Reimbursement Landscape for the WATCHMAN Left Atrial Appendage

Reimbursement Landscape for the WATCHMAN™ Left Atrial Appendage Closure (LAAC) Device 1 SH-304702 -AB OCT 2015

Important Information Health economic and reimbursement information provided by Boston Scientific Corporation is gathered from third-party sources and is subject to change without notice as a result of complex and frequently changing laws, regulations, rules and policies. This information is presented for illustrative purposes only and does not constitute reimbursement or legal advice. Boston Scientific encourages providers to submit accurate and appropriate claims for services. It is always the provider’s responsibility to determine medical necessity, the proper site for delivery of any services and to submit appropriate codes, charges and modifiers for services that are rendered. Boston Scientific recommends that you consult with your payers, reimbursement specialists and/or legal counsel regarding coding, coverage and reimbursement matters. Boston Scientific does not promote the use of its products outside their FDA-approved labeling. Payer policies will vary and should be verified prior to treatment for limitations on diagnosis, coding or site of service requirements. The coding options listed within this guide are commonly used codes and are not intended to be an all-inclusive list. Providers are responsible for making appropriate decisions related to coding and reimbursement submissions. We recommend consulting your relevant manuals for appropriate coding options. CPT Copyright 2014 American Medical Association. All rights reserved. CPT is a registered trademark of the American Medical Association. Applicable FARS/DFARS Restrictions Apply to Government Use. Fee schedules, relative value units, conversion factors and/or related components are not assigned by the AMA, are not part of CPT, and the AMA is not recommending their use. The AMA does not directly or indirectly practice medicine or dispense medical services. The AMA assumes no liability for data contained or not contained herein Payer policies will vary and should be verified prior to treatment for limitations on diagnosis, coding or site of service requirements. The coding options listed within this guide are commonly used codes and are not intended to be an all-inclusive list. We recommend consulting your relevant manuals for appropriate coding options. 2 SH-304702 -AB OCT 2015

Agenda • Overview of WATCHMAN Reimbursement Landscape - Coverage - Other structural heart technologies • Physician Coding and Payment • Hospital Coding and Payment • Appeals Process • Reimbursement Resources 3 SH-304702 -AB OCT 2015

Overview of Reimbursement Landscape for the WATCHMAN™ Device 4 SH-304702 -AB OCT 2015
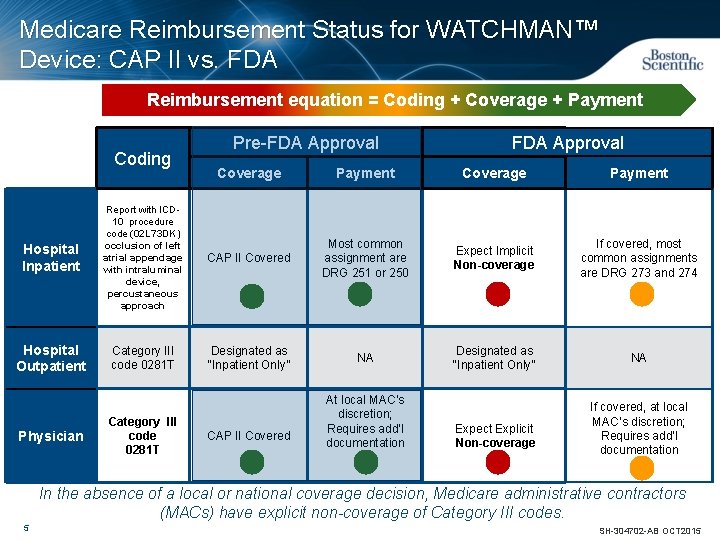
Medicare Reimbursement Status for WATCHMAN™ Device: CAP II vs. FDA Reimbursement equation = Coding + Coverage + Payment Coding Pre-FDA Approval Coverage Payment Hospital Inpatient Report with ICD 10 procedure code (02 L 73 DK) occlusion of left atrial appendage with intraluminal device, percustaneous approach CAP II Covered Most common assignment are DRG 251 or 250 Expect Implicit Non-coverage If covered, most common assignments are DRG 273 and 274 Hospital Outpatient Category III code 0281 T Designated as “Inpatient Only” NA Physician Category III code 0281 T CAP II Covered At local MAC’s discretion; Requires add’l documentation Expect Explicit Non-coverage If covered, at local MAC’s discretion; Requires add’l documentation In the absence of a local or national coverage decision, Medicare administrative contractors (MACs) have explicit non-coverage of Category III codes. 5 SH-304702 -AB OCT 2015

