Regulation of Pyruvate Kinase Allosterically activated by fructose1

Regulation of Pyruvate Kinase • Allosterically activated by fructose-1, 6 -bisphosphate – increase flow through glycolysis • Allosterically inhibited by signs of abundant energy supply (all tissues) – ATP – acetyl-Co. A and long-chain fatty acids – alanine (enough amino acids) • Inactivated by phosphorylation in response to signs of glucose depletion (glucagon) (liver only) – Glucose from liver is exported to the brain and other vital organs.
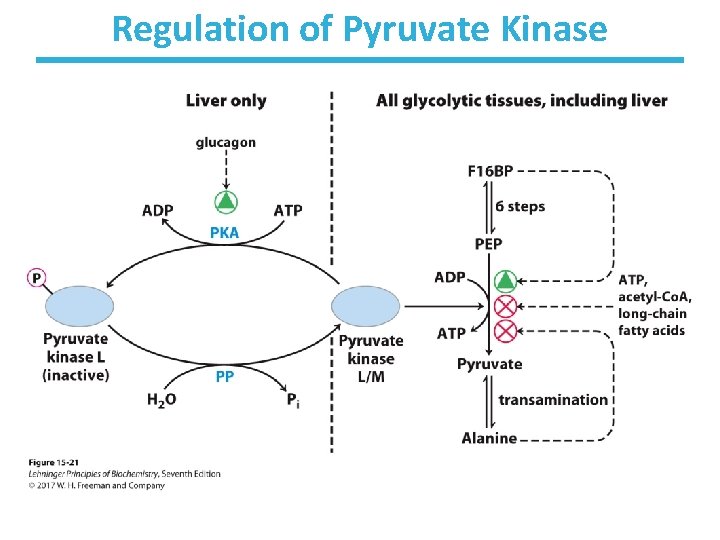
Regulation of Pyruvate Kinase

Two Alternative Fates for Pyruvate • Pyruvate can be a source of new glucose. – store energy as glycogen – generate NADPH via pentose phosphate pathway • Pyruvate can be a source of acetyl-Co. A. – store energy as body fat – make ATP via citric acid cycle • Acetyl-Co. A stimulates glucose synthesis via gluconeogenesis by activating pyruvate carboxylase.
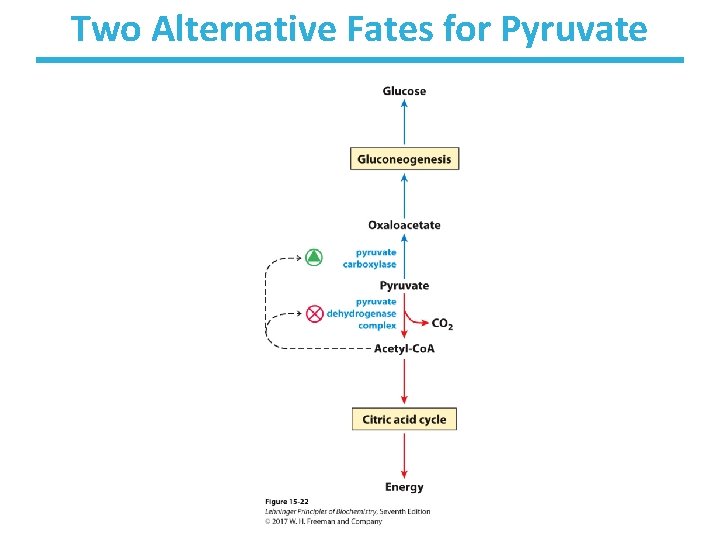
Two Alternative Fates for Pyruvate

The Amount of Many Metabolic Enzymes Is Controlled by Transcription TABLE 15 -5 Some of the Many Genes Regulated by Insulin Change in gene expression Role in glucose metabolism Increased expression Hexokinase II Hexokinase IV Phosphofructokinase-1 (PFK-1) PFK-2/FBPase-2 Pyruvate kinase Essential for glycolysis, which consumes glucose for energy Glucose 6 -phosphate dehydrogenase 6 -Phosphogluconate dehydrogenase Malic enzyme Produce NADPH, which is essential for conversion of glucose to lipids ATP-citrate lyase Pyruvate dehydrogenase Produce acetyl-Co. A, which is essential for conversion of glucose to lipids Acetyl-Co. A carboxylase Fatty acid synthase complex Stearoyl-Co. A dehydrogenase Acyl-Co. A–glycerol transferases Essential for conversion of glucose to lipids Decreased expression PEP carboxykinase Glucose 6 -phosphatase (catalytic subunit) Essential for glucose production by gluconeogenesis
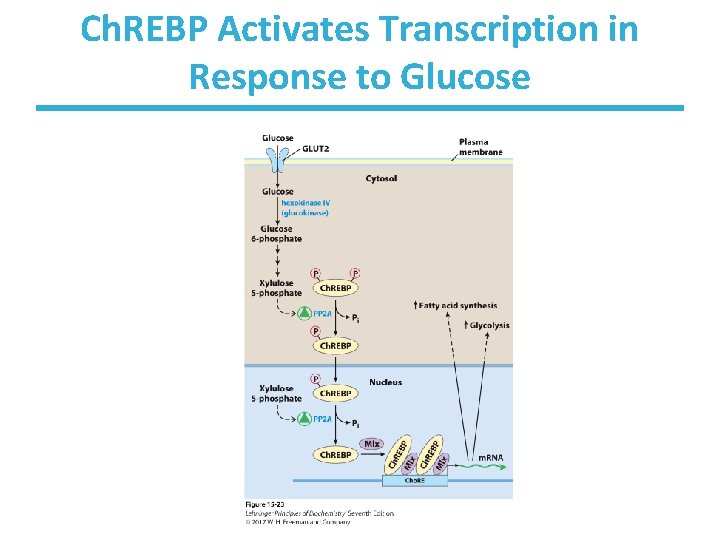
Ch. REBP Activates Transcription in Response to Glucose
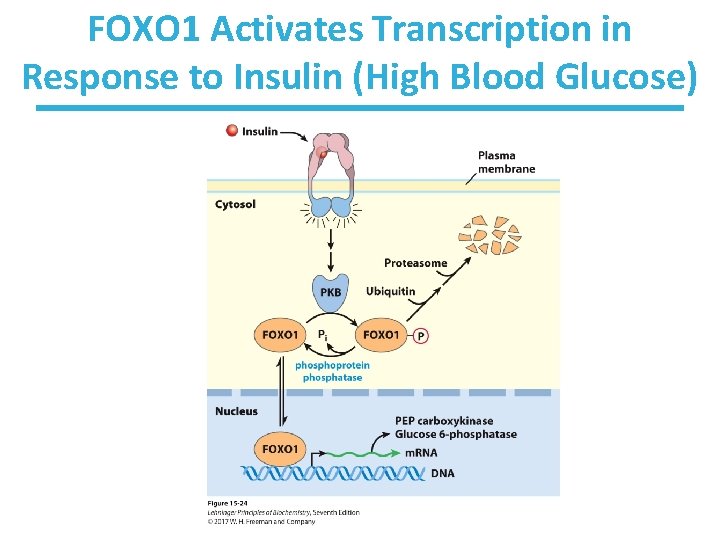
FOXO 1 Activates Transcription in Response to Insulin (High Blood Glucose)
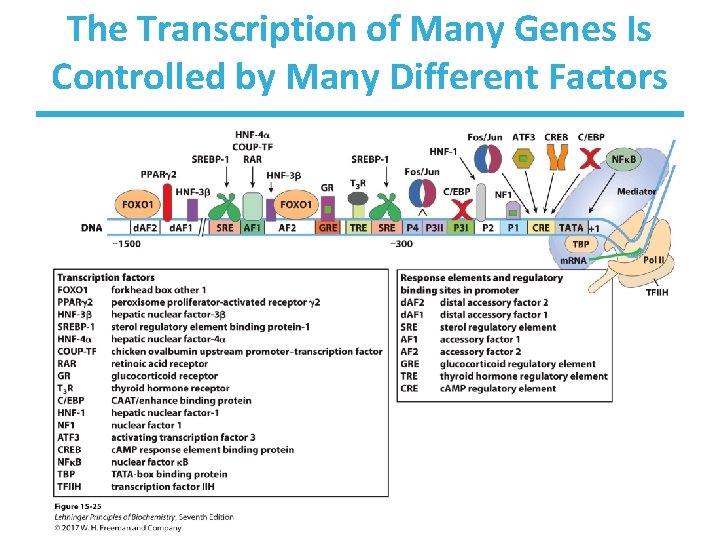
The Transcription of Many Genes Is Controlled by Many Different Factors

Glucose Can Be Stored for Later Use as Glycogen • Glycogen is a branched polymer of α(1 4)-linked glucose with α(1 6) linkages every 12 to 14 glucose units. • Glycogen storage occurs mainly in the liver and muscle. • Glycogen is degraded to glucose units for use in energy production. • Glycogen can be made from excess blood glucose or recycling of glucogenic metabolites like lactate or certain amino acids.
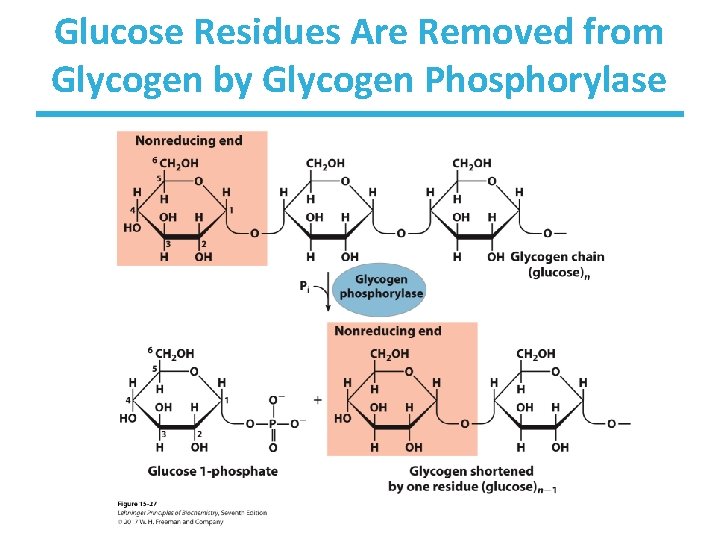
Glucose Residues Are Removed from Glycogen by Glycogen Phosphorylase

Dealing with Branch Points in Glycogen • Glycogen phosphorylase works on nonreducing ends until it reaches four residues from an ( 1 6) branch point. • Debranching enzyme transfers a block of three residues to the nonreducing end of the chain. • Debranching enzyme cleaves the single remaining ( 1 6)-linked glucose, which becomes a free glucose unit (i. e. , NOT glucose-1 phosphate).
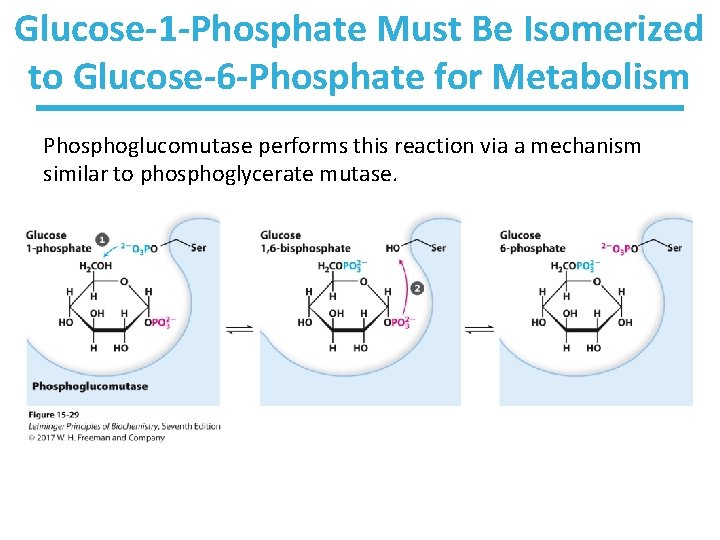
Glucose-1 -Phosphate Must Be Isomerized to Glucose-6 -Phosphate for Metabolism Phosphoglucomutase performs this reaction via a mechanism similar to phosphoglycerate mutase.
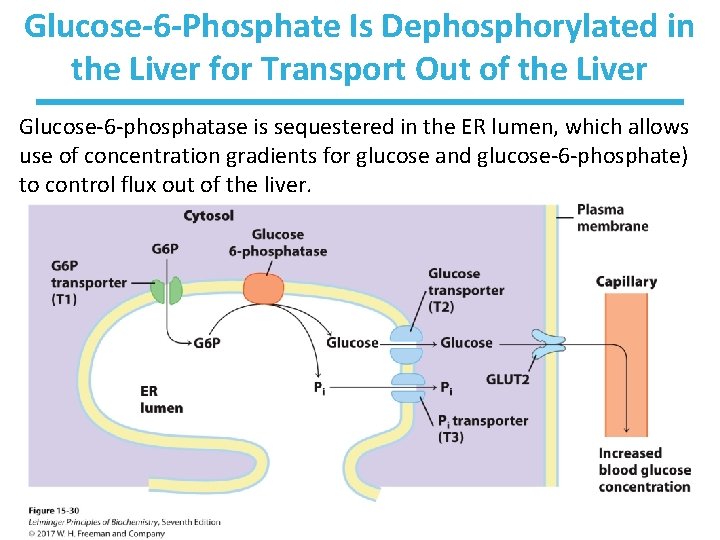
Glucose-6 -Phosphate Is Dephosphorylated in the Liver for Transport Out of the Liver Glucose-6 -phosphatase is sequestered in the ER lumen, which allows use of concentration gradients for glucose and glucose-6 -phosphate) to control flux out of the liver.

Glycogen Synthesis from Glucose Occurs in Multiple Steps • Synthesis of glycogen requires more enzymes and metabolic intermediates than glycogen degradation. • Blood glucose must be: • phosphorylated • labeled with UDP • added to glycogen Multiple steps allow for multiple points in regulation.
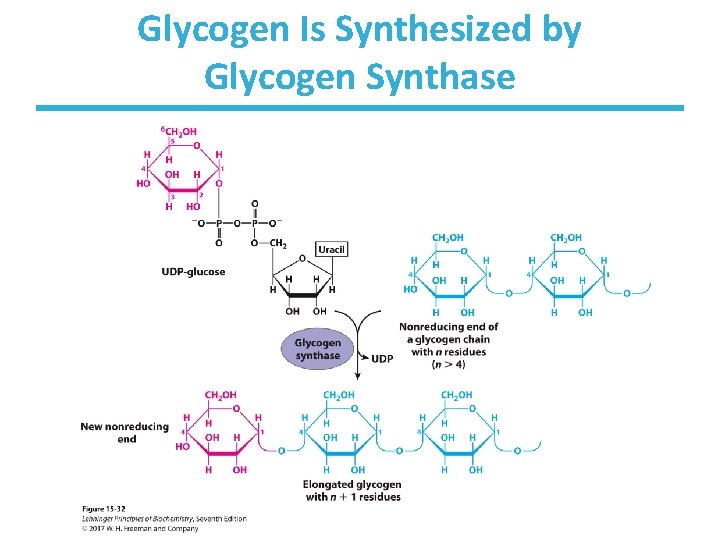
Glycogen Is Synthesized by Glycogen Synthase
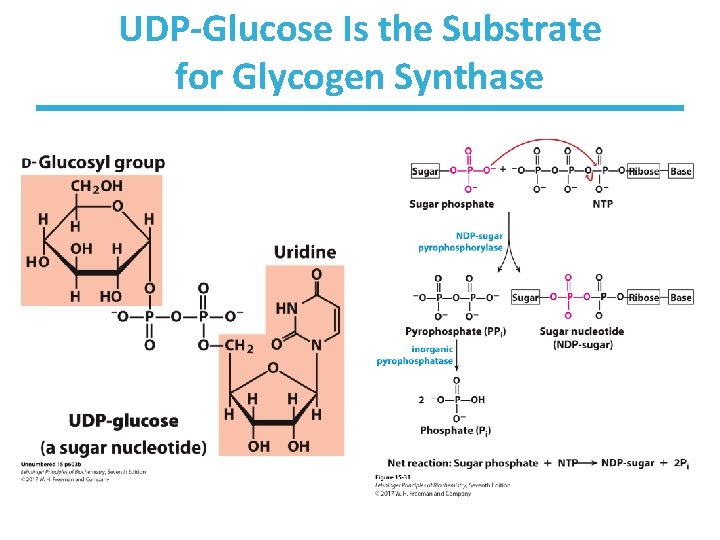
UDP-Glucose Is the Substrate for Glycogen Synthase
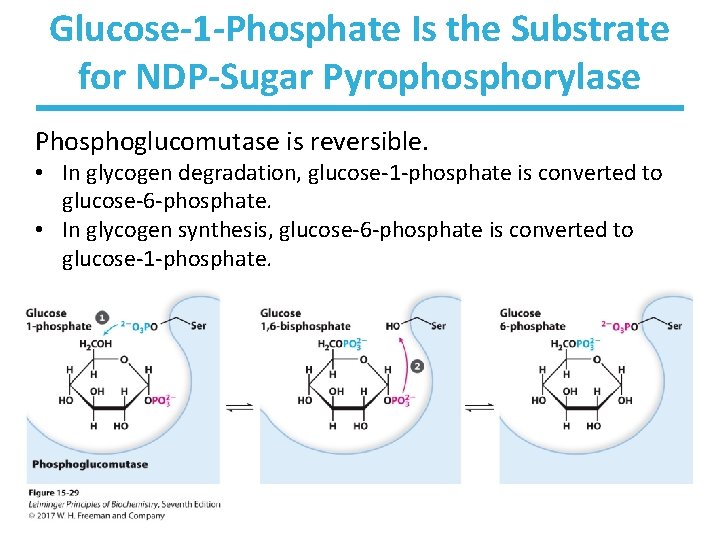
Glucose-1 -Phosphate Is the Substrate for NDP-Sugar Pyrophosphorylase Phosphoglucomutase is reversible. • In glycogen degradation, glucose-1 -phosphate is converted to glucose-6 -phosphate. • In glycogen synthesis, glucose-6 -phosphate is converted to glucose-1 -phosphate.
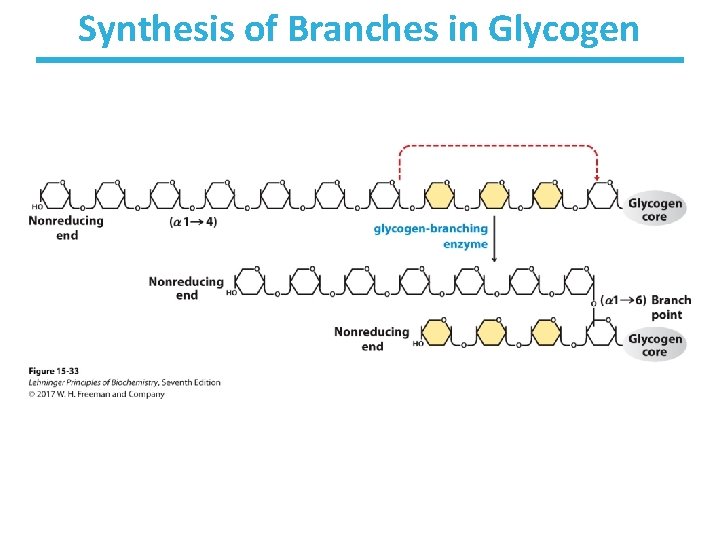
Synthesis of Branches in Glycogen
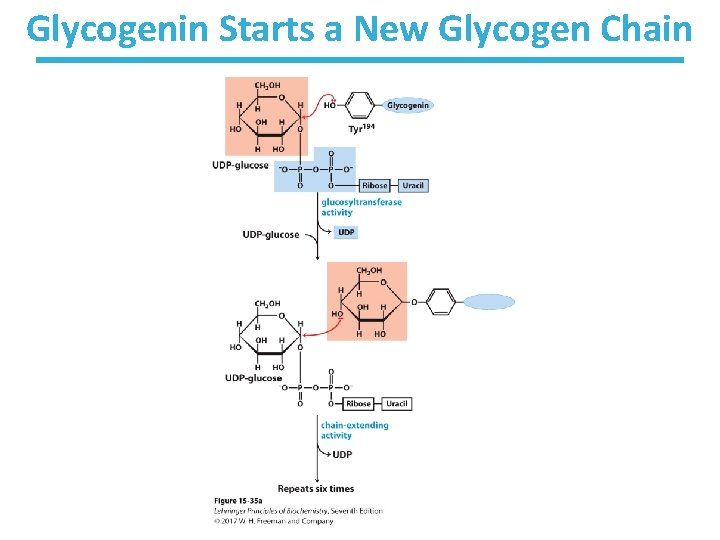
Glycogenin Starts a New Glycogen Chain
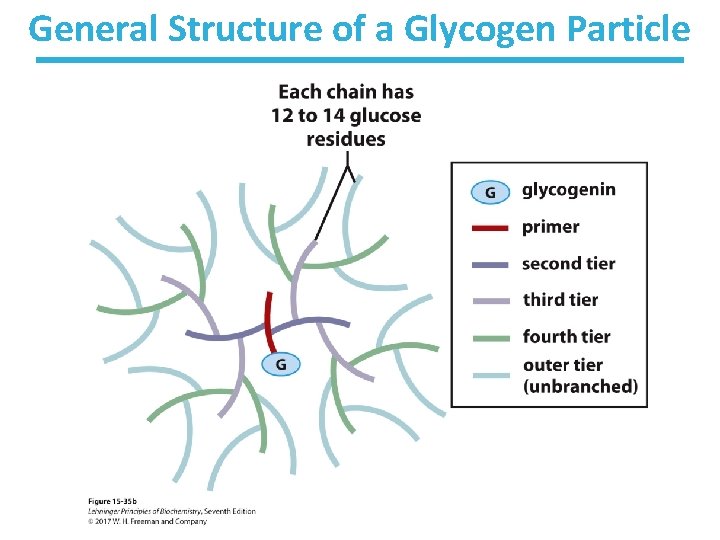
General Structure of a Glycogen Particle
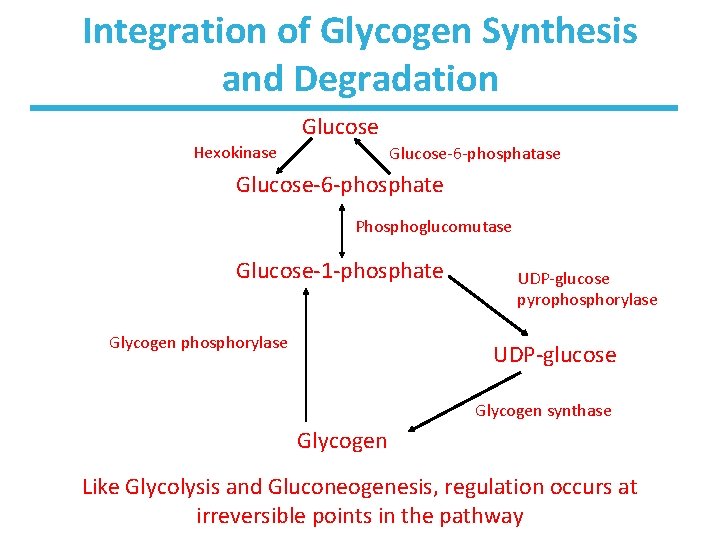
Integration of Glycogen Synthesis and Degradation Glucose Hexokinase Glucose-6 -phosphate Phosphoglucomutase Glucose-1 -phosphate Glycogen phosphorylase UDP-glucose pyrophosphorylase UDP-glucose Glycogen synthase Glycogen Like Glycolysis and Gluconeogenesis, regulation occurs at irreversible points in the pathway
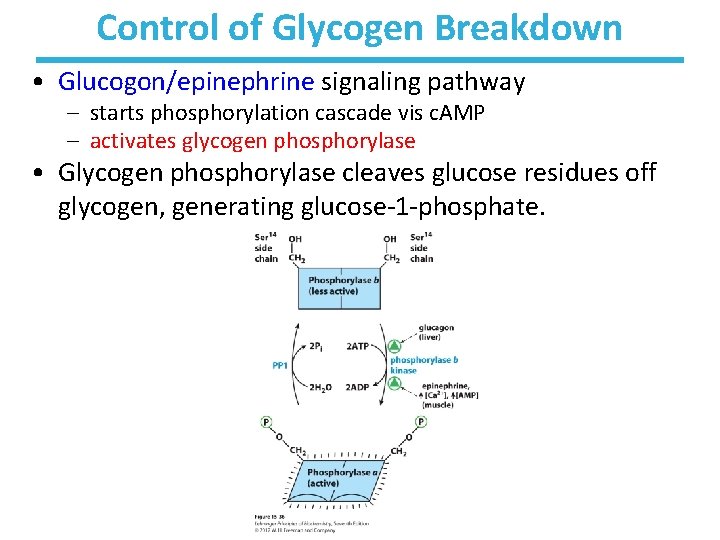
