Regulatory Requirement for Drug Coated Balloons Andrew Farb

Regulatory Requirement for Drug Coated Balloons Andrew Farb, MD Division of Cardiovascular Devices Center for Devices and Radiological Health (CDRH) andrew. farb@fda. hhs, gov DES Bioabsorbable and DCB Technologies CRT 2013 Washington, DC February 25, 2013 1

Andrew Farb, MD I/we have no real or apparent conflicts of interest to report.
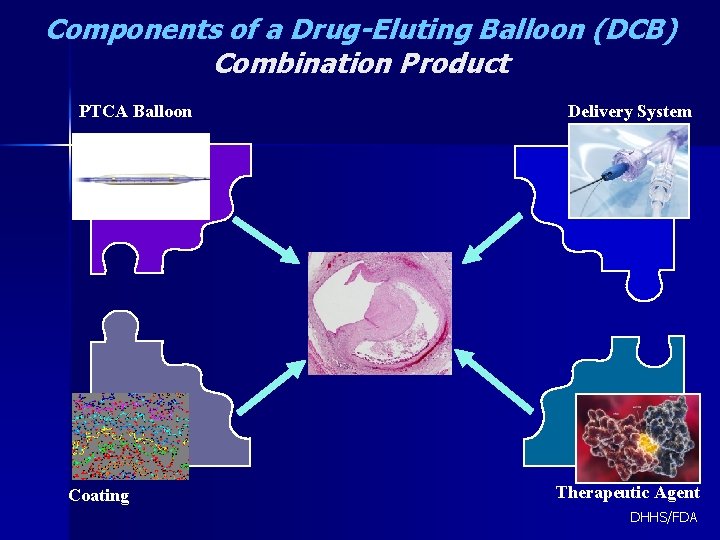
Components of a Drug-Eluting Balloon (DCB) Combination Product PTCA Balloon Coating Delivery System Therapeutic Agent DHHS/FDA

DCB Regulatory Submission Pathway Expectation that the DCB acts as both a dilatation catheter and drug-delivery device Otherwise primarily a drug delivery device Different regulatory path Investigational Device Exemption (IDE) Significant risk product (Class III) Required to conduct clinical trial in the US Premarket Approval Application (PMA) Comprehensive review of bench testing, animal studies, & all clinical data Establish a reasonable assurance of safety and effectiveness 4

Targeting IDE and PMA Approval To initiate an IDE study: Identify and justify bench & animal studies to provide adequate safety information to support the IDE Leverage available clinical information Include risk mitigation strategies in the clinical study protocol For PMA approval: Complete characterization of the finished sterilized product Some nonclinical studies can be performed concurrent with clinical studies Consider benefit-risk principles Underlying clinical condition (e. g. , life limiting or life threatening) Limitations of alternative therapies with respect to safety & effectiveness Discuss test methods early with FDA to avoid wasted steps and mis-spent resources 5

DCB Nonclinical Testing Benchmarks The finalized DCB: Can be reproducibly manufactured with respect to coating integrity and uniformity and shelf life stability Retains a consistent uniform amount of residual drug on the balloon following catheter advancement to the target lesion Provides consistent drug delivery to the artery that: Delivers drug along the length of the target lesion Uniform delivery can be affected by vessel geometry and plaque characteristics Can inhibit neointimal growth (expectation of effectiveness) Does not produce local toxic effects acutely and later (due to drug retention in surrounding tissue) Does not result systemic drug concentrations that produce off-target adverse events 6

DCB Bench Testing See FDA Guidance documents For PTCA component: Class II Special Controls Guidance for PTCA Catheters For Drug component: Draft Coronary DES Guidance Bench tests to support indication (e. g. , coronary vs. peripheral arteries, ISR, small vessels, bifurcation side branches) Testing to evaluate expected worst case clinical use and encompass product matrix Address use of DCB for multiple inflations (if applicable) 7

DCB Bench Testing Manufacturing Coating uniformity & integrity Processing effects on coating Spatial distribution of coating Drug retention & release from balloon Particulate generation (size & solubility) Realities of bench testing evaluation Some traditional test methods not applicable or lack acceptance criteria Acceptable in vivo animal study safety data and available clinical data may be leveraged to address concerns regarding: High pharm tox drug levels Particulate generation 8

In Vitro and In Vivo PK Evaluation Assess: Effect of surface texture and balloon folding on drug bioavailability Drug lost/retained during balloon transit and inflation Serum and tissue levels Local tissue dose/response to establish the lowest effective dose 9

Animal Studies: Safety Assess simulated use performance & handling Deliverability Balloon inflation (+multiple inflations, if applicable) Balloon withdrawal Focus on local toxicity from high dose drug delivery Assess healing Thrombus & Inflammation Re-endothelialization: SEM Edge effects & remodeling (positive/negative) Safety margin overdose studies Assess downstream effects 10

Animal Studies Other Considerations GLP or justify non-GLP Yucatan miniature swine or Sinclair adults Establish expectation of effectiveness Inhibition of neointimal proliferation Expected time points for tissue harvest & analysis 3 -5 days 30 days 90 days* 180 days *Can be used to support initiation of IDE if results acceptable 11

Clinical Studies Early feasibility and traditional feasibility studies Initial assessment of device handling & performance, proof of concept, and safety Acceptable results to justify a larger pivotal trial Pivotal trial RCT recommended Worldwide clinical experience Leverage supportive data of DCB safety and effectiveness 12

Coronary Artery Pivotal Trial Elements Primary endpoint - Target lesion failure Target lesions & control group Denovo CAD (e. g. , workhouse, small vessels, SB bifurcation treatment) Control: Approved DES or BMS (in patients not able or unlikely to comply with prolonged DAPT use) In-stent restenosis Control: Approved DES for ISR or justify off-label use of an approved DES If non-inferiority design vs. DES, chose clinically acceptable delta that also maintains headroom vs. historical outcomes associated with BMS (denovo lesions) or brachytherapy (ISR) 13

Antiplatelet Therapy for Coronary Applications Propose and justify duration of P 2 Y 12 receptor inhibitor use post-DCB use Capture subject compliance with dual antiplatelet therapy 14

Femoropopliteal Pivotal Trial Elements Primary effectiveness endpoint: Patency at 1 year Primary safety endpoint: Composite of 30 day death and 1 year amputation, re-intervention, or limb-related death Control: PTA with bailout stenting or approved stents (discuss options with FDA) Trial designs Vs. PTA Superiority for effectiveness Non-inferiority for safety Vs. Stent – Discuss superiority & non-inferiority designs P-ARC effort underway to propose consensus definitions for events and study endpoints 15

Other Clinical Trial Considerations Uniform pre-dilatation procedure Clarify balloon inflation time & single use or multiple inflations of an individual DCB Assess procedural results Acute device success Achievement of a acceptable post-deployment stenosis with the test device alone Arterial dissection rates & recoil Procedure success: Includes DCB plus adjunctive procedures/devices Need for adjunctive stent implantation may confound acute & late results Clinical success: Device success without in-hospital MACE 16

Final Regulatory Comments Early discussions with FDA via the pre-submission process recommended Nonclinical testing program Discuss non-US clinical data – Study design, events definitions & adjudication, long-term follow-up, poolability Post-approval studies (PAS) Long-term follow-up of subjects in pre-market studies Separate PAS cohort to evaluate real world use and capture low frequency events Satisfactory data for a coronary indication does not automatically equal satisfactory data for a peripheral indication and vice versa 17
- Slides: 17