Relation between Dmax based sizing and lesionlevel outcomes

Relation between Dmax based sizing and lesion-level outcomes in Absorb BVS and Xience EES treated patients. - AIDA trial sub-study Ruben Y. G. Tijssen MD Laura S. Kerkmeijer MD Joanna J. Wykrzykowska MD, Ph. D Academic Medical Center Amsterdam

Potential conflicts of interest Speaker's name : Ruben Y. G. Tijssen, AMSTERDAM ☑ I have no conflicts or interest to report

The AIDA trial - Background • The ABSORB bioresorbable vascular scaffold - approved by the FDA in 2016 and was CE marked in 2010. • Before the device was commercialized, no adequately powered, randomized (allcomers) studies, addressing safety and efficacy, had been performed. • After safety review at the 11 th of November 2016, the DSMB recommended early reporting of the data of the AIDA trial because of safety concerns of scaffold thrombosis. • The preliminary outcomes per 20 -Dec-2016 were presented at Euro. PCR 2017, and were published in the NEJM in June 2017 (NEJM 2017; 376: 2319 -2328) • The full 2 -year results were presented at Euro. PCR 2018 en were simultaneously published in Euro. Intervention.
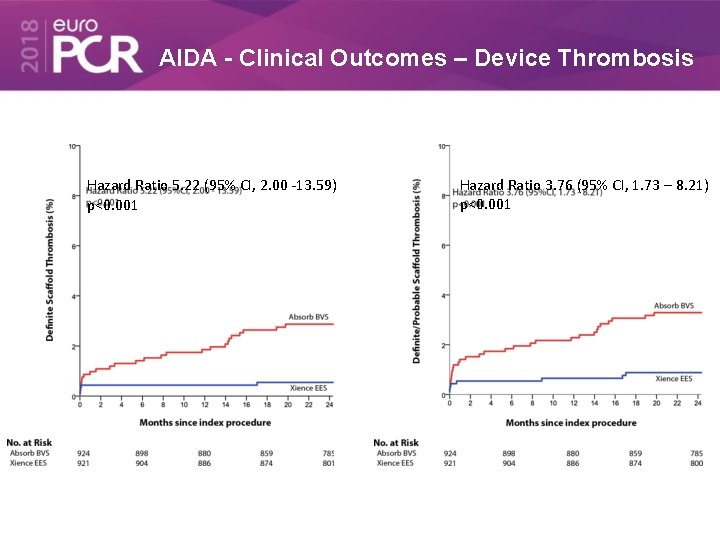
AIDA - Clinical Outcomes – Device Thrombosis Hazard Ratio 5. 22 (95% CI, 2. 00 -13. 59) p<0. 001 Hazard Ratio 3. 76 (95% CI, 1. 73 – 8. 21) p<0. 001

No significant Predictors of device thrombosis

Dmax QCA Background • In spite of the increased risk of scaffold thrombosis, AIDA met its non-inferiority margin for its primary endpoint of TLF. • To date, however, the exact cause of the increased incidence of scaffold thrombosis remains unknown. • Analysis from Absorb cohort B, II, EXTEND showed that an oversized Absorb BVS in a small vessel was associated with higher rates of MACE at 1 year, while undersized Absorb BVS implantation was associated with a higher risk of MACE between 1 and 3 years. 1, 2 • The aim of this study was to investigate the lesion oriented outcomes related to appropriate lumen to device sizing based on quantitative coronary angiography in both the Absorb BVS and the Xience EES arm of the AIDA trial. 1). Katagiri et al. Relation between Bioresorbable Scaffold Sizing Using QCA-Dmax and Long-term Clinical Outcomes in 1, 232 Patients from 3 Study Cohorts (ABSORB Cohort B, ABSORB EXTEND, and ABSORB II). , Euro. Intervention 2018. 2. ) Ishibashi et al. Relation Between Bioresorbable Scaffold Sizing Using QCA-Dmax and Clinical Outcomes at 1 Year in 1, 232 Patients From 3 Study Cohorts (ABSORB Cohort B, ABSORB EXTEND, and ABSORB II). JACC CI, 2015

Endpoint - Statistical Analysis • This presentation summarizes information of lesion-oriented outcomes of scaffold thrombosis and target lesion revascularization subdived by their non-oversize or oversize status. • Lesions were treated with oversized devices if: – The proximal and distal in-segment diameter (Dmax) were smaller than the nominal device size • Multiple device / lesion: – The proximal Dmax smaller than the nominal device size of the proximal device – The distal Dmax smaller than the nominal device size of the distal device • All analyses were conducted using the per-protocol population.

QCA analysis • The current analysis includes all study lesions that received at least one study device for which baseline angiogram suited for QCA was available • Analyses were performed by 12 QCA readers – blinded for clinical events. • Pre- and post-procedural measurements were performed in either: • (1) multiple matched views; (2) single matched view (3) no matched views. • All analysis were performed with Cardiovascular Angiography Analysis System (CAAS, Pie Medical)

Academic QCA corelab • QCA corelab supervision: Prof. dr. P. W. Serruys (NHLI, Imperial College London) Dr. Y. Onuma (Erasmus MC, Cardialysis BV, Rotterdam) • Academic QCA corelab: Ruben Tijssen 1, Laura Kerkmeijer 1, Yuki Katagiri 1, MD; Kuniaki Takahashi 1, MD; Norihiro Kogame 1, MD; Ply Chichareon 1, MD; Rodrigo Modolo 1, MD; Taku Asano 1, MD; Martina Nassif 1, MD; Deborah N. Kalkman 1, MD; Yohei Sotomi 1, MD; Carlos Collet 1, MD 1 AMC Heart Center, Amsterdam
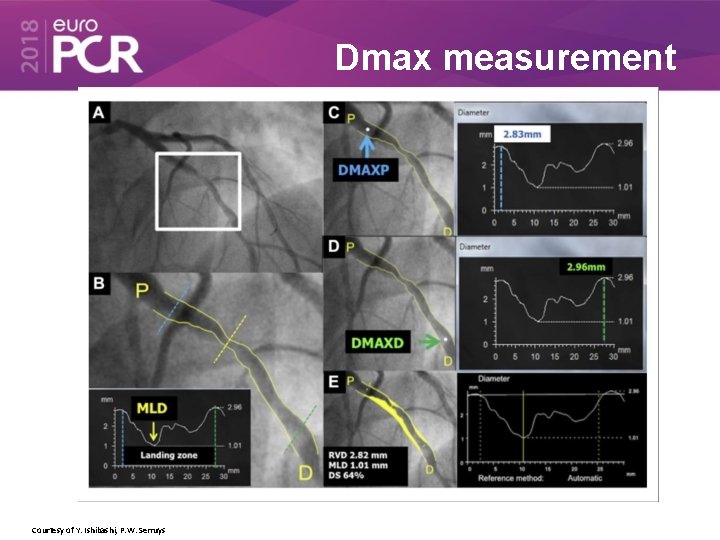
Dmax measurement Courtesy of Y. Ishibashi, P. W. Serruys
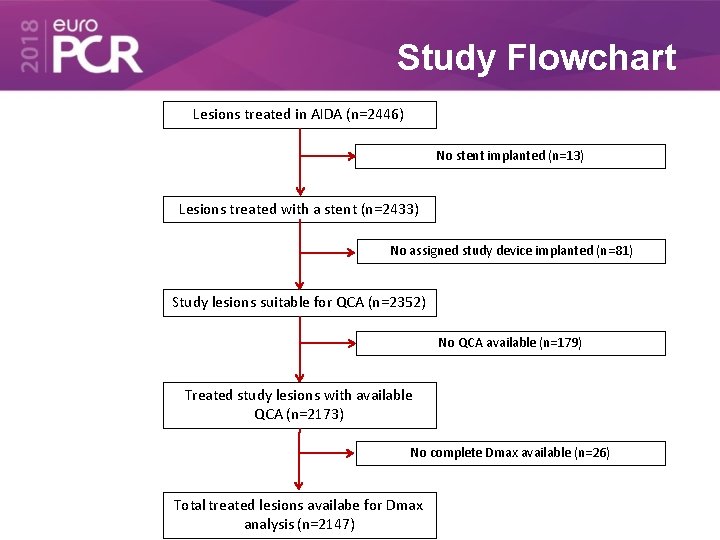