Control of Glycogen Breakdown • Glucogon/epinephrine signaling pathway – starts phosphorylation cascade vis c. AMP – activates glycogen phosphorylase • Glycogen phosphorylase cleaves glucose residues off glycogen, generating glucose-1 -phosphate.
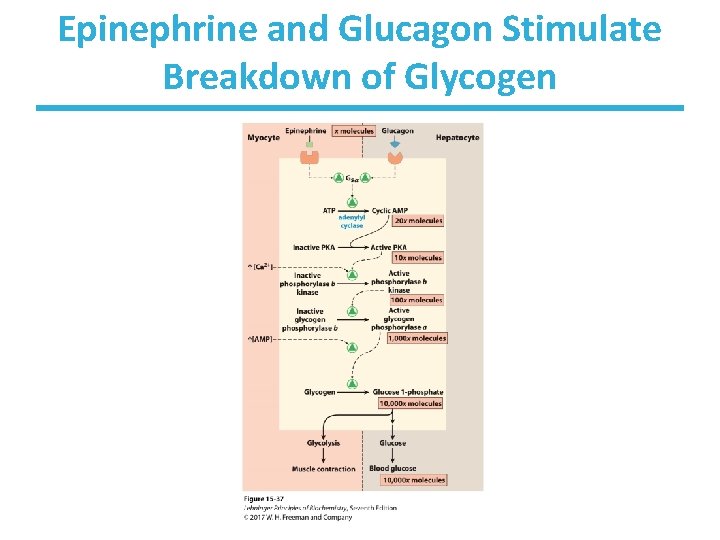
Epinephrine and Glucagon Stimulate Breakdown of Glycogen

Control of Glycogen Synthesis • Insulin-signaling pathway – increases glucose import into muscle – stimulates the activity of muscle hexokinase – activates glycogen synthase • Increased hexokinase activity enables activation of glucose. • Glycogen synthase makes glycogen for energy storage.
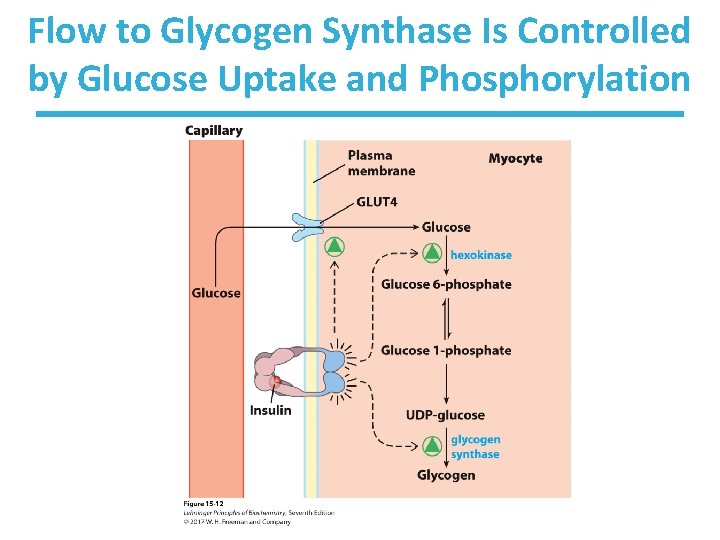
Flow to Glycogen Synthase Is Controlled by Glucose Uptake and Phosphorylation
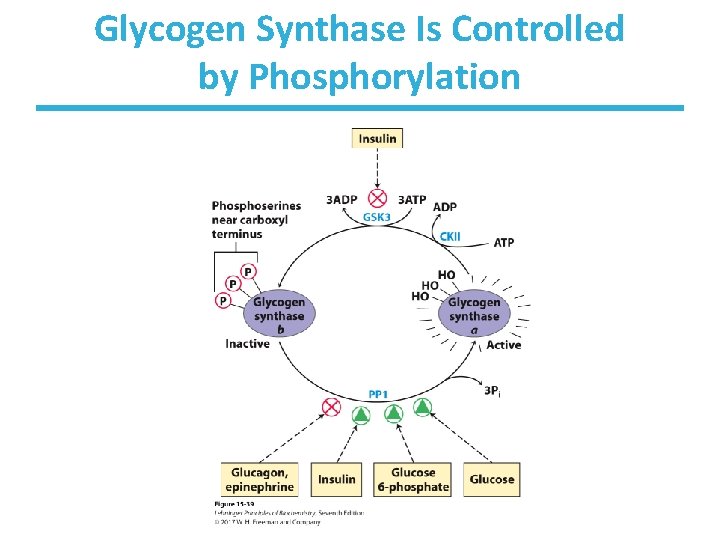
Glycogen Synthase Is Controlled by Phosphorylation
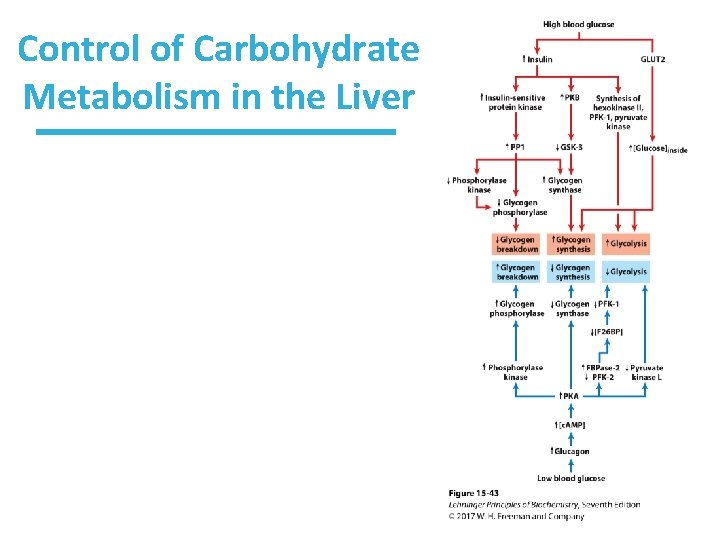
Control of Carbohydrate Metabolism in the Liver
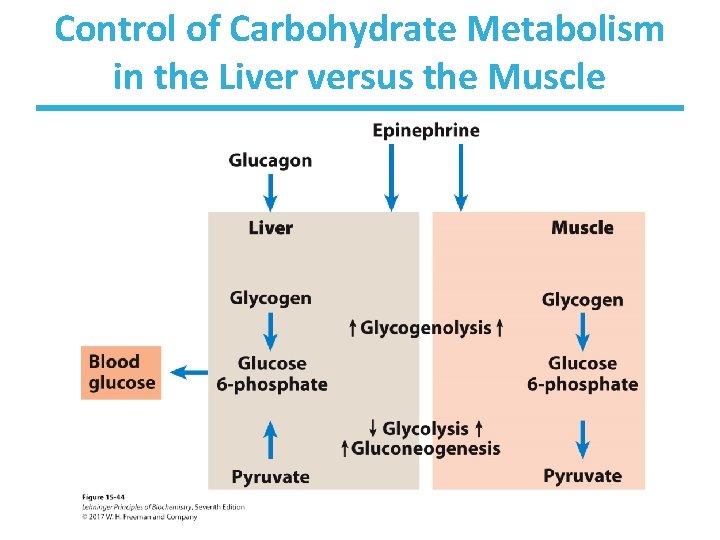
Control of Carbohydrate Metabolism in the Liver versus the Muscle
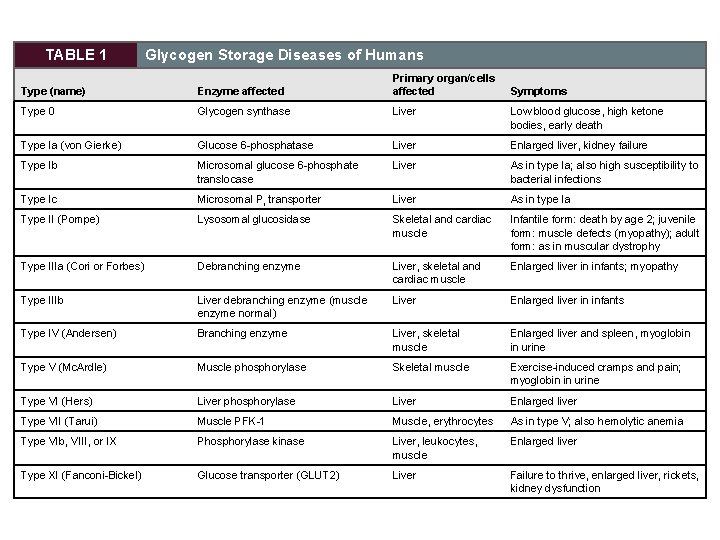
TABLE 1 Glycogen Storage Diseases of Humans Type (name) Enzyme affected Primary organ/cells affected Type 0 Glycogen synthase Liver Low blood glucose, high ketone bodies, early death Type Ia (von Gierke) Glucose 6 -phosphatase Liver Enlarged liver, kidney failure Type Ib Microsomal glucose 6 -phosphate translocase Liver As in type Ia; also high susceptibility to bacterial infections Type Ic Microsomal Pi transporter Liver As in type Ia Type II (Pompe) Lysosomal glucosidase Skeletal and cardiac muscle Infantile form: death by age 2; juvenile form: muscle defects (myopathy); adult form: as in muscular dystrophy Type IIIa (Cori or Forbes) Debranching enzyme Liver, skeletal and cardiac muscle Enlarged liver in infants; myopathy Type IIIb Liver debranching enzyme (muscle enzyme normal) Liver Enlarged liver in infants Type IV (Andersen) Branching enzyme Liver, skeletal muscle Enlarged liver and spleen, myoglobin in urine Type V (Mc. Ardle) Muscle phosphorylase Skeletal muscle Exercise-induced cramps and pain; myoglobin in urine Type VI (Hers) Liver phosphorylase Liver Enlarged liver Type VII (Tarui) Muscle PFK-1 Muscle, erythrocytes As in type V; also hemolytic anemia Type VIb, VIII, or IX Phosphorylase kinase Liver, leukocytes, muscle Enlarged liver Type XI (Fanconi-Bickel) Glucose transporter (GLUT 2) Liver Failure to thrive, enlarged liver, rickets, kidney dysfunction Symptoms

Chapter 15: Summary In this chapter, we learned that: • living organisms regulate the flux of metabolites through metabolic pathways by – increasing or decreasing enzyme concentrations – activating or inactivating key enzymes in the pathway • the activity of key enzymes in glycolysis and gluconeogenesis is tightly and coordinately regulated via various activating and inhibiting metabolites • glycogen synthesis and degradation is regulated by hormones insulin, epinephrine, and glucagon that report on the levels of glucose in the body
- Slides: 30