WATCHMAN™ Reimbursement pathway is not uncommon for new and innovative technologies Coverage gaps are routine for new technologies as Medicare and payers do not make coverage decisions prior to FDA approval Recent Technology FDA Approval Coding Coverage Payment Mitra. Clip™ (TMVR) 10/24/2013 Unlisted code: prior to 2014 Obtained NCD in August 2014 DRG 273 or 274, New Tech Add-on payment effective 10. 1. 2014 Obtained NCD May 2012 New DRGs 266267 effective FY 2015 Cat III code: Jan 2014 SAPIEN™ Valve (TAVR) 11/2/2011 Cat. III code: Jan 2011; CPT code: Jan 2013 WATCHMAN™ 03/13/2015 Cat III code: Jan 2012 TBD DRG 273 or 274 “Reimbursement” = Coding + Coverage + Payment Rates The WATCHMAN Therapy has established Coding, with associated Hospital Payment. Boston Scientific is working with CMS to define the Coverage Pathway Denials may be common so providers should seek coverage on a case-bycase basis by appealing denials if they occur. 6 SH-304702 -AB OCT 2015

Physician Coding and Reimbursement 7 SH-304702 -AB OCT 2015

Physician Reimbursement: Category III Code for WATCHMAN™ LAA Closure Procedure Category III CPT Code – Effective January 1, 2012 Code 0281 T Code Description Percutaneous transcatheter closure of the left atrial appendage with implant, including fluoroscopy, transseptal puncture, catheter placement(s), left atrial angiography, left atrial appendage angiography, radiological supervision and interpretation RVU Not assigned Payment Payer Discretion with supporting documentation Category III CPT codes: • Claim manually reviewed by payers. • No national payment rate established. Payment will vary and will be at discretion of the payer. • To ensure appropriate payment, physicians will select an existing CPT code (i. e. PFO or ASD procedure) as a reference to benchmark work and resources similar to WATCHMAN LAA closure procedure. CPT Copyright 2014. American Medical Association. All rights reserved. CPT is a registered trademark of the American Medical Association. Applicable FARS/DFARS Restrictions Apply to Government Use. Fee schedules, relative value units, conversion factors and/or related components are not assigned by the AMA, are not part of CPT, and the AMA is not recommending their use. The AMA does not directly or indirectly practice medicine or dispense medical services. The AMA assumes no liability for data contained or not contained herein 8 SH-304702 -AB OCT 2015

Physician Category III code claims: Benchmarking a comparator CPT code to WATCHMAN™ Device Process for submitting Category III code to payers includes: • For private payers, physicians should seek prior authorization. (Traditional Medicare does not perform prior authorizations. ) • Definition or description of the nature, extent, and need for the procedure • Provider’s time & effort - Utilize a comparator CPT code (with similar work, resources, time, competencies, and risk) for establishing equivalent work units as Category III codes do not have RVUs. • Equipment /device utilized • Copy of the FDA approval letter (BSC can supply) • Published clinical literature supporting the use of LAA closure with the WATCHMAN Device indicated for patient population Salaried physicians will need to identify RVU benchmark in communicating with their hospital administrators prior to performing WATCHMAN LAAC. 9 SH-304702 -AB OCT 2015

Inpatient Hospital Coding and Reimbursement 10 SH-304702 -AB OCT 2015

Hospital Inpatient Reimbursement Inpatient Coding ICD-10 procedure code: 02 L 73 DK Occlusion of left atrial appendage with intraluminal device, percutaneous approach DRG Assignment MS-DRG Descriptor FY 2016 National Base Payment Range 273 Percutaneous Cardiovascular Procedure without Coronary Artery Stent with MCC $20, 961 274 Percutaneous Cardiovascular Procedure without Coronary Artery Stent without MCC $14, 288 * Medicare Program: FY 2016 Hospital Inpatient Prospective Payment System, Final Rule; Updated October, 2015. Hospital inpatient payment rates will vary by hospital and are highly dependent on: patient’s condition, market conditions (capital and labor), new technology payments, medical education payments, disproportionate share payments, outlier payments and quality-based initiatives (readmissions reduction policy, and value-based incentive programs). • Medicare restricts WATCHMAN procedures to inpatient only site-ofservice 11 – “ 2 -Midnight Rule” not applicable to procedures restricted to inpatient only list. SH-304702 -AB OCT 2015

Best practices: Prior authorization and Appeals 12 SH-304702 -AB OCT 2015

Appeals Process: Medicare • Traditional Medicare does not offer prior authorizations – – Providers perform procedure based on medical necessity Consider Advanced Beneficiary Notice (ABN) Providers submit claims to MAC for processing Providers either receive payment or denial (*For Medicare Advantage Plans- physicians & hospitals should seek prior authorization) • Medicare has defined appeals process – Redetermination by CMS contractor (i. e. Medicare administrative Contractor - MAC) is the first step • Most providers generally proceed at initial “Redetermination” level – 120 day filing deadline from receipt of denial – Form CMS-20027 available to submit appeal Providers should anticipate appealing WATCHMAN ™ Device Medicare cases as this is the only mechanism for payment reconsideration. 13 SH-304702 -AB OCT 2015

Appeals Process: Private Payers Providers have the opportunity to request prior authorizations • Seek prior authorization from Private Payers – Payers may have non-coverage policies for Category III codes and LAAC procedures – Check payers’ policies as plans vary – BSC has sample prior authorization templates available • Providers should anticipate denials for prior authorizations. – This is not uncommon for new technologies – Verify payer timelines & processes for submitting appeals • Providers need to APPEAL pre-surgical prior authorizations upon receipt – – – 14 Provide supporting clinical documentation Defend WATCHMAN Implant therapy as most appropriate for your patient Request peer-to-peer review with like specialty (i. e. EP or cardiologist) Continue to follow up until decision obtained Request second level appeal if necessary SH-304702 -AB OCT 2015

Common themes of a successful appeal 1. Paint the Clinical Picture- Be specific! o Describe the WATCHMAN™ LAAC procedure o Provide rationale on why WATCHMAN Device is a medically appropriate alternative (compared to warfarin) for your patient o Provide clinical documentation (prior attempted treatments that failed) o Talk about patient quality of life 2. Highlight the extensive body of WATCHMAN clinical trials o Most studied device in the LAAC space with four pivotal trials o Over 2, 000 patients studied to date o Demonstrated safety of WATCHMAN Device in most recent clinical data (e. g. PREVAIL and PROTECT 4 year) 3. Provide copy of FDA approval letter o WATCHMAN Device is not an investigational device o Established safety and efficacy 15 SH-304702 -AB OCT 2015

When should I anticipate coverage? Coverage is dependent on timing & review cycles of payers & policy makers that are beyond BSC’s direct control What should I expect: • Coverage gaps are routine for novel technologies • Establishing coverage could not begin until after FDA approval – BSC has been working proactively with FDA, CMS, and physician specialty societies to minimize coverage gap • A coverage gap is anticipated now that we have FDA approval – BSC is proactively working with CMS to define coverage • In the near-term, it will be necessary for clinicians/hospitals to seek coverage on a case-by-case basis, and appeal denials if they occur 16 SH-304702 -AB OCT 2015

HE&R Resources for WATCHMAN 17 SH-304702 -AB OCT 2015

BSC has Reimbursement Tools & Resources for WATCHMAN™ LAAC Device • WATCHMAN Reimbursement Guide • Pre-authorization and Appeals Templates • WATCHMAN Hospital Economic Analysis (request from WATCHMAN Sales Manager) • Physician Category III Code Guide • Documentation & Impact on MS-DRG Guide • BSC Reimbursement Support Line at 1 -800 -CARDIAC All Reimbursement resources are easily accessible at www. bostonscientific. com As BSC continues to receive reimbursement updates on WATCHMAN, we will provide you timely communications. WATCHMAN is a registered or unregistered trademark of Boston Scientific Corporation or its affiliates. All other trademarks are the property of their respective owners. 18 SH-304702 -AB OCT 2015
- Slides: 18