Study Flowchart Lesions treated in AIDA (n=2446) No stent implanted (n=13) Lesions treated with a stent (n=2433) No assigned study device implanted (n=81) Study lesions suitable for QCA (n=2352) No QCA available (n=179) Treated study lesions with available QCA (n=2173) No complete Dmax available (n=26) Total treated lesions availabe for Dmax analysis (n=2147)
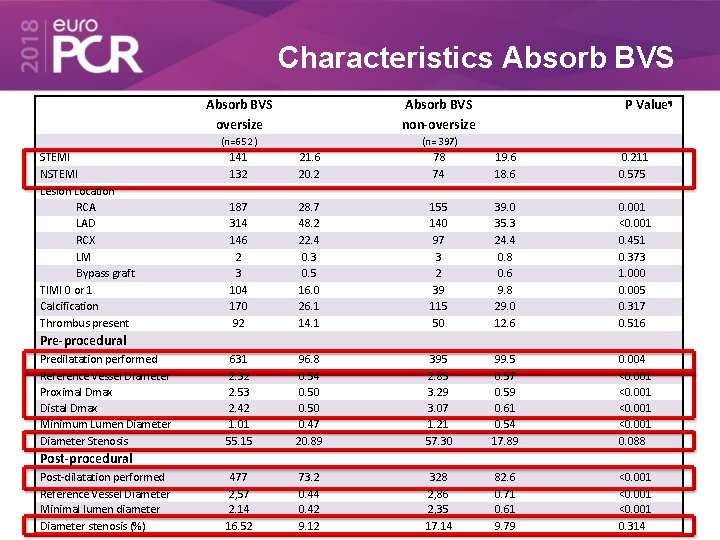
Characteristics Absorb BVS STEMI NSTEMI Lesion Location RCA LAD RCX LM Bypass graft TIMI 0 or 1 Calcification Thrombus present Absorb BVS oversize Absorb BVS non-oversize (n=652 ) (n= 397) P Value¶ 141 132 21. 6 20. 2 78 74 19. 6 18. 6 0. 211 0. 575 187 314 146 2 3 104 170 92 28. 7 48. 2 22. 4 0. 3 0. 5 16. 0 26. 1 14. 1 155 140 97 3 2 39 115 50 39. 0 35. 3 24. 4 0. 8 0. 6 9. 8 29. 0 12. 6 0. 001 <0. 001 0. 451 0. 373 1. 000 0. 005 0. 317 0. 516 631 2. 32 2. 53 2. 42 1. 01 55. 15 96. 8 0. 54 0. 50 0. 47 20. 89 395 2. 85 3. 29 3. 07 1. 21 57. 30 99. 5 0. 57 0. 59 0. 61 0. 54 17. 89 0. 004 <0. 001 0. 088 477 2, 57 2. 14 16. 52 73. 2 0. 44 0. 42 9. 12 328 2, 86 2, 35 17. 14 82. 6 0. 71 0. 61 9. 79 <0. 001 0. 314 Pre-procedural Predilatation performed Reference Vessel Diameter Proximal Dmax Distal Dmax Minimum Lumen Diameter Stenosis Post-procedural Post-dilatation performed Reference Vessel Diameter Minimal lumen diameter Diameter stenosis (%)

Characteristics Xience EES STEMI NSTEMI Lesion Location RCA LAD RCX LM Bypass graft TIMI flow (pre-PCI) Calcification Thrombus present Xience EES oversize Xience EES non-oversize P Value¶ (n=648 ) 166 135 25. 6 20. 8 (n= 450) 85 102 18. 9 22. 7 0. 010 0. 502 178 304 161 2 3 109 170 102 27. 5 46. 9 24. 8 0. 3 0. 5 16. 8 26. 2 15. 7 143 172 130 4 1 36 128 42 31. 8 38. 2 28. 9 0. 2 8. 0 28. 4 9. 3 0. 138 0. 004 0. 114 0. 234 0. 648 <0. 001 0. 448 0. 002 607 2. 31 2. 58 2. 50 0. 93 58. 35 93. 7 0. 50 0. 49 0. 36 16. 35 406 2. 79 3. 26 3. 04 1. 07 60. 8 90. 2 0. 60 0. 65 0. 44 15. 5 0. 039 <0. 001 0. 014 295 2. 62 2. 26 13. 39 45. 5 0. 47 0. 44 9. 26 243 2, 87 2, 41 15, 79 54. 0 0. 54 0. 49 9. 29 0. 006 <0. 001 Pre-procedural Predilatation performed Reference Vessel Diameter Proximal Dmax Distal Dmax Minimum Lumen Diameter Stenosis Post-procedural Post-dilatation performed Reference Vessel Diameter Minimal lumen diameter Diameter stenosis (%)
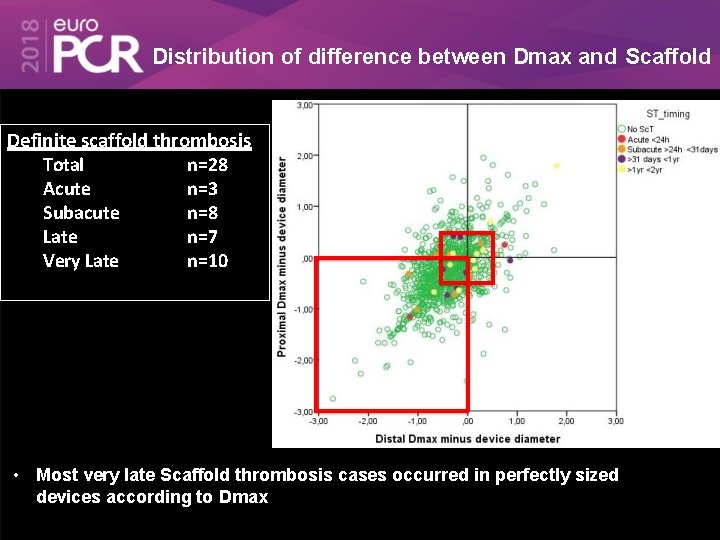
Distribution of difference between Dmax and Scaffold Definite scaffold thrombosis Total n=28 Acute n=3 Subacute n=8 Late n=7 Very Late n=10 • Most very late Scaffold thrombosis cases occurred in perfectly sized devices according to Dmax
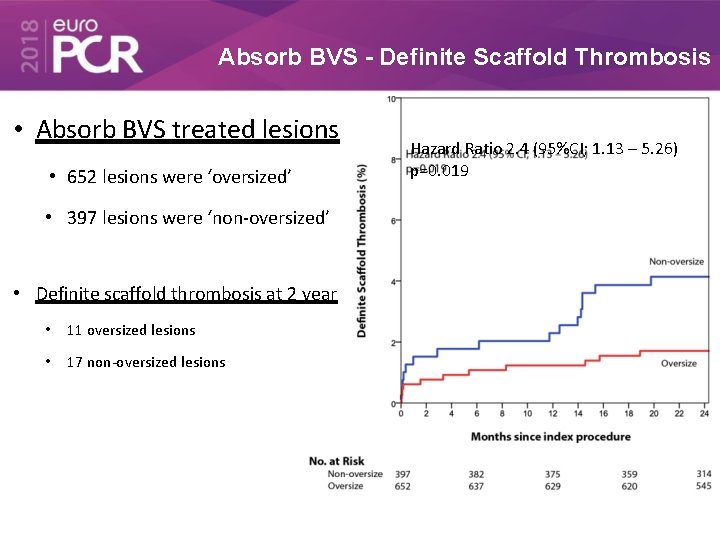
Absorb BVS - Definite Scaffold Thrombosis • Absorb BVS treated lesions • 652 lesions were ‘oversized’ • 397 lesions were ‘non-oversized’ • Definite scaffold thrombosis at 2 year • 11 oversized lesions • 17 non-oversized lesions Hazard Ratio 2. 4 (95%CI; 1. 13 – 5. 26) p=0. 019

Definite Scaffold Thrombosis 31 day landmark analysis Definite Scaffold Thrombosis Non-oversize Oversize n=10 n=7 Hazard Ratio 2. 42 (95%CI; 0. 92 – 6. 36) p=0. 069 1 year landmark analysis Definite Scaffold Thrombosis Non-oversize Oversize n=7 n=3 Hazard Ratio 4. 00 (95%CI; 1. 03 – 15. 45) p=0. 003
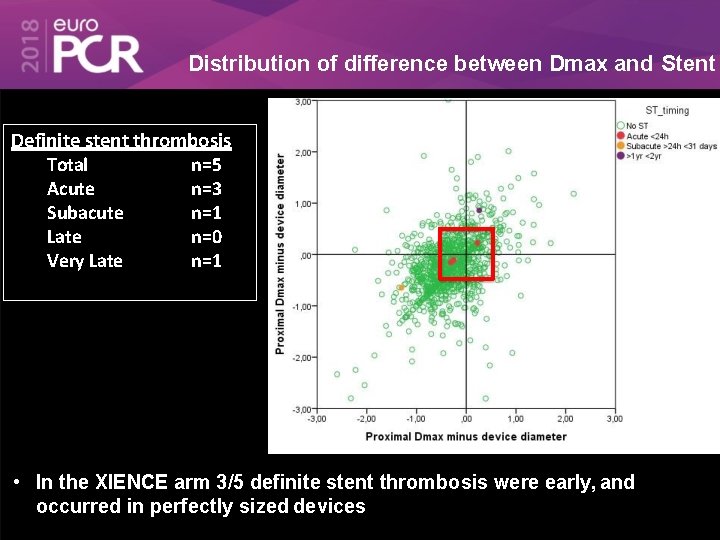
Distribution of difference between Dmax and Stent Definite stent thrombosis Total n=5 Acute n=3 Subacute n=1 Late n=0 Very Late n=1 • In the XIENCE arm 3/5 definite stent thrombosis were early, and occurred in perfectly sized devices
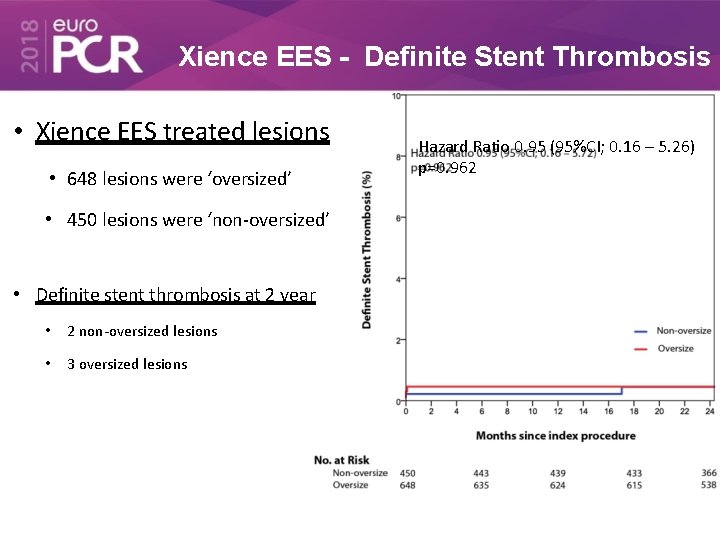
Xience EES - Definite Stent Thrombosis • Xience EES treated lesions • 648 lesions were ‘oversized’ • 450 lesions were ‘non-oversized’ • Definite stent thrombosis at 2 year • 2 non-oversized lesions • 3 oversized lesions Hazard Ratio 0. 95 (95%CI; 0. 16 – 5. 26) p=0. 962
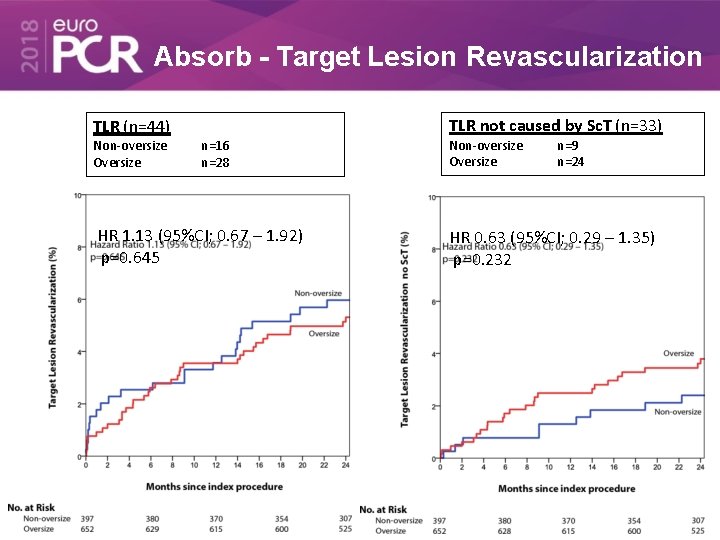
Absorb - Target Lesion Revascularization TLR (n=44) Non-oversize Oversize TLR not caused by Sc. T (n=33) n=16 n=28 HR 1. 13 (95%CI; 0. 67 – 1. 92) p=0. 645 Non-oversize Oversize n=9 n=24 HR 0. 63 (95%CI; 0. 29 – 1. 35) p=0. 232
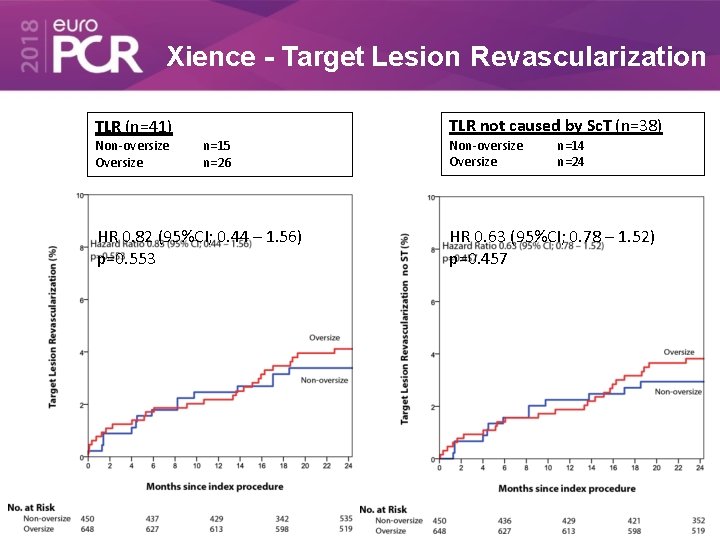
Xience - Target Lesion Revascularization TLR (n=41) Non-oversize Oversize TLR not caused by Sc. T (n=38) n=15 n=26 HR 0. 82 (95%CI; 0. 44 – 1. 56) p=0. 553 Non-oversize Oversize n=14 n=24 HR 0. 63 (95%CI; 0. 78 – 1. 52) p=0. 457

