Regulation Molecular Motors I Andy Howard Introductory Biochemistry

Regulation; Molecular Motors I Andy Howard Introductory Biochemistry 7 November 2013 Regulation; Molecular Motors I 11/06/2012

Regulation and motors Regulation happens at several levels n Even though it isn’t really an enzyme, hemoglobin can teach us how allostery in enzymes works. n After that we’ll talk about molecular motors. n 11/06/2012 Regulation; Molecular Motors I P. 2 of 65

Globins & Motor Topics n Regulation n n Availability Allostery PTM Protein-protein interactions Globins n n Oxygen binding Tertiary structure Quarternary structure R and T states n Globins (concluded) n n n Allostery Bohr effect BPG as an effector Sickle-cell anemia Molecular Motors Microtubules n n n Tubulin Structure Cilia & flagella 11/06/2012 Regulation; Molecular Motors I P. 3 of 65

Enzyme availability n n n The enzyme has to be where the reactants are in order for it to act Even a highly proficient enzyme has to have a nonzero concentration How can the cell control [E]tot? n n n Transcription (and translation) Protein processing (degradation) Compartmentalization 11/06/2012 Regulation; Molecular Motors I P. 4 of 65
Transcriptional control n n n m. RNAs have short lifetimes Therefore once a protein is degraded, it will be replaced and available only if new transcriptional activity for that protein occurs Many types of transcriptional effectors n n n Proteins can bind to their own gene Small molecules can bind to gene Promoters can be turned on or off 11/06/2012 Regulation; Molecular Motors I P. 5 of 65

Protein degradation n n All proteins have finite half-lives; Enzymes’ lifetimes often shorter than structural or transport proteins Degraded by slings & arrows of outrageous fortune; or Activity of the proteasome, a molecular machine that tags proteins for degradation and then accomplishes it 11/06/2012 Regulation; Molecular Motors I P. 6 of 65

Compartmentalization n If the enzyme is in one compartment and the substrate in another, it won’t catalyze anything Several mitochondrial catabolic enzyme act on substrates produced in the cytoplasm; these require elaborate transport mechanisms to move them in Therefore, control of the transporters confers control over the enzymatic system 11/06/2012 Regulation; Molecular Motors I P. 7 of 65

Allostery n n Remember we defined this as an effect on protein activity in which binding of a ligand to a protein induces a conformational change that modifies the protein’s activity Ligand may be the same molecule as the substrate or it may be a different one Ligand may bind to the same subunit or a different one These effects happen to non-enzymatic proteins as well as enzymes 11/06/2012 Regulation; Molecular Motors I P. 8 of 65

Substrates as allosteric effectors (homotropic) n n n Standard example: binding of O 2 to one subunit of tetrameric hemoglobin induces conformational change that facilitates binding of 2 nd (& 3 rd & 4 th) O 2’s So the first oxygen is an allosteric effector of the activity in the other subunits Effect can be inhibitory or accelerative 11/06/2012 Regulation; Molecular Motors I P. 9 of 65

Other allosteric effectors (heterotropic) n n n Covalent modification of an enzyme by phosphate or other PTM molecules can turn it on or off Usually catabolic enzymes are stimulated by phosphorylation and anabolic enzymes are turned off, but not always Phosphatases catalyze dephosphorylation; these have the opposite effects 11/06/2012 Regulation; Molecular Motors I P. 10 of 65
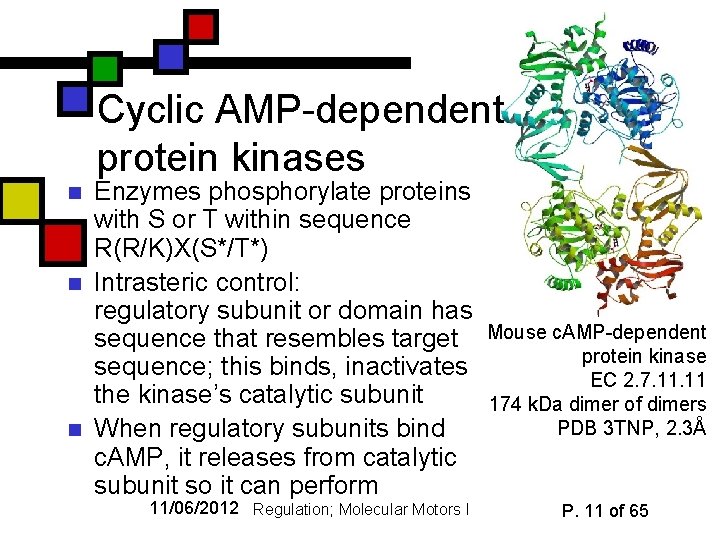
Cyclic AMP-dependent protein kinases n n n Enzymes phosphorylate proteins with S or T within sequence R(R/K)X(S*/T*) Intrasteric control: regulatory subunit or domain has sequence that resembles target sequence; this binds, inactivates the kinase’s catalytic subunit When regulatory subunits bind c. AMP, it releases from catalytic subunit so it can perform 11/06/2012 Regulation; Molecular Motors I Mouse c. AMP-dependent protein kinase EC 2. 7. 11 174 k. Da dimer of dimers PDB 3 TNP, 2. 3Å P. 11 of 65
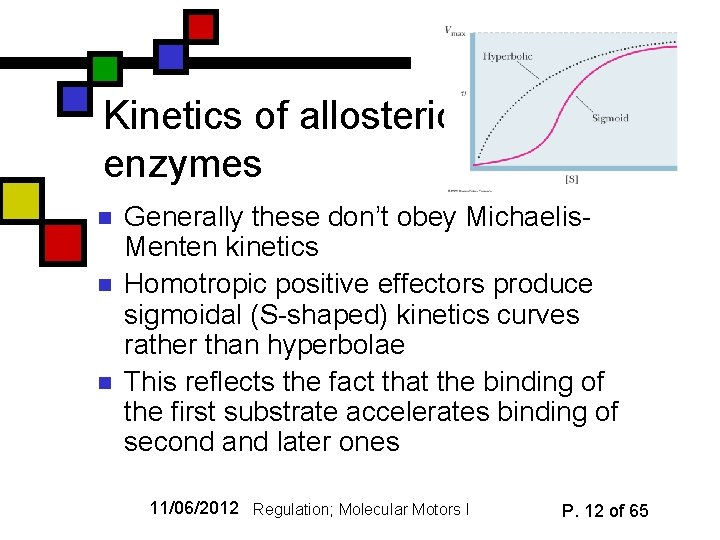
Kinetics of allosteric enzymes n n n Generally these don’t obey Michaelis. Menten kinetics Homotropic positive effectors produce sigmoidal (S-shaped) kinetics curves rather than hyperbolae This reflects the fact that the binding of the first substrate accelerates binding of second and later ones 11/06/2012 Regulation; Molecular Motors I P. 12 of 65

T R State transitions n n Many allosteric effectors influence the equilibrium between two conformations One is typically more rigid and inactive, the other is more flexible and active The rigid one is typically called the “tight” or “T” state; the flexible one is called the “relaxed” or “R” state Allosteric effectors shift the equilibrium toward R or toward T 11/06/2012 Regulation; Molecular Motors I P. 13 of 65
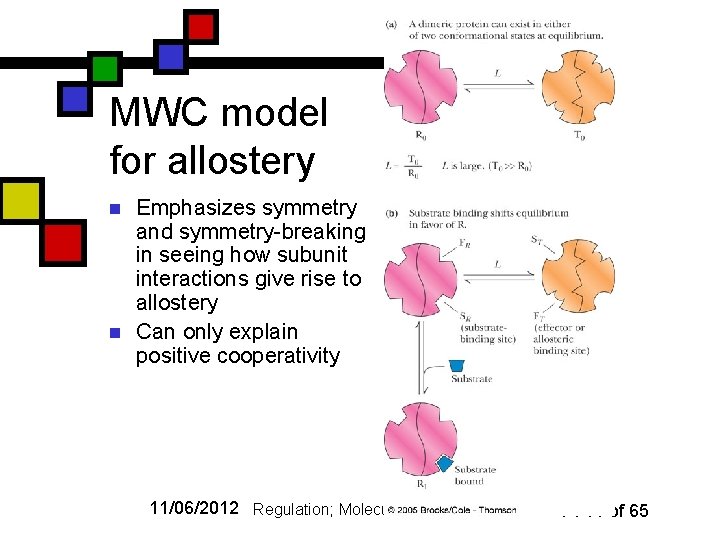
MWC model for allostery n n Emphasizes symmetry and symmetry-breaking in seeing how subunit interactions give rise to allostery Can only explain positive cooperativity 11/06/2012 Regulation; Molecular Motors I P. 14 of 65

Koshland (KNF) model n n Emphasizes conformational changes from one state to another, induced by binding of effector Ligand binding and conformational transitions are distinct steps … so this is a sequential model for allosteric transitions Allows for negative cooperativity as well as positive cooperativity 11/06/2012 Regulation; Molecular Motors I P. 15 of 65

Oligomerization and allostery n n n Often the R T transition is influenced by enzyme oligomerization Tryptophan synthase: dimerization shifts equilibrium toward R Amino acid kinases: hinge motions of monomers engage cooperativity Thermatoga N-acetylglutamate kinase: 194 k. Da hexamer, trimer shown EC 2. 7. 2. 8; PDB 2 BTY, 2. 75¸Å 11/06/2012 Regulation; Molecular Motors I P. 16 of 65
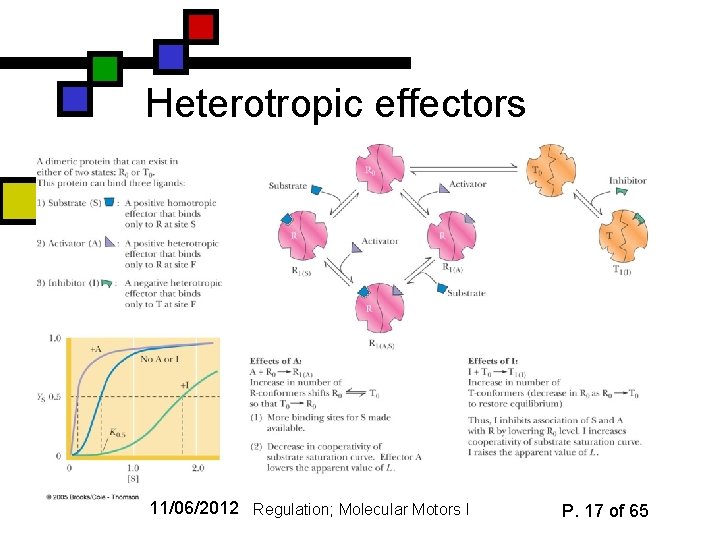
Heterotropic effectors 11/06/2012 Regulation; Molecular Motors I P. 17 of 65

Post-translational modification n n We’ve already looked at phosphorylation Proteolytic cleavage of the enzyme to activate it is another common PTM mode Some proteases cleave themselves (auto -catalysis); in other cases there’s an external protease involved Blood-clotting cascade involves a series of catalytic activations 11/06/2012 Regulation; Molecular Motors I P. 18 of 65

Zymogens n n n As mentioned earlier, this is a term for an inactive form of a protein produced at the ribosome Proteolytic post-translational processing required for the zymogen to be converted to its active form Cleavage may happen intracellularly, during secretion, or extracellularly 11/06/2012 Regulation; Molecular Motors I Bacillus subtilisin + prosequence 35 k. Da heterodimer EC 3. 4. 21. 62 PDB 3 CNQ, 1. 71Å P. 19 of 65
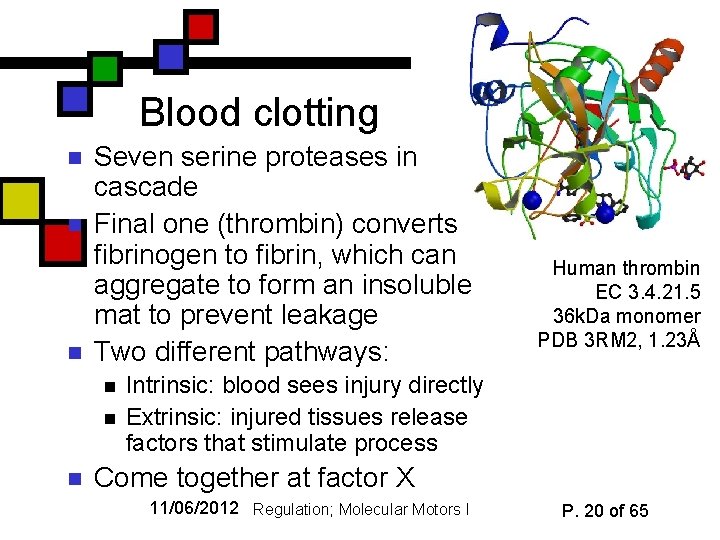
Blood clotting n n n Seven serine proteases in cascade Final one (thrombin) converts fibrinogen to fibrin, which can aggregate to form an insoluble mat to prevent leakage Two different pathways: n n n Human thrombin EC 3. 4. 21. 5 36 k. Da monomer PDB 3 RM 2, 1. 23Å Intrinsic: blood sees injury directly Extrinsic: injured tissues release factors that stimulate process Come together at factor X 11/06/2012 Regulation; Molecular Motors I P. 20 of 65
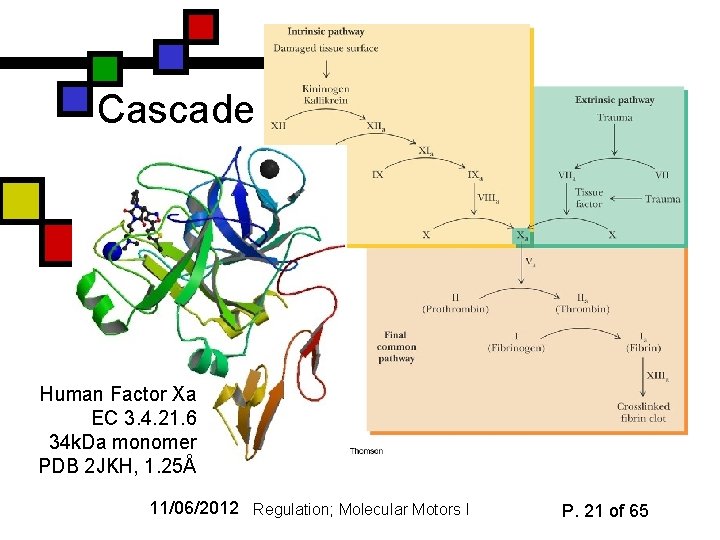
Cascade Human Factor Xa EC 3. 4. 21. 6 34 k. Da monomer PDB 2 JKH, 1. 25Å 11/06/2012 Regulation; Molecular Motors I P. 21 of 65

Protein-protein interactions n n n One major change in biochemistry in the last 25 years is the increasing emphasis on protein-protein interactions in understanding biological activities Many proteins depend on exogenous partners for modulating their activity up or down Example: cholera toxin’s enzymatic component depends on interaction with human protein ARF 6 Vibrio cholerae toxin A 1 subunit + human ARF 6: 37 k. Da heterodimer PDB 2 A 5 D, 1. 8Å 11/06/2012 Regulation; Molecular Motors I P. 22 of 65

Globins as aids to understanding n n Myoglobin and hemoglobin are wellunderstood non-enzymatic proteins whose properties help us understand enzyme regulation Hemoglobin is described as an “honorary enzyme” in that it “catalyzes” the reaction O 2(lung) O 2 (peripheral tissues) 11/06/2012 Regulation; Molecular Motors I P. 23 of 65

Setting the stage for this story n n n Myoglobin is a 16 k. Da monomeric O 2 -storage protein found in peripheral tissues Has Fe-containing prosthetic group called heme; iron must be in Fe 2+ state to bind O 2 It yields up dioxygen to various oxygenrequiring processes, particularly oxidative phosphorylation in mitochondria in rapidly metabolizing tissues 11/06/2012 Regulation; Molecular Motors I P. 24 of 65

Why is myoglobin needed? n Free heme will bind O 2 nicely; why not just rely on that? n Protein has 3 functions: Immobilizes the heme group n Discourages oxidation of Fe 2+ to Fe 3+ n Provides a pocket that oxygen can fit into n 11/06/2012 Regulation; Molecular Motors I P. 25 of 65

Setting the stage II n n n Hemoglobin (in vertebrates, at least) is a tetrameric, 64 k. Da transport protein that carries oxygen from the lungs to peripheral tissues It also transports acidic CO 2 the opposite direction Its allosteric properties are what we’ll discuss 11/06/2012 Regulation; Molecular Motors I P. 26 of 65

n Structure determinations Myoglobin & hemoglobin were the first 2 proteins to have their 3 -D structures determined experimentally n n Photo courtesy EMBL Myoglobin: Kendrew, 1958 Hemoglobin: Perutz, 1958 Most of the experimental tools that crystallographers rely on were developed for these structure determinations Nobel prizes for both, 1965 (small T!) 11/06/2012 Regulation; Molecular Motors I Photo courtesy Oregon State Library P. 27 of 65
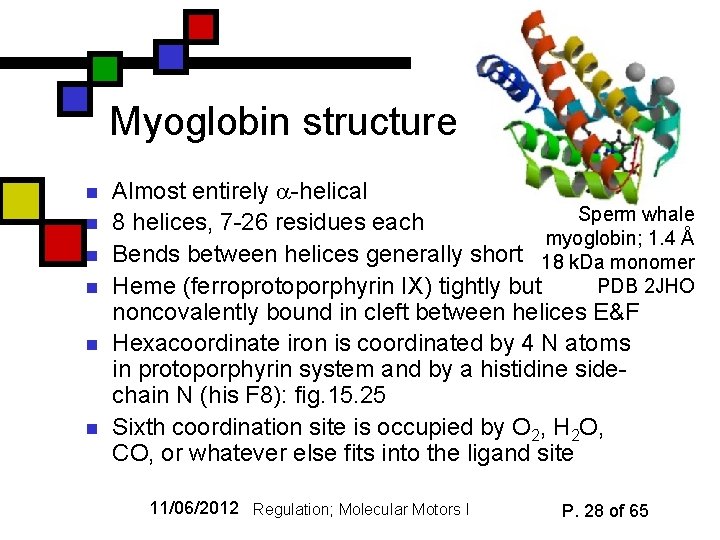
Myoglobin structure n n n Almost entirely -helical Sperm whale 8 helices, 7 -26 residues each myoglobin; 1. 4 Å Bends between helices generally short 18 k. Da monomer PDB 2 JHO Heme (ferroprotoporphyrin IX) tightly but noncovalently bound in cleft between helices E&F Hexacoordinate iron is coordinated by 4 N atoms in protoporphyrin system and by a histidine sidechain N (his F 8): fig. 15. 25 Sixth coordination site is occupied by O 2, H 2 O, CO, or whatever else fits into the ligand site 11/06/2012 Regulation; Molecular Motors I P. 28 of 65

O 2 binding alters myoglobin structure a little n n n Deoxymyoglobin: Fe 2+ is 0. 55Å out of the heme plane, toward his F 8, away from O 2 binding site Oxymyoglobin: moves toward heme plane—now only 0. 26Å away (fig. 15. 26) This difference doesn’t matter much here, but it’ll matter a lot more in hemoglobin! 11/06/2012 Regulation; Molecular Motors I P. 29 of 65
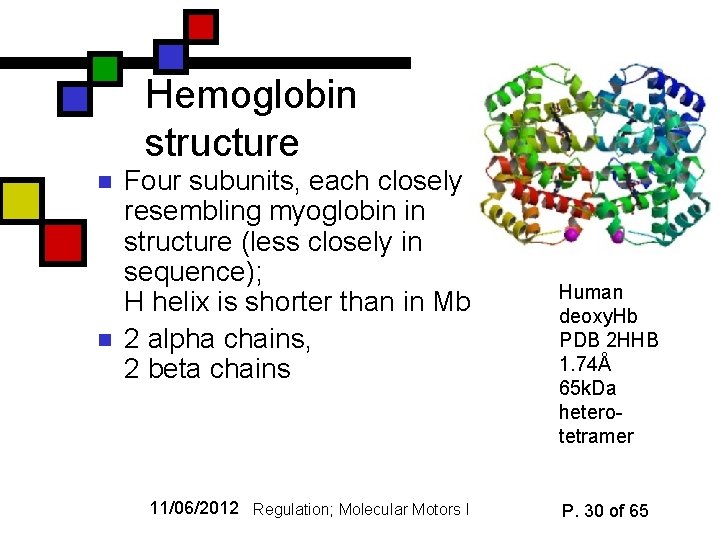
Hemoglobin structure n n Four subunits, each closely resembling myoglobin in structure (less closely in sequence); H helix is shorter than in Mb 2 alpha chains, 2 beta chains 11/06/2012 Regulation; Molecular Motors I Human deoxy. Hb PDB 2 HHB 1. 74Å 65 k. Da heterotetramer P. 30 of 65
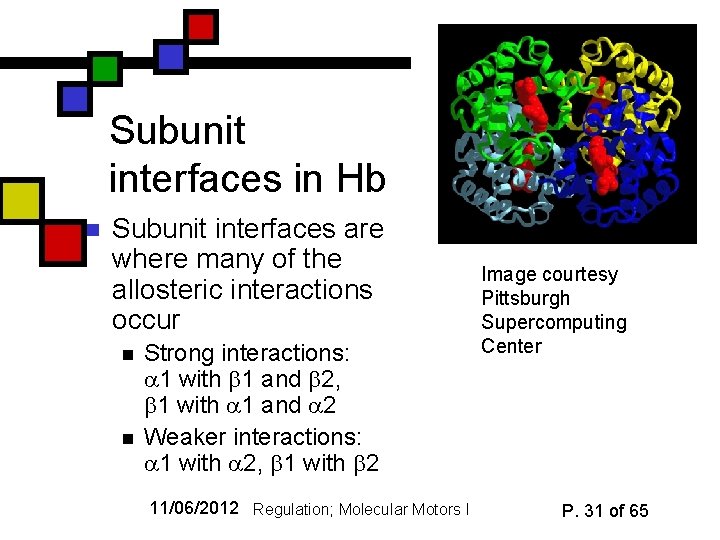
Subunit interfaces in Hb n Subunit interfaces are where many of the allosteric interactions occur n n Strong interactions: 1 with 1 and 2, 1 with 1 and 2 Weaker interactions: 1 with 2, 1 with 2 11/06/2012 Regulation; Molecular Motors I Image courtesy Pittsburgh Supercomputing Center P. 31 of 65
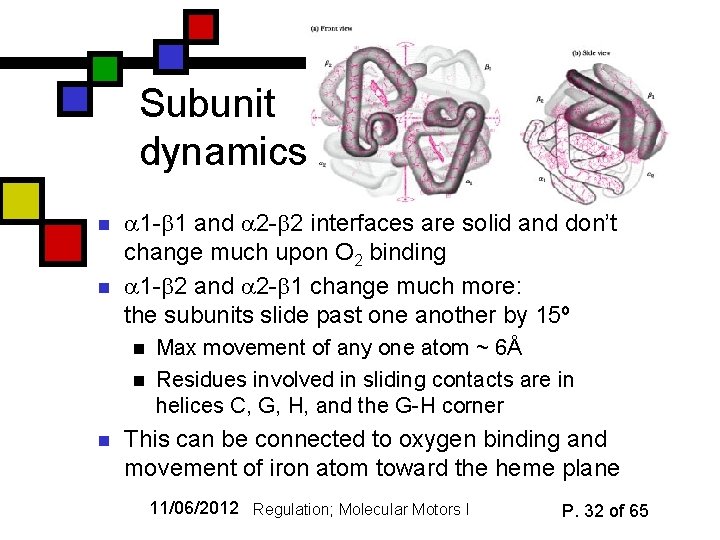
Subunit dynamics n n 1 - 1 and 2 - 2 interfaces are solid and don’t change much upon O 2 binding 1 - 2 and 2 - 1 change much more: the subunits slide past one another by 15º n n n Max movement of any one atom ~ 6Å Residues involved in sliding contacts are in helices C, G, H, and the G-H corner This can be connected to oxygen binding and movement of iron atom toward the heme plane 11/06/2012 Regulation; Molecular Motors I P. 32 of 65

Conformational states n n We can describe this shift as a transition from one conformational state to another The stablest form for deoxy. Hb is described as a “tense” or T state n n Heme environment of beta chains is almost inaccessible because of steric hindrance That makes O 2 binding difficult to achieve The stablest form for oxy. HB is described as a “relaxed” or R state Accessibility of beta chains substantially enhanced 11/06/2012 Regulation; Molecular Motors I P. 33 of 65
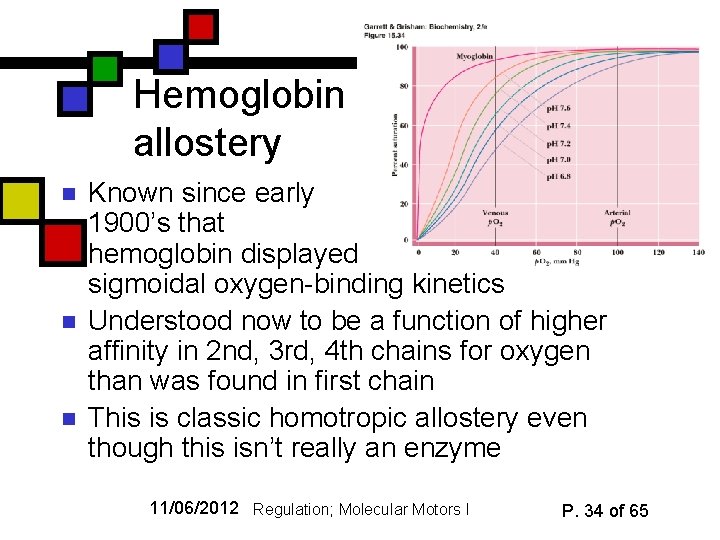
Hemoglobin allostery n n n Known since early 1900’s that hemoglobin displayed sigmoidal oxygen-binding kinetics Understood now to be a function of higher affinity in 2 nd, 3 rd, 4 th chains for oxygen than was found in first chain This is classic homotropic allostery even though this isn’t really an enzyme 11/06/2012 Regulation; Molecular Motors I P. 34 of 65

R T states and hemoglobin n We visualize each Hb monomer as existing in either T (tight) or R (relaxed) states; T binds oxygen reluctantly, R binds it enthusiastically Deoxy. Hb is stablest in T state Binding of first Hb stabilizes R state in the other subunits, so their affinity is higher 11/06/2012 Regulation; Molecular Motors I P. 35 of 65

Binding and p. O 2 n n Hill found that binding could be modeled by a polynomial fit to p. O 2 Sir Archibald V. Hill Kinetics worked out in 1910’s: didn’t require protein purification, photo courtesy nobelprize. org just careful in vitro measurements of blood extracts 11/06/2012 Regulation; Molecular Motors I P. 36 of 65

Hill coefficients n n n Actual equation is on next page Relevant parameters to determine are P 50, the oxygen partial pressure at which half the O 2 -binding sites are filled, and n, a unitless value characterizing the cooperativity n is called the Hill coefficient. 11/06/2012 Regulation; Molecular Motors I P. 37 of 65

p. O 2 and fraction oxygenated n n n If Y is fraction of globin that is oxygenated and p. O 2 is the partial pressure of oxygen, then Y/(1 -Y) = (p. O 2 /P 50)n 4 th-edition formulation: P 50 n K so Y/(1 -Y) = p. O 2 n / K P 50 is a parameter corresponding to halfoccupied hemoglobin (Y=1/2) n n n work out the algebra: When p. O 2 = P 50, Y/(1 -Y) = 1 n=1 so Y = 1/2. Note that the equation on p. 496 of the enhanced 3 rd edition is wrong! 11/06/2012 Regulation; Molecular Motors I P. 38 of 65

Real Hill parameters (p. 503) n Human hemoglobin has n ~ 2. 8, P 50 ~ 26 Torr n n n Perfect cooperativity, tetrameric protein: n =4 No cooperativity at all would be n = 1. Lung p. O 2 ~ 100 Torr; peripheral tissue 10 -40 Torr So lung has Y~0. 98, periphery has Y~0. 06! That’s a big enough difference to be functional If n=1, Ylung=0. 79, Ytissue=0. 28; not nearly as good a delivery vehicle! 11/06/2012 Regulation; Molecular Motors I P. 39 of 65

MWC theory n n n Monod, Wyman, Changeux developed mathematical model describing T R transitions and applied it to Hb Accounts reasonably well for sigmoidal kinetics and Hill coefficient values Key assumption: ligand binds only to R state, so when it binds, it depletes R in the T R equilibrium, so that tends to make more R 11/06/2012 Regulation; Molecular Motors I Jacques Monod Photo Courtesy Nobelprize. org P. 40 of 65

Koshland’s contribution n n Conformational changes between the two states are also clearly relevant to the discussion His papers from the 1970’s articulating the algebra of hemoglobin-binding kinetics are amazingly intricate 11/06/2012 Regulation; Molecular Motors I Dan Koshland Photo Courtesy U. of California P. 41 of 65

Added complication I: p. H n n n Oxygen affinity is p. H dependent That’s typical of proteins, especially those in which histidine is involved in the activity (remember it readily Christian Bohr undergoes protonation and photo courtesy deprotonation near neutral p. H) Wikipedia Bohr effect (also discovered in early 1900’s): lower affinity at low p. H (fig. 15. 33) 11/06/2012 Regulation; Molecular Motors I P. 42 of 65

How the Bohr effect happens n n R form has an effective p. Ka that is lower than T One reason: n n In the T state, his 146 is close to asp 94. That allows the histidine p. Ka to be higher In R state, his 146 is farther from asp 94 so its p. Ka is lower. 11/06/2012 Regulation; Molecular Motors I Cartoon courtesy Jon Robertus, UT Austin P. 43 of 65
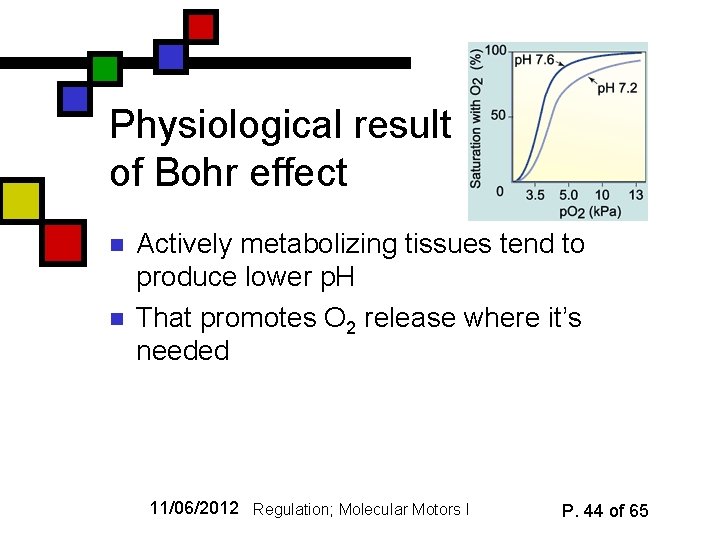
Physiological result of Bohr effect n n Actively metabolizing tissues tend to produce lower p. H That promotes O 2 release where it’s needed 11/06/2012 Regulation; Molecular Motors I P. 44 of 65
![CO 2 also promotes dissociation n High [CO 2] lowers p. H because it CO 2 also promotes dissociation n High [CO 2] lowers p. H because it](http://slidetodoc.com/presentation_image_h2/f3d9cc0f64c448469b8ffdc3faa4e73e/image-45.jpg)
CO 2 also promotes dissociation n High [CO 2] lowers p. H because it dissolves with the help of the enzyme carbonic anhydrase and dissociates: H 2 O + CO 2 H 2 CO 3 H+ + HCO 3 Bicarbonate transported back to lungs When Hb gets re-oxygenated, bicarbonate gets converted back to gaseous CO 2 and exhaled 11/06/2012 Regulation; Molecular Motors I P. 45 of 65
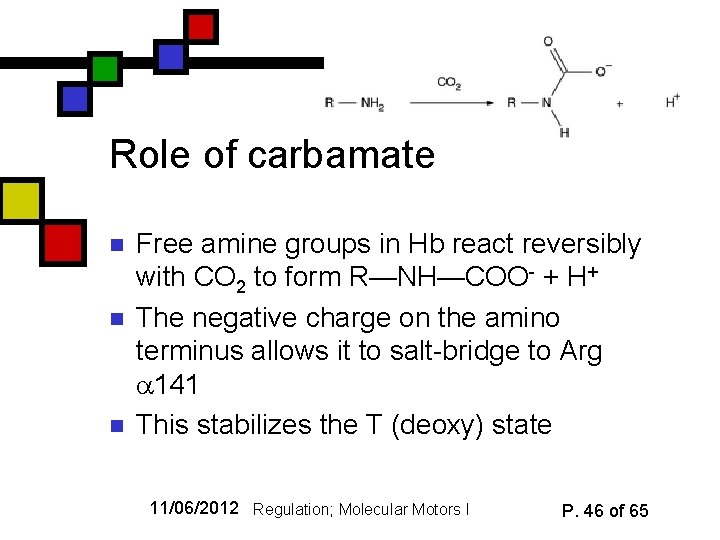
Role of carbamate n n n Free amine groups in Hb react reversibly with CO 2 to form R—NH—COO- + H+ The negative charge on the amino terminus allows it to salt-bridge to Arg 141 This stabilizes the T (deoxy) state 11/06/2012 Regulation; Molecular Motors I P. 46 of 65
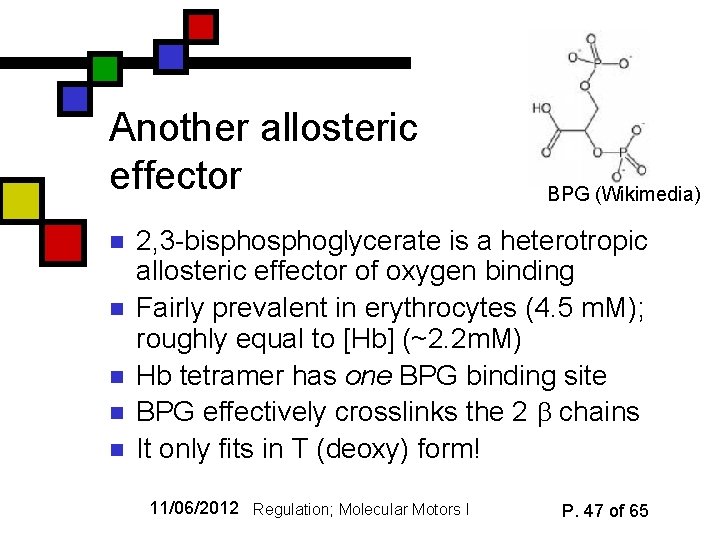
Another allosteric effector n n n BPG (Wikimedia) 2, 3 -bisphoglycerate is a heterotropic allosteric effector of oxygen binding Fairly prevalent in erythrocytes (4. 5 m. M); roughly equal to [Hb] (~2. 2 m. M) Hb tetramer has one BