RENAL FUNCTION TEST PRESENTED BY H GROUP Mohini

RENAL FUNCTION TEST PRESENTED BY H GROUP Mohini, monika R, Ankita, Kinjal, Kinal, monika

INTRODUCTION: � Kidneys are the organ that filter waste products from the blood. � The kidneys serve three essential function: 1. 2. 3. They function as filter, removing metabolic product and toxins from the blood and excreting them through the urine. They regulate the body’s fluid status, electrolyte balance and acid-base balance. The kidney produce or activate hormones that are involved erythrogenesis, Ca²˖ metabolism and the regulation of blood pressure and blood flow.

� Renal function may be assessed by measuring blood urea and serum creatinine. Renal function decreases with age , which must be taken into account when interpreting test values. � These tests primarly evaluate glomerular function by assessing the glomerular filtration � In many renal diseases, urea and creatinine accumulate in the blood because they are not excreted properly � These tests also aid in determining drug dosage for drugs excreted through the kidneys

What is Renal Function Test? � Renal function tests are use to detect the presence of renal diseases and assess their progress.
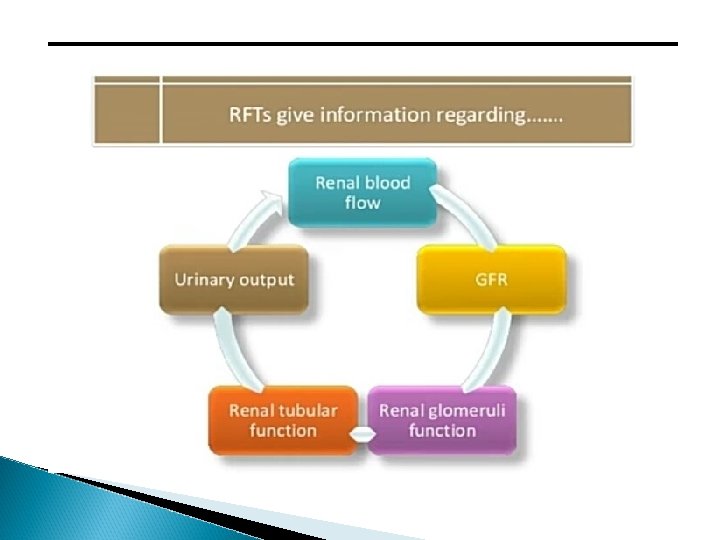

TESTS INVOLVED IN RFT: 1. 2. 3. 4. 5. 6. 7. 8. Urea Ammonia Para Thyroid Hormone Calcium Uric acid Potassium Creatinine clearance Glomerular filtration rate

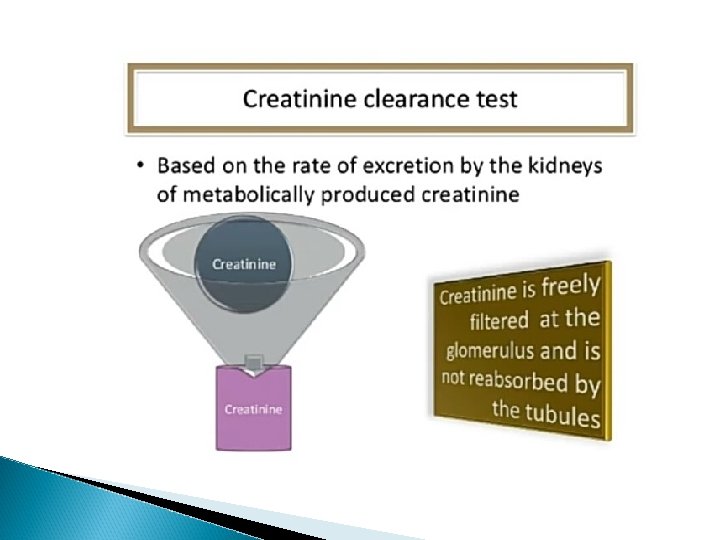
CREATININE: � In the muscles creatine is converted to creatine phosphate which becomes the source of a high energy phosphate bond for the immediate reformation of ATP. � Creatinine is the byproduct of muscle energy metabolism and is produce at a constant rate according to the muscle mass of the individual. It is the substance that is easily excreted by the kidney. � By this method we can also estimate serum creatinine and urine creatinine.

� Jaffe method Principle: In this colorimetric method creatinine reacts with picrate ion formed in alkaline medium to develop a red-orange colour. The colour produced from the sample is then compared in a colorimeter at wavelength of 520 nm with that produced by known amount of creatinine under the same condition. � Creainine + picric acid →creatinine picrate (orange)

This divides into two types 1. Manual method and 2. Automated kinetic method The automated kinetic assay of creatinine is a method with the Jafee reaction but by using special type of spectrometer or autoanalyser.

Method: � To get clear supernatant, take 1 ml of distilled water, serum, sodium tungstat and sulfuric acid. � Mix it well and centrifuge it for 5 min. � Label three tubes as blank, standard and test respectively. Blank Standard Test Sample - - 2 ml d/w 2 ml 2 ml Na. OH 1 ml 1 ml Picric acid 1 ml 1 ml

� Mix it well and incubate at room temperature for 10 min. � Take the OD against the blank at 520 nm. Calculation formula: Creatinine(mg/d. L)= abs. of test/ abs. of std. *std. conc.

Creatinine Clearance �A measure of the amount of creatinine eliminated from the blood by the kidney. � The value is given in unit of millions per minute, representing the volume of blood cleared by the kidney per minute. � Calculation: C cr=U x V/P Where, U = Urine creatinine concentration in mg/dl, P = Serume creatinine in mg/dl and V = Volume of urine in ml/mt.

Referance Range � Male: 0. 7 -1. 3 mg/d. L � Female: 0. 4 -1. 1 mg/d. L

Interpretation High creatinine level causes: Ø Acute and Chronic kidney disease Ø Ureter obstruction Ø Dehydration Ø Glomerulonephritis

PTH: PARATHYROID HORMONE PTH is a hormone secreted by the parathyroid gland There are four parathyroid glands located behind the thyroid
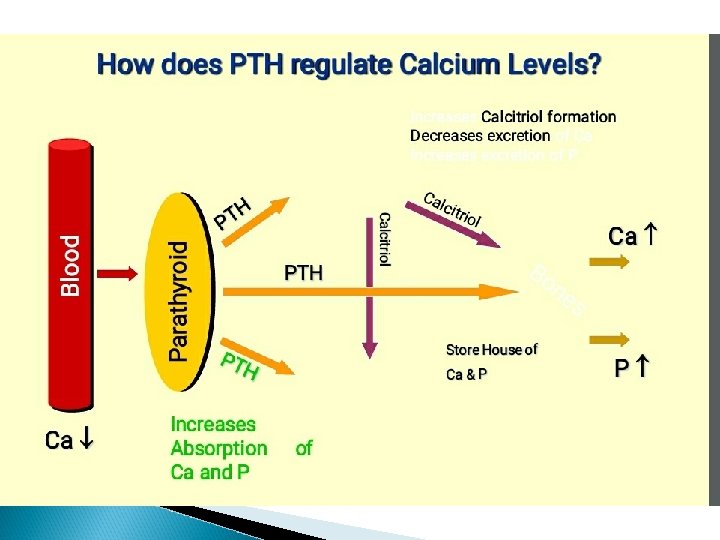

Role of PTH � To regulate calcium levels (in blood) � The parathyroid glands major function – regulate the calcium level in the body within a very narrow range (8. 5 – 10. 2 mg/dl)so that the nervous and muscular system can function properly. � Activation of vitamin D is very essential for calcium absorption from GI tract. � Vitamin D has to be converted into 1, 25 dihydroxycholeciferol in the liver and kidney in the presence of PTH. � Vitamin D � PTH 1, 25 - dihydroxycholeciferol is also increased the formation of 1, 25 dihydroxycholeciferol from 25 hydroxycholecalciferol.

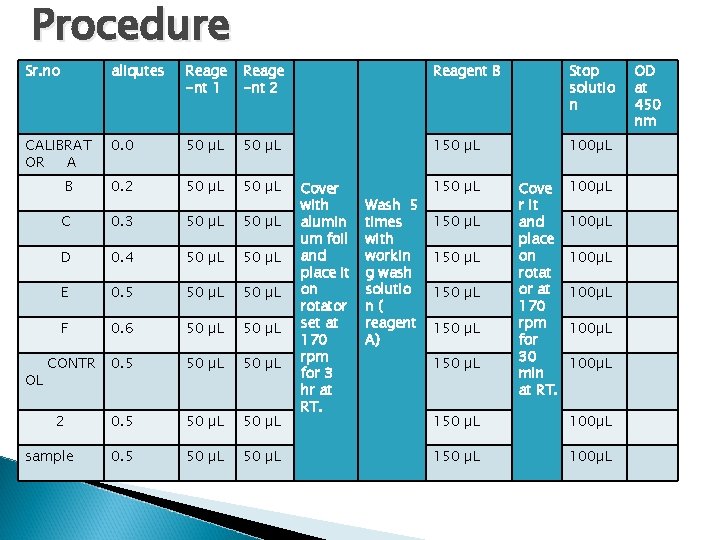
� PRINCIPLE OF THE TEST The DRG Intact PTH Immunoassay is a two-site ELISA [Enzyme-Linked Immuno. Sorbent Assay] for the measurement of the biologically intact 84 amino acid chain of PTH. One antibody is prepared to bind only the N- terminal PTH 1 -34. and this antibody is labeled with horseradish peroxides(HRP) for detection. -incubation -washed unbound components And the another end c -terminal bind with biotinlaylded anti-PTH. Make a sandwich complex necessary for detection. -add substrate (TMB) -stopping solution( 1 N sulfuric acid) Yellow color which is praposnal to PTH concentration.
Procedure Sr. no aliqutes Reage -nt 1 Reage -nt 2 Reagent B Stop solutio n CALIBRAT OR A 0. 0 50 µL 150 µL 100µL B 0. 2 50 µL C 0. 3 50 µL D 0. 4 50 µL E 0. 5 50 µL F 0. 6 50 µL 0. 5 50 µL OL CONTR 2 sample Cover with alumin um foil and place it on rotator set at 170 rpm for 3 hr at RT. Wash 5 times with workin g wash solutio n( reagent A) 150 µL 150 µL Cove r it and place on rotat or at 170 rpm for 30 min at RT. 100µL 100µL 150 µL 100µL OD at 450 nm

� Where, � Reagent 1= biotinylated PTH antibody � Reagent 2= peroxidase labeled PTH ab � Reagent A= saline with surfactant � Stop solution = 1 N sulfuric acid � Referance range : = 10 -65 pg/ml

URIC ACID Introduction

Sometime the body produces too much uric acid or doesn’t filter out enough of it and that time uric acid level increase in blood. Main two condition are observed, 1. Hyperuricemia 2. Hypouricemia


URIC ACID BLOOD TEST � Introduction A uric acid blood test also known as a serum uric acid measurment, determine how much uric acid is present in your blood. The test can help determine how well your body produce and removes uric acid.

� Intended use This reagent is for in vitro determination of uric acid in serum/plasma. � Clinical significance Uric acid is a metabolism of purines, nucleic acids and nucleo-proteins. Consequently, abnormal levels may be indicative of a disorder in the metabolism or in some genetic diseases.

� Causes of increase and decrease of uric acid level - High level of uric acidin your blood can also indicate of a variety of conditions, inluding: - diabetes - gout (acute arthritis) - chemotherapy & radiation - leukemia(bone marrow disorders) - hypoparathyroidism - kidney disorders(stones) - multiple myeloma - metastasized cancer
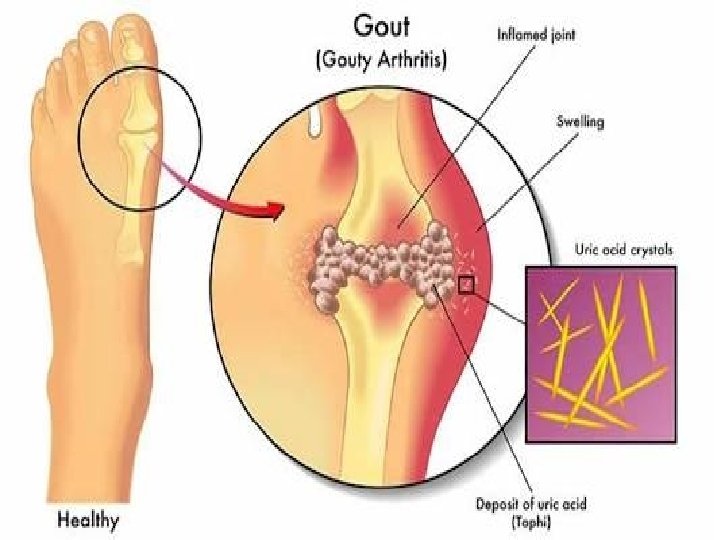

- low levels of uric acid in the blood may inluding: - wilson’s disease - fanconi syndrome(cystinosis) - alcoholism - liver or kidney disease - a diet low in purines

� Methodology 1. chemical method phototungstic acid method 2. enzymatic method The reagent is based on Trinder’s reaction, enzymatic and colorimetric method.


� Raference range serum/plasma for women – 2. 5 -6. 8 mg/dl for men – 3. 6 - 7. 7 mg/dl

� Limitations of uric acid testing are as follows: - Methodological interference and in cases of vitamin C, levodopa, and alphamethyldopa. - Early purine rich diet - several exercise increases uric acid - Rapid degradation of uric acid, which occurs at room temp. in the plasma of patients with tumor lysis syndrome.

UREA: � Urea is the chief nitrogenous waste of body. � Urea is the end product of protein metabolism. � After filtered by glomeruli. It is partilly reabsorbed by the renal tubules. Methodology: 1. Kinetic method 2. Enzymatic method

Principle: � Urea in the sample is hydrolized enzymatically into ammonia(NH 4+) and carbon dioxide (co 2). � Ammonia ions formed reacts with αketoglutarate in a reaction catalysed by glutmate dehydrogenase(GLDH) with simultaneous oxidation of NADH to NAD+. � Urea + 2 H₂O Urease 2 NH₄⁺+ CO₃²⁻ GLDH � NH₄ ⁺ +2 -Oxoglitarate +NADH L-glutamate +NAD ⁺+H₂O

� The decrease in concentration of NADH, is proportional to urea concentration in the sample. ASSAY PROCEDURE: PIPETTE IN TUBES MARKED WORKING REAGENT STANDARD SAMPLE STANDARD TEST 1000μl 20μl - - 20μl Mix well and aspirate standard followed by samples.

Calculation: Urea (mg/dl)=(∆ Abs of test/∆Abs of std)*conc. Of standard(mg/dl) REFERENCE VALUE: serum/plasma: 13 -45 mg/dl

Urea clearance test: � Urea clearance test is less than the GFR and it is influnced by the protein content of the diet. � Approximately 40% of filtered urea is normaly reabsorbed by tibules � The sensitivity of urea clearance is much less than the creatinine clearance because plasma concentration of urea is affected by number of factors. � Like, Dietary protien fluid intake infaction surgery, etc. � Nornaml value of urea clearance: 75% ml/min. � Urea clearance is defined as the volume(ml) of plasma that would be completely cleared of urea per minue.

� It is calculated by the formula: Cm= U*V/P Cm= Maximum Urea clearance. U = Urea concentration in urine (mg/dl). V = Urine excreted per minute in ml. P = Urea concentration in plasma. � If the output of urine is more than 2 ml per minute. � This is referred to as maximum urea clearance.

Standard Urea Clerance: � the urea clearance drastically changes when the volume of urine is less than 2 ml/min. � This is known as standard urea clearance(C) and the normal value is around 54 ml/min. Diagnostic importance: � A Urea clearance value below 75% of the normal is serious. Since it is an indicator of renal damage. � Blood urea level is found to increas only when the clearance falls below 50% normal. � Normal level of blood urea: 20 -40 mg/dl.

Causes for increasedblood urea: Pre-renal condition: -Dehydration: Severe vomiting, intestinal obstruction, diarrhea, diabetic coma, severe burns, fever and severe infections. Renal diseases: 1. Acute glomerulonephritis 2. Nephrosis 3. Malignant hypertension 4. Chronic pyelonephritis

Decreased blood urea: � Urea concentration in serum may be low in late pregnancy, in starvation, in diet grossly deficient in protein and in hepatic failure. � Azotemia: = � Increase in the blood level of NPN(creatinine, urea, uric acid) is referred to as azotemia and is the hallmark of kidney failure.

Calcium: � Calcium plays an important role in: � nerve impulse transmission, � muscle contraction, � pancreatic insulin release, � as a core factor for some enzyme reactions and blood coagulation, � and most important bone and tooth structural integrity. Normal total calcium values 8. 8‐ 10. 3 mg/dl.

� Hypocalcemia usually implies a deficiency in either the production or response to parathyroid hormone(PTH) or vitamin D � Hypercalcemiais an increased calcium concentration and it is usually associated with malignancy or metastatic diseases.

Reference ranges in males � Younger � Age � Age than 12 months : not established 1 -14 yr : 9. 6 -10. 6 mg/dl 15 -16 yr : 9. 5 -10. 5 mg/dl 17 -18 yr : 9. 5 -10. 4 mg/dl 19 -21 yr : 9. 3 -10. 3 mg/dl 22 yr and older : 8. 9 -10. 1 mg/dl

OCPC Method � Principle: calcium in an alkaline medium combines with o – Cresolphthalein complexone to form a purple coloured complex. Intensity of the colour formed is directly proportional to the amount of calcium present in the sample. � Calcium + OCPC Purple coloured complex

Procedure � Wavelength/filter: 570 nm(Hg 578 nm)/yellow � Temperature : R. T. � Light path : 1 cm � Pipette into clean dry test tubes labelled as blank(B), standard(S), and Test(T)

� Mix Addition sequence B (ml) S (ml) T (ml) Buffer Reagent(L 1) 0. 5 Colour. Reagent(L 2) 0. 5 Distilled water 0. 02 Calcium Standard (S) Sample - 0. 5 - - 0. 02 well and incubate at R. T. (25 ℃) for 5 min. Measure the absorbance of the standard(Abs. S), and test sample(Abs. T) against the blank, within 60 min.

Calculations � Calcium in mg/dl = Abs. T ______ Abs. S X 10

Arsenazo iii method : � Principle : Calcium combines specifically with Arsenazo iii at a neutral p. H to forma blue purple coloured complex. Intensity of the colour formed is directly proportional to the amount of calcium present in the sample. � Calcium + Arsenazo iii Blue purple coloured complex

Procedure � Wavelength/filter: 650 nm(Hg 623 nm)/Red � Temperature : R. T. � Light path : 1 cm � Pipette into clean dry test tubes labelled as blank(B), standard(S), and Test(T)

� Mix Addition sequence B (ml) Colour. Reagent(L 1) 1. 0 Distilled water 0. 01 Calcium Standard (S) Sample - S (ml) T (ml) 1. 0 - - 0. 01 well and incubate at R. T. (25 ℃) for 5 min. Measure the absorbance of the standard (Abs. S), and test sample (Abs. T) against the blank, within 60 min.

Calculations : � Calcium in mg/dl = Abs. T ______ Abs. S X 10

Potassium � Potassium is the most abundant intercellular cation approximately 3500 m. Eq of potassium is contained in the body of a 70 kg adult. Only 10% of the body potassium is extracellular. Normal values are 3. 5‐ 5. 0 m. Eq/L or mmol/L � The role or function of potassium is in the maintenace of proper electrical conduction in cardiac and skeletal muscles. � Potassium measurements are used to monitor electrolyte balance in a diagnosis and treatment of disease condition characterized by low or high blood potassium levels.

Potassium is regulated by: � Kidneys � Aldosterone � Arterial � Insulin p. H � Potassium intake � Sodium delivery to distal tubules

Principle � An Ion-Selective Electrode makes use of the unique properties of certain membrane materials to develop an electrical potential for the measurement of ions in solution. � The potassium electrodes are based on neutral carriers.

Reference range § § § Serum : K+ : 3. 3 – 5. 1 mmol/L Plasma : K+ : 3. 3 – 4. 5 mmol/L Urine : K+ : 25 – 125 mmol/L (diet dependent)

Interpritation: � Hypokalemia can occur. The kidneys are responsible for approximately 90% of daily potassium loss. Other losses occur mainly through GI system. � A low potassium level has many causes but usually results from vomiting, diarrhea, adhrenal gland disorders. � A low potassium level can make muscles feel weak, cramp, twitch, or even become paralyzed, and abnormal heart rhthms may develop. � Hyperkalemia most common results from decreased renal elimination, excessive intake or from cellular break down.

AMMONIA PRINCIPLE OF THE METHOD: � Ammonia combines with α-ketoglutarate and NADPH in the presence of glutamate dehydrogenase (GLDH) to yield glutamate and NADP+. The corresponding decrease in absorbance at 340 nm is proportional to the plasma ammonia concentration. � α-ketoglutarate + NH₃+NADPH glutamate+ NADP⁺ GLDH

Procedure: � 1. Assay conditions: � Wavelength: 340 nm � *Cuvette: 1 cm light path � Constant temperature: 25/30/37ºC � * Please try not to use flow cell. Exchangeable cuvettes are suggested to avoid cargover in manual photometers. � 2. Adjust the instrument to zero with distilled water. � 3. Pipette into a cuvette:
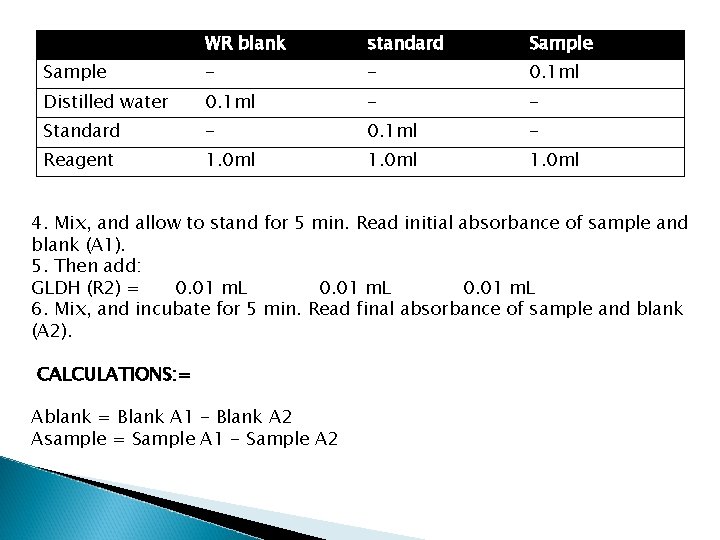
WR blank standard Sample - - 0. 1 ml Distilled water 0. 1 ml - - Standard - 0. 1 ml - Reagent 1. 0 ml 4. Mix, and allow to stand for 5 min. Read initial absorbance of sample and blank (A 1). 5. Then add: GLDH (R 2) = 0. 01 m. L 6. Mix, and incubate for 5 min. Read final absorbance of sample and blank (A 2). CALCULATIONS: = Ablank = Blank A 1 - Blank A 2 Asample = Sample A 1 - Sample A 2

� Conc. of � � � Ammonia Asample - Ablank = --------Astandard - Ablank REFERENCE VALUES(2) Plasma ammonia: 10 - 47 μmol/L 0. 17 - 80 μg/d. L 0. 017 - 0, 080 mg/d. L x Standard conc

GLOMERULAR FILTRATION RATE q. Glomerular filtration rate(GFR) is the volume of fluid filtered from the renal(kidney) glomerular capillaries in to the Bowman’s capsule per unit time. q. Central GFR. of the physiological maintenance of

PRINCIPAL � GFR is equal to the clearance rate when any solution is freely filtered & is neither reabsorbed non secreted by the kidneys. � GFR is typically recorded in units of volume per tine � EX: milliliters per minutes(ml/min), compare to filtration fraction

� The rate at which plasma is filtered by the kindney glomeruli. � An important measurement in the evaluation of kidney fuction � GFR = 125 ml plasma/min or, 180 L/day � Plasma volume (70 -kg young adult man)= about 3 L, the Kidneys filter the plasma some 60 time in a day.
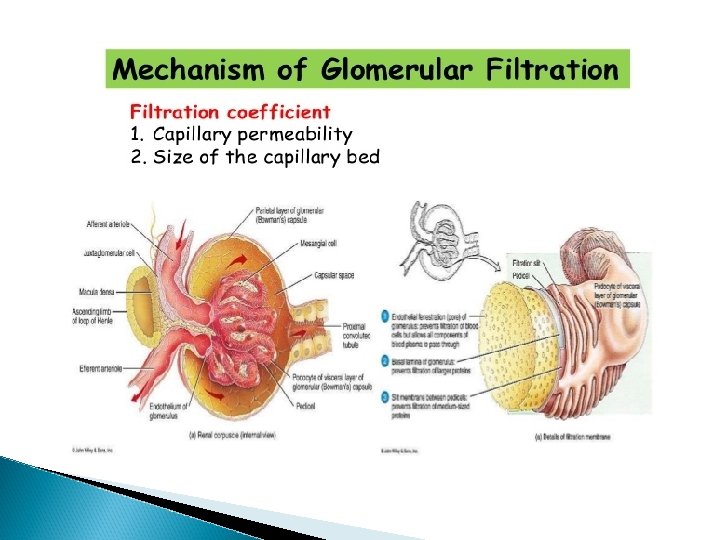
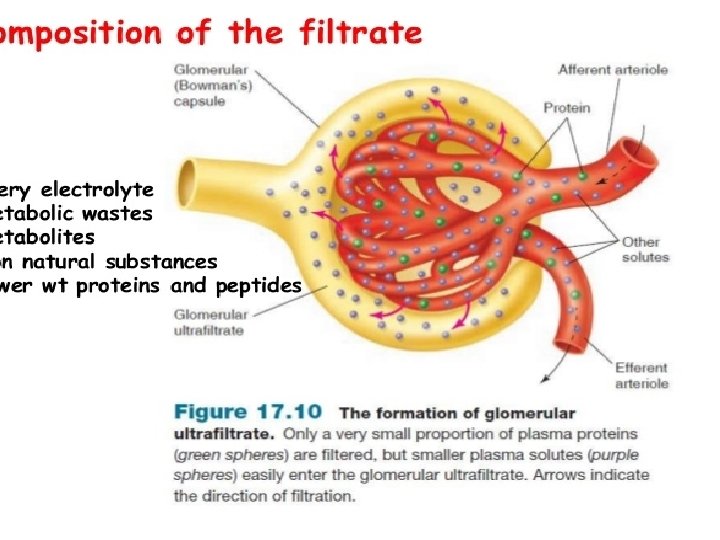

Factors affecting GFR 1. 2. 3. 4. 5. 6. 7. Change in renal blood flow Glomerular capillary hydrostatic pressure Change in capsular hydrostatic pressure Oncotic pressure Glomerular capillary permeability Effective filtration surface area Size, shape & electrical change of the macromolecules

Measurement of GFR � Modern imaging techniques � Measuring substances renal clearance of various

Pathological condition that affect GFR 1. 2. 3. Nephrotic syndrome Nephritic syndrome Single kidney

CLEARANCE � Measurement or calculation of the glomerular filtration reat(GFR) � Considered the most sensitive chemical test for assessing function � Collect a 24 hour or timed urine speciment & a blood specimen from the patient � Measure the creatinine in the serum $ urine soecimen � Calculate the clearnce

e. GFR� e. GFR=30849. 2 X (serum creatinine)1. 154(age)-0. 203 � (If female X (0. 742))
- Slides: 72