Conclusions • This AIDA substudy is the first to present Dmax analysis in both ABSORB BVS and Xience EES treated lesions. • It is also the first study to present the effect of Dmax lumen/device matching on lesion-oriented outcomes. • We found that definite scaffold thrombosis (both early and (very) late) occurred more frequently in non-oversized Absorb BVS treated lesions. • Most (very) late definite scaffold thrombosis cases occurred in perfectly sized Absorb BVS. • In the XIENCE arm 3/5 definite stent thrombosis were early, and occurred in perfectly sized devices.

Limitations 1. ) Device sizing based on QCA was not mandatory in AIDA. 2. ) Routine intravascular imaging has not been performed therefore, the study does not provide mechanistic insights in the occurence of events due to a device / artery mismatch. 3. ) This AIDA sub-study is a post-hoc study, therefore it is subject to underpowering, as all post-hoc studies.

Thank you for your attention!
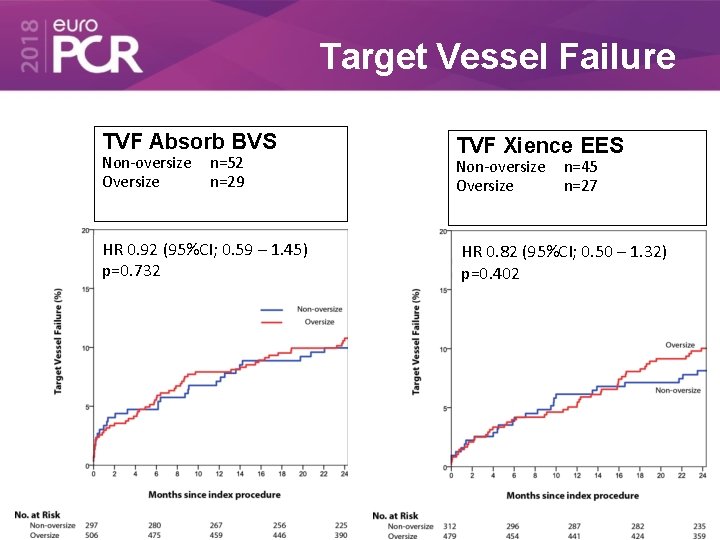
Target Vessel Failure TVF Absorb BVS Non-oversize Oversize n=52 n=29 HR 0. 92 (95%CI; 0. 59 – 1. 45) p=0. 732 TVF Xience EES Non-oversize Oversize n=45 n=27 HR 0. 82 (95%CI; 0. 50 – 1. 32) p=0. 402

Conclusions • This AIDA substudy is the first to present Dmax analysis in both ABSORB BVS and Xience EES treated lesions. • It is also the first study to present the effect of Dmax lumen/device matching on lesion-oriented outcomes. • We found that definite scaffold thrombosis (both early and (very) late) occurred more frequently in non-oversized Absorb BVS treated lesions. • Most (very) late definite scaffold thrombosis cases occurred in perfectly sized Absorb BVS. • In the XIENCE arm 3/5 definite stent thrombosis were early, and occurred in perfectly sized devices.
- Slides: 25