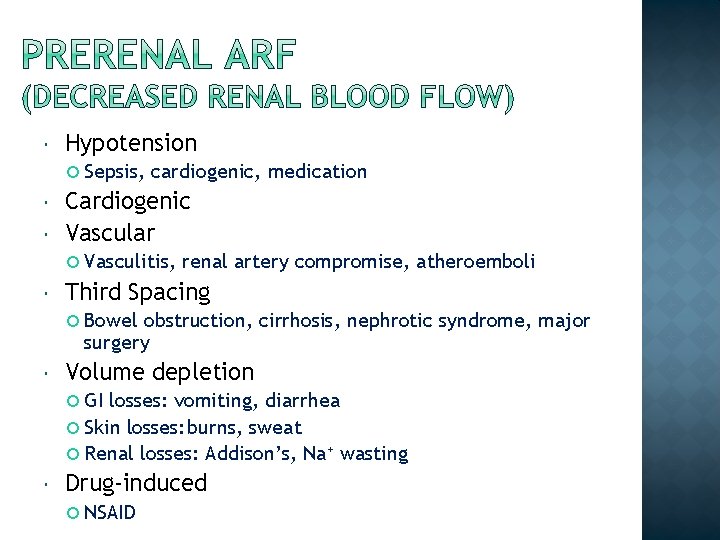
Hypotension Sepsis, cardiogenic, medication Cardiogenic Vascular Vasculitis, renal artery compromise, atheroemboli Third Spacing Bowel obstruction, cirrhosis, nephrotic syndrome, major surgery Volume depletion GI losses: vomiting, diarrhea Skin losses: burns, sweat Renal losses: Addison’s, Na+ wasting Drug-induced NSAID
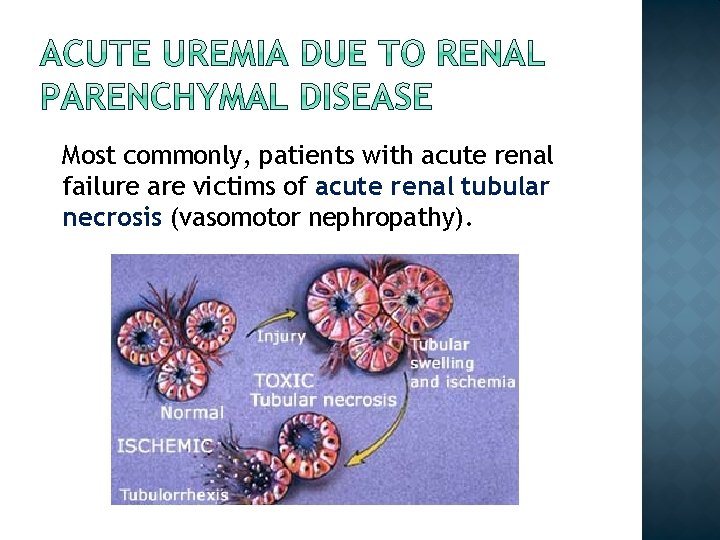
Most commonly, patients with acute renal failure are victims of acute renal tubular necrosis (vasomotor nephropathy).

Hemorrhage Burns Diarrhea and vomiting Pancreatitis Diuretics Myocardial infarction Congestive cardiac failure Endotoxic shock Snake-bite Myoglobinaemia Hepatorenal syndrome
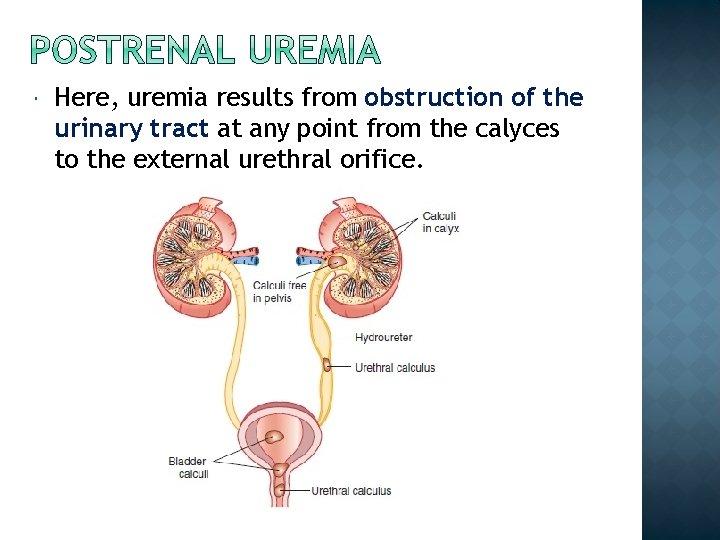
Here, uremia results from obstruction of the urinary tract at any point from the calyces to the external urethral orifice.
Oliguria Polyuria Recovery phase

Azotemia Hypoosmotic hyperhydration Hyperkaliemia Acidosis

PATHOGENESIS OF UREMIA Retention of nitrogenous wastes Increased intracellular Na+ and water Decreased intracellular K+ Increased levels of bioactive substances normally cleared renally (hormones) Decreased levels of hormones and other mediators produced by the kidney Decreased basal body temperature Diminished lipoprotein lipase activity

ACUTE RENAL FAILURE Laboratory changes during acute renal failure: ü hyperkalaemia ( ECG abnormalities) ü decreased bicarbonate ü elevated urea ü elevated creatinine ü elevated uric acid ü Hypocalcaemia ü Hyperphoshataemia

Renal failure Lethargy Anorexia Pericarditis Neuropathy Nausea and vomiting Pruritis Dyspnea

Azotemia Hypohydration isoosmotic Hypokaliemia Acidosis

Eliminate the toxic insult Hemodynamic support Respiratory support Fluid management Electrolyte management Medication dose adjustment Dialysis

Oliguria: <400/24 hr 85% will require dialysis >400/24 hr 30 -40% will require dialysis Mechanical ventilation Acute myocardial infarction Arrhythmia Hypoalbuminemia Multi-system organ failure

Diminish risk of nosocomial infection Prevention of nephrotoxicity avoid/reduce nephrotoxins correct hypokalemia, hypomagnesemia correct/treat other systemic diseases Pharmacology avoid overlapping nephrotoxins follow drug levels closely Attention to fluid status
CHRONIC RENAL FAILURE ØChronic renal failure is a chronic progressive disease that presents by an ongoing loss of kidney function and performance ØThis process has normally a duration of several years leading to end-stage renal failure ØIn this stage dialysis or kidney transplantation become obligatory!

Chronic renal failure represents progressive and irreversible destruction of kidney structures. Chronic renal failure can results from a number of conditions that cause permanent loss of nephrons : Diabetes Hypertension Glomerulonephritis Polycystic kidney disease
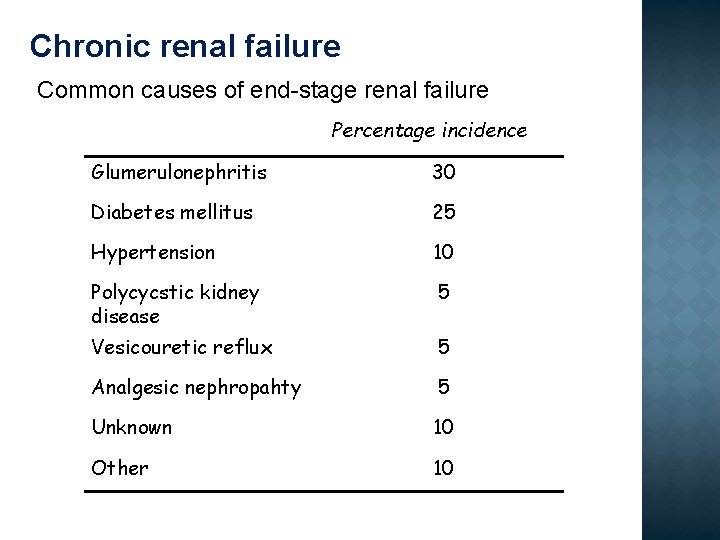
Chronic renal failure Common causes of end-stage renal failure Percentage incidence Glumerulonephritis 30 Diabetes mellitus 25 Hypertension 10 Polycycstic kidney disease 5 Vesicouretic reflux 5 Analgesic nephropahty 5 Unknown 10 Other 10

Chronic renal failure…and diabetes mellitus excretion of glucose with resulting excessive water excretion early influence on kidney function Pathology of diabetic nephropathy early events: ü thickening of the glumerular basement membrane ü expansion of the mesangium ü thickening of efferent and afferent arterioles often superimposed by hypertensive or infective damage progression of thickening of arteries and arterioles scarring and fibrosis


Chronic renal failure Stages Stage of chronic renal failure GFR (m. L/Min) Symptoms of uraemia Serum biochemical derangemen t Mild renal impairment > 75 None Not clearly progressive Mild 50 – 75 None Subtle Early bone disease Moderate 25 – 50 Mild Anaemia starts Severe 10 – 25 Moderate Salt and water retention evident Severe Dialysis or transplantation necessary End-Stage < 5 - 10 Severe Comment

Polyuria Oliguria Uremia

Concentrated function loss Isoosmotic urine - Hypoosmotic urine - Nocturia - Azotemia Urea outcome from organism through the skin, gastrointestinal tract, lungs

Azotemia Uremia Hyperkaliemia Hypocalcaemia (vit D ↓ ) Acidosis Hypertension Anemia Uremic coma

Chronic renal failure Main consequences Mechanism Example Consequence Decreased excretion Uraemic toxins Salt and water Phosphate Acid Potassium Uraemic syndrom Volumne overload, hypertension Hyperparathyroidism Metabolic acidose Hyperkalaemia Decreased biosynthesi s Erythropoietin Activation of vitamin D Anaemia Osteomalacia, Hyperparathyroidism Altered metabolism Dyslipidaemia Sex hormones Atherogenesis Abnormal reproductive function

Factors causing progression Ø sustaining primary disease Ø systemic hypertension Ø Intraglomerular hypertension Ø Proteinuria Ø Nephrocalcinosis Ø Dyslipidaemia Ø Imbalance between renal energy demands and supply

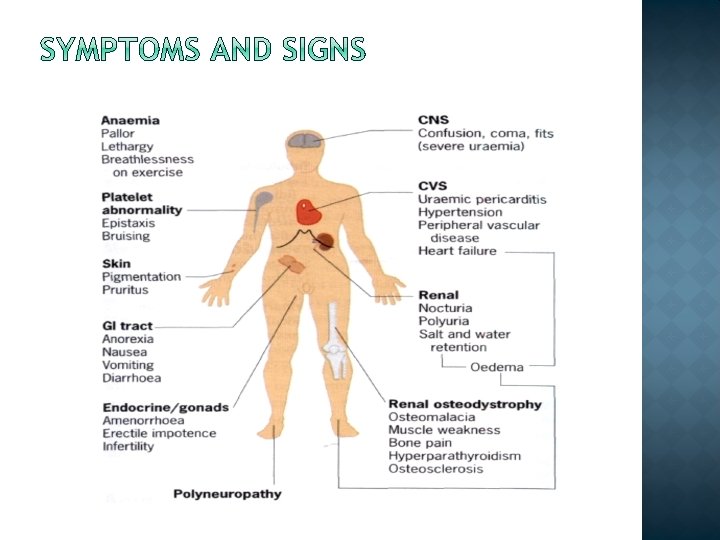
Impaired renal function causes pathologic changes in different secondary organ systems � Cardiovascular system hypertension � Bone disturbed vitamin D, parathyroid hormone metabolism changed Ca 2+ metabolism � Neuromuscular neuropathies by uraemic toxins � Blood anaemia by erythropoietin deficiency � Skins metastatic calcification � Endocrine system disturbed hormone production

Erythropoietin production decreased Hypothyroidism Insulin resistance Growth hormone decreased Gonadal dysfunction Parathyroid hormone and Vitamin D 3 Hyperlipidemia

Renal failure Differentiation between acute and chronic renal failure Acute Chronic History Haemoglobin concentration Normal Low Renal size Normal Reduced Renal osteodystrophy Absent Present Peripheral neuropathy Absent Present Serum Creatinine concentration (days Long (monthyears) Short -week) Acute reversible Chronic increase irreversible

Conservative Therapy Hemodialysis Peritoneal Dialysis Transplant Nothing

GOALS: Detect & treat potentially reversible causes of renal failure Preserve existing renal function Treat manifestations Prevent complications Provide for comfort

Removal of soluble substances and water from the blood by diffusion through a semi-permeable membrane. HISTORY Early animal experiments began 1913 1 st human dialysis 1940’s by Dutch physician Willem Kolff Considered experimental through 1950’s, for acute renal failure only. 1960 Dr. Scribner developed Scribner Shunt 1960’s Machines expensive, scarce, no funding. “Death Panels” panels within community decided who got to dialyze.

During dialysis Fluid and electrolyte related hypotension Cardiovascular arrythmias Associated with exsanguination Neurologic seizures Musculoskeletal cramping Other fever & sepsis the extracorporeal circuit 119
- Slides: 119