Regulation of oxygen consumption by local oxygen concentration

Regulation of oxygen consumption by local oxygen concentration in pre-vascular tissue spheroids Eric Krauland Shawdee Eshghi December 12, 2002 BE. 400
Motivations n n Mammalian embryos are served by diffusion until implantantion in uterine wall Embryos have adapted to survive in low oxygen conditions Embryoid bodies serve as a good in vitro model of embryogenesis, recreating gastrulation, hematopoiesis, and angiogenesis Pre-vascular tumors provide another model system to study tissue/cellular response to diffusional transport of oxygen
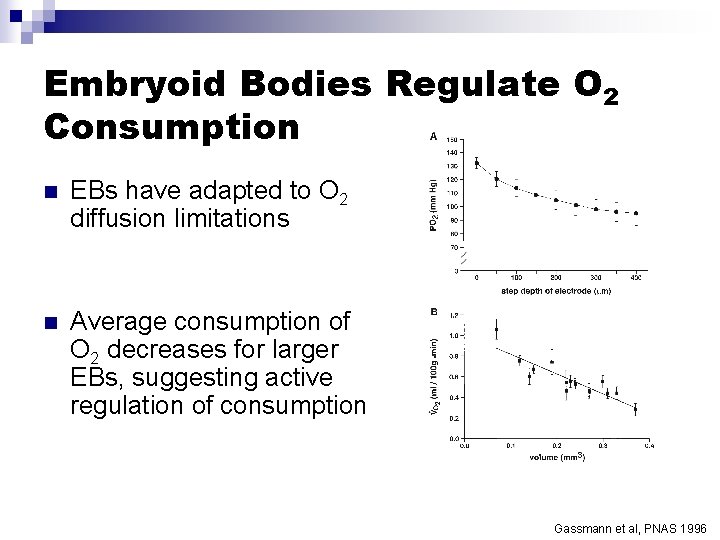
Embryoid Bodies Regulate O 2 Consumption n EBs have adapted to O 2 diffusion limitations n Average consumption of O 2 decreases for larger EBs, suggesting active regulation of consumption Gassmann et al, PNAS 1996
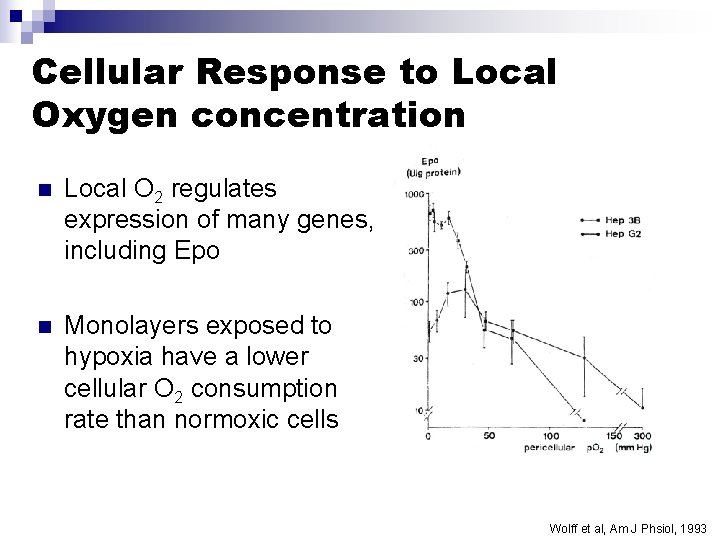
Cellular Response to Local Oxygen concentration n Local O 2 regulates expression of many genes, including Epo n Monolayers exposed to hypoxia have a lower cellular O 2 consumption rate than normoxic cells Wolff et al, Am J Phsiol, 1993
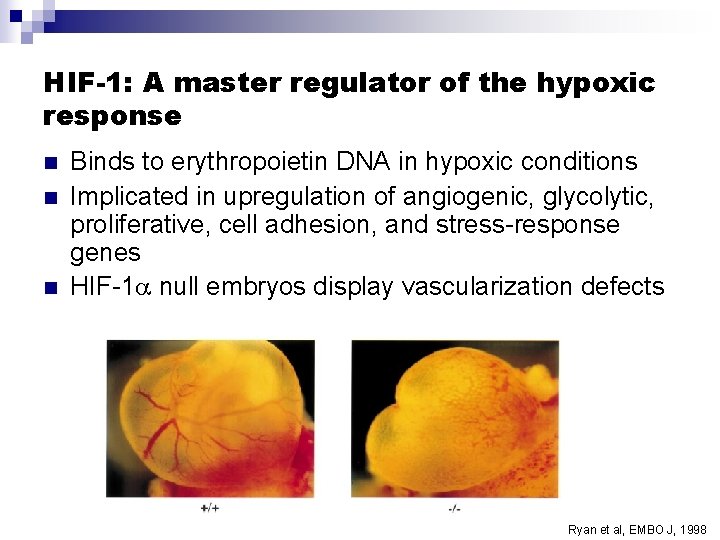
HIF-1: A master regulator of the hypoxic response n n n Binds to erythropoietin DNA in hypoxic conditions Implicated in upregulation of angiogenic, glycolytic, proliferative, cell adhesion, and stress-response genes HIF-1 null embryos display vascularization defects Ryan et al, EMBO J, 1998
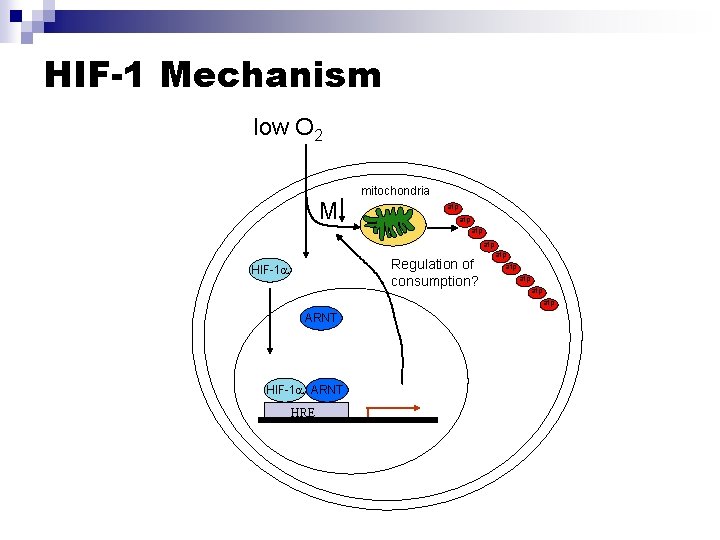
HIF-1 Mechanism low O 2 M mitochondria atp atp Regulation of HIF-1 Ub Ub Ub consumption? HIF-1 atp atp atp ARNT HIF-1 ARNT HRE

Questions to be addressed: 1) Do embryoid bodies and tumor spheroids sense O 2 concentration and regulate consumption on a cellular/local basis? 2) Does this control mechanism rely on HIF-1 gene regulation?
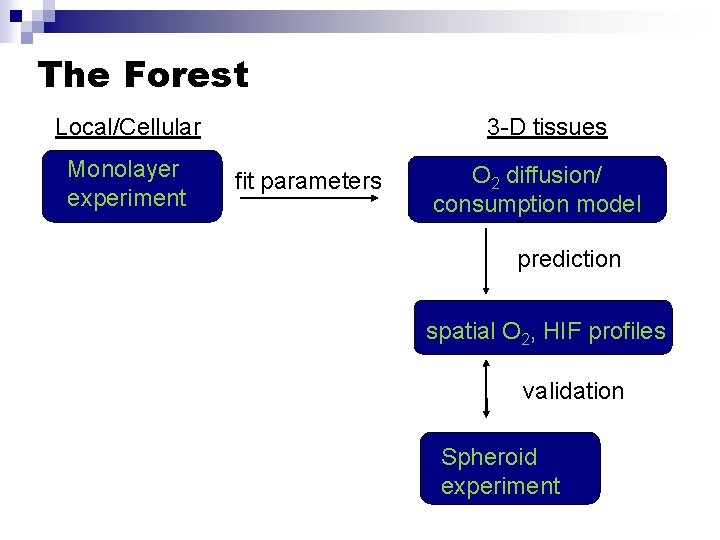
The Forest Local/Cellular Monolayer experiment 3 -D tissues fit parameters O 2 diffusion/ consumption model prediction spatial O 2, HIF profiles validation Spheroid experiment
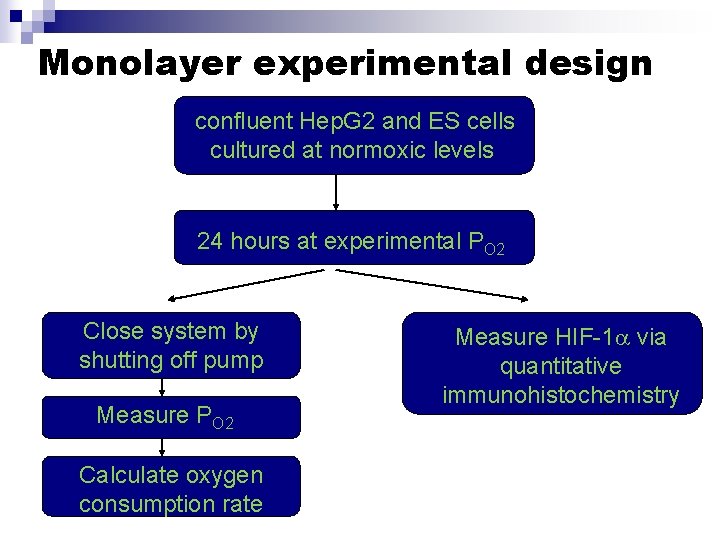
Monolayer experimental design confluent Hep. G 2 and ES cells cultured at normoxic levels 24 hours at experimental PO 2 Close system by shutting off pump Measure PO 2 Calculate oxygen consumption rate Measure HIF-1 via quantitative immunohistochemistry
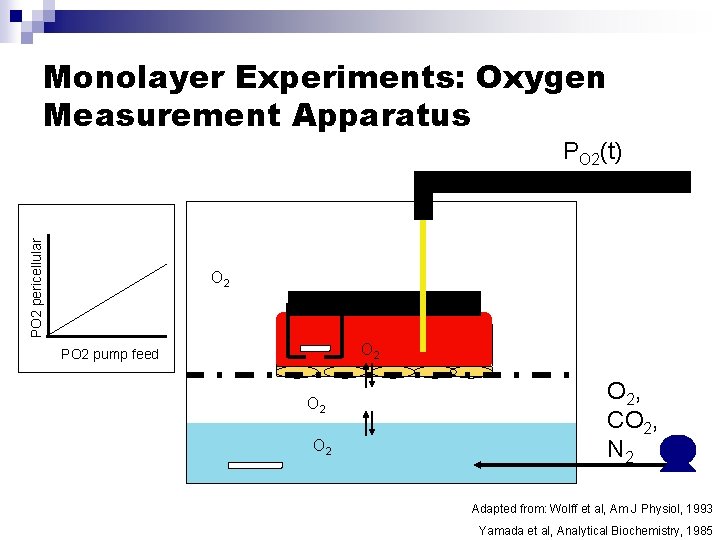
Monolayer Experiments: Oxygen Measurement Apparatus PO 2 pericellular PO 2(t) O 2 PO 2 pump feed O 2 O 2, CO 2, N 2 Adapted from: Wolff et al, Am J Physiol, 1993 Yamada et al, Analytical Biochemistry, 1985
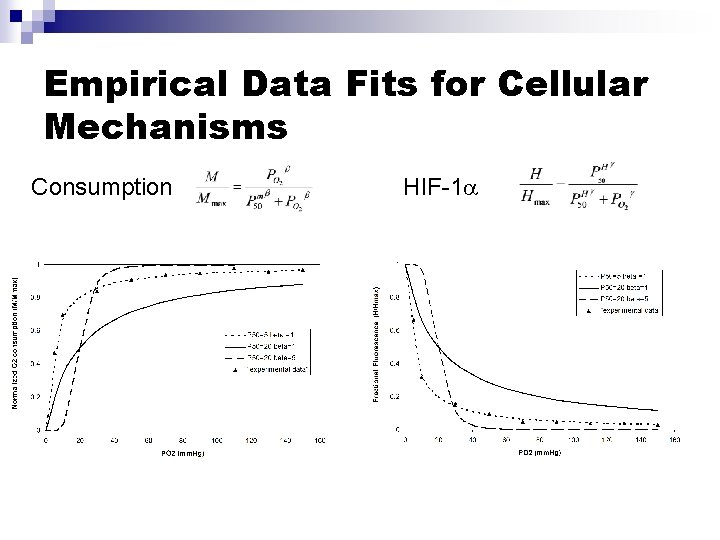
Empirical Data Fits for Cellular Mechanisms Consumption HIF-1

Tissue Diffusion/Consumption Model Governing Equations: Geometry: Spherical Tissue Mass PO 2(R)=PR MO 2(PO 2) K, r (1) zero BCs: (2) PO 2(r) (3) r=R • PO 2 Partial pressure of Oxygen • K Krogh’s Diffusion coefficient • MO 2 Vol. Tissue Consumption of O 2 • Tissue cellular density Empirical Consumption Equation: (1) (4) Under pseudo-steady conditions PO 2/ t 0:
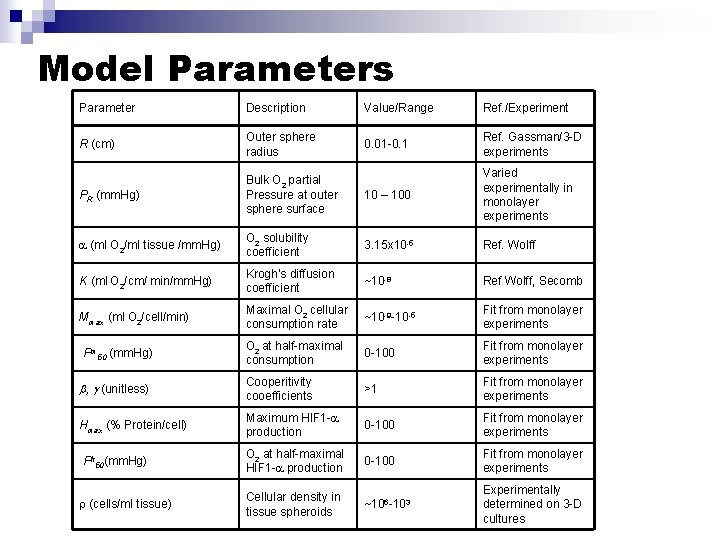
Model Parameters Parameter Description Value/Range Ref. /Experiment R (cm) Outer sphere radius 0. 01 -0. 1 Ref. Gassman/3 -D experiments PR (mm. Hg) Bulk O 2 partial Pressure at outer sphere surface 10 – 100 Varied experimentally in monolayer experiments (ml O 2/ml tissue /mm. Hg) O 2 solubility coefficient 3. 15 x 10 -5 Ref. Wolff K (ml O 2/cm/ min/mm. Hg) Krogh’s diffusion coefficient ~10 -8 Ref Wolff, Secomb Mmax (ml O 2/cell/min) Maximal O 2 cellular consumption rate ~10 -9 -10 -5 Fit from monolayer experiments Pm 50 (mm. Hg) O 2 at half-maximal consumption 0 -100 Fit from monolayer experiments , (unitless) Cooperitivity cooefficients >1 Fit from monolayer experiments Hmax (% Protein/cell) Maximum HIF 1 - production 0 -100 Fit from monolayer experiments Ph 50(mm. Hg) O 2 at half-maximal HIF 1 - production 0 -100 Fit from monolayer experiments (cells/ml tissue) Cellular density in tissue spheroids ~106 -103 Experimentally determined on 3 -D cultures

Non-dimensionalize Model Defining Dimensionless Parameters: Get non-dimensional governing equation and B. C. : (symmetry) 3 Dimensionless parameters-govern behavior of differential equation: 1) 2) 3)
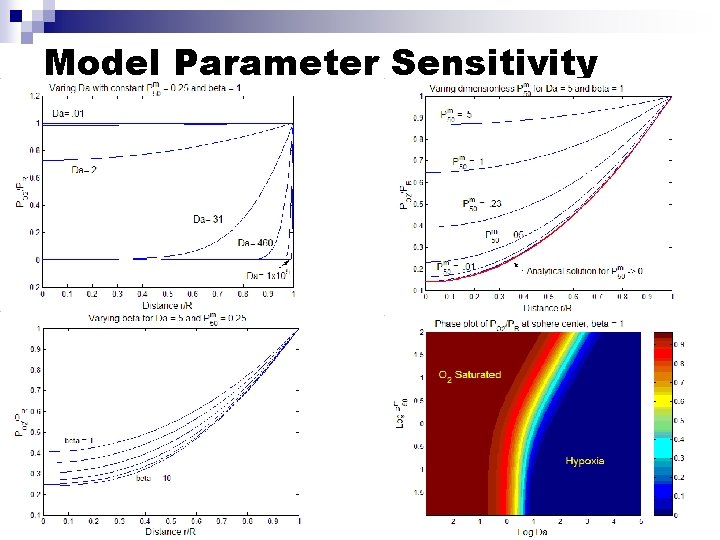
Model Parameter Sensitivity
The Forest: Again Local/Cellular Monolayer experiment 3 -D tissues fit parameters O 2 diffusion/ consumption model prediction spatial O 2, HIF profiles validation Spheroid experiment
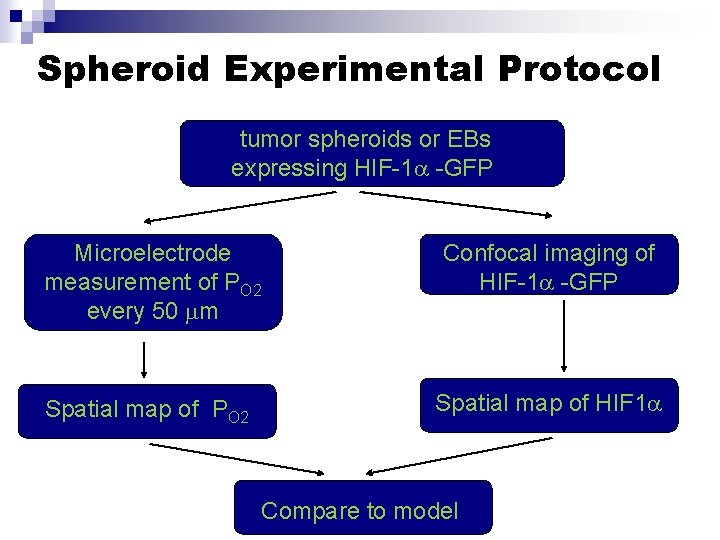
Spheroid Experimental Protocol tumor spheroids or EBs expressing HIF-1 -GFP Microelectrode measurement of PO 2 every 50 m Confocal imaging of HIF-1 -GFP Spatial map of PO 2 Spatial map of HIF 1 Compare to model
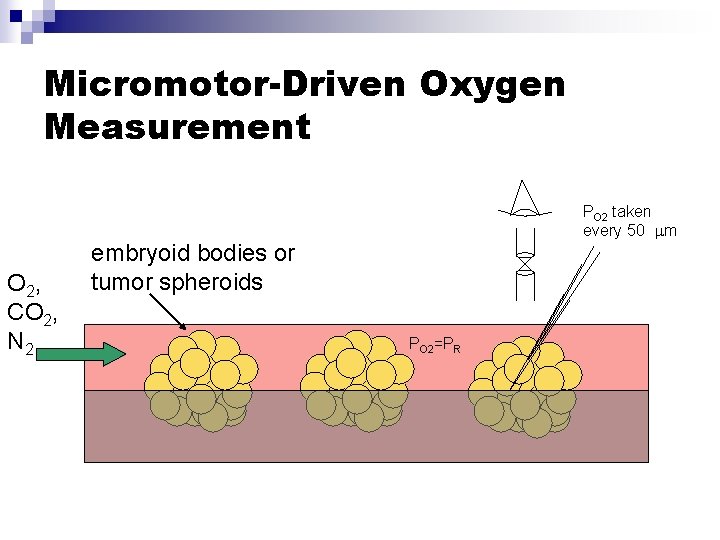
Micromotor-Driven Oxygen Measurement O 2, CO 2, N 2 PO 2 taken every 50 m embryoid bodies or tumor spheroids PO 2=PR
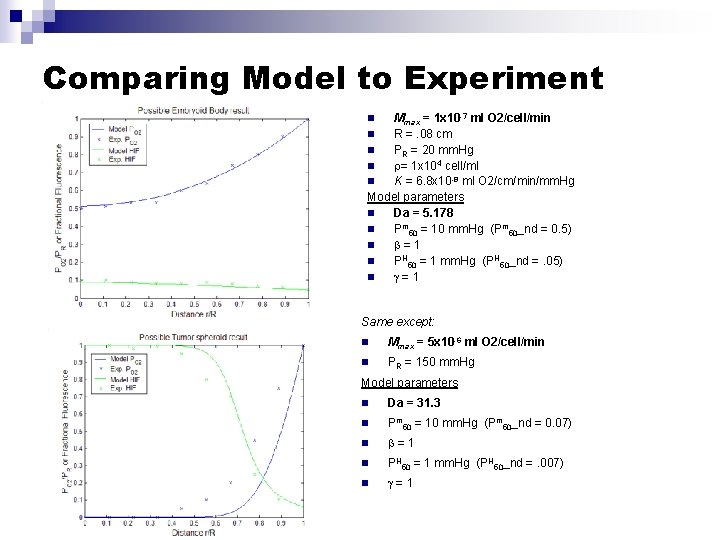
Comparing Model to Experiment Mmax = 1 x 10 -7 ml O 2/cell/min n R =. 08 cm n PR = 20 mm. Hg n = 1 x 104 cell/ml n K = 6. 8 x 10 -8 ml O 2/cm/min/mm. Hg Model parameters n Da = 5. 178 n Pm 50 = 10 mm. Hg (Pm 50_nd = 0. 5) n =1 n PH 50 = 1 mm. Hg (PH 50_nd =. 05) n =1 n Same except: n Mmax = 5 x 10 -6 ml O 2/cell/min n PR = 150 mm. Hg Model parameters n Da = 31. 3 n Pm 50 = 10 mm. Hg (Pm 50_nd = 0. 07) n =1 n PH 50 = 1 mm. Hg (PH 50_nd =. 007) n =1

Analysis of Results n Tested local sensing/consumption regulation of cells and correlation between O 2 presence and HIF 1 persistence in the cell ¨ Correlation between experiment and model validates local sensing and regulation hypothesis ¨ Poor correlation does not disprove local sensing of oxygen but suggest other methods of oxygen regulation

Critique: Did our experimental plan address question 2? ->Is O 2 consumption modified by HIF-1 gene regulation? No n Experimental design does not directly test role of HIF-1 in regulation of oxygen consumption n Changes in oxygen consumption due to local oxygen sensing could be independent of HIF-1 expression n HIF-1 changes due to O 2 could regulate other downstream events (angiogenesis, etc) and not cellular consumption
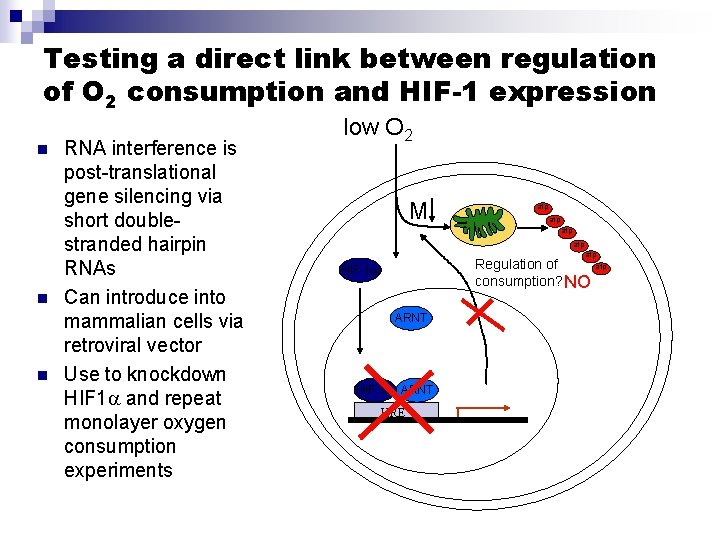
Testing a direct link between regulation of O 2 consumption and HIF-1 expression n RNA interference is post-translational gene silencing via short doublestranded hairpin RNAs Can introduce into mammalian cells via retroviral vector Use to knockdown HIF 1 and repeat monolayer oxygen consumption experiments low O 2 M atp atp atp Regulation of consumption? NO HIF-1 ARNT HIF-1 ARNT HRE
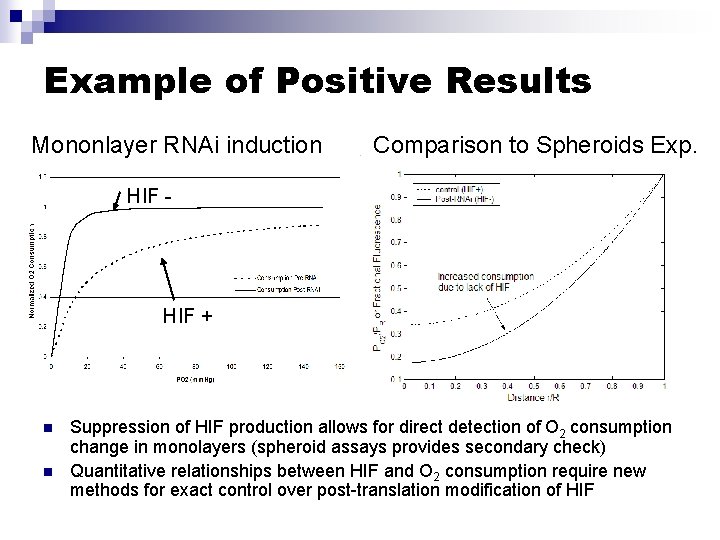
Example of Positive Results Mononlayer RNAi induction Comparison to Spheroids Exp. HIF - HIF + n n Suppression of HIF production allows for direct detection of O 2 consumption change in monolayers (spheroid assays provides secondary check) Quantitative relationships between HIF and O 2 consumption require new methods for exact control over post-translation modification of HIF

Model Critique - continued n Model does not account for possibility of regulation of consumption via cell-cell signaling O 2 diffusion into tissue Signal downregulates outer cell consumption Low O 2 M O 2 diffusible signal M Leads to greater or cell-cell oxygen in the tissue contact to outer cells

Possibilities for further experimentation n Assay downstream glycolytic genes for role in regulation of oxygen consumption by repeating monolayer and spheroid experiments n Test (mine) for soluble factors that may control metabolic rates in early embryonic tissues and/or tumors n Explore “community effect” in regulation of oxygen metabolism

Project summary n n n Developed and implemented model for oxygen diffusion in pre-vascular tissue spheroids with consumption rate dependent on local oxygen concentration Proposed experiments to determine model parameters and validate dependence of oxygen consumption rate on local oxygen concentration Proposed experiments to determine if regulation of oxygen consumption rate is mediated through HIF 1 expression

References Bichet S et al, Oxygen tension modulates -glibn switching in embryoid bodies. FASEB J. 1999 Feb; 13(2): 285 -95 Frasch et al, Early Signals in Cardiac Development. Circ Res. 2002 Sep 20; 91(6): 457 -69 Gassmann, M et al. Oxygen supply and oxygen-dependent gene expression in differentiating embryonic stem cells. 1996 PNAS 93: 2867 -2872 Harris, AL. Hypoxia--A Key Regulatory Factor in Tumour Growth. Nat Rev Cancer 2002 Jan; 2(1): 38 -47 Iyer, NV et al. Cellular and developmental control of O 2 homeostasis by hypoxia-inducible factor 1. Genes Dev. 1998 12: 149 -162 Kotch LE et al, Defective Vascularization of HIF-1 -Null Embryos is Not Associated with VEGF Deficiency but with Mesenchymal Cell Death. Dev Biol. 1999 May 15; 209(2): 254 -67 Krogh, A. The Comparative Physiology of Respiratory Mechanisms. Philadelphia: University of Pennsylvania Press. 1941 Simon et al, Stem Cells. HIF and the Development of Stem Cells of the Cardiovascular System. 2001; 19(4): 279 -86 Ravi R et al, Regulation of tumor angiogenesis by p 53 -induced degradation of hypoxia-inducible factor 1. Genes Dev. 2000 Jan 1; 14(1): 34 -44 Risau et al, Molecular Mechanisms of Vasculogenesis and Embryonic Angiogenesis. J Cell Physiol. 1997 Nov; 173(2): 206 -10 Ryan et al, HIF-1 is required for solid tumor formation and embryonic vascularization. EMBO J. 1998 Jun 1; 17(11): 3005 -15 Wartenberg, M et al, Tumor-induced angiogenesis studied in confrontation cultures of muticellular tumor spheroids and embryoid bodies frown from pluripotent embryonic stem cell. FASEB J 2001 15: 995 -1005 Wolff, M, J Fandrey and W. Jelkmann. Microelectrode measurements of pericellular PO 2 in erythrpoietin-producing human hepatoma cultures. Am J. Physiol 1993 Yamada, T et al, Oxygen Consumption of Mammalian Myocardial Cells in Culture: Measurements in Beating Cells Attached to the Substrate of the Culture Dish, Analytical Biochemistry 1985 145, 302 -307 www. visembryo. com
- Slides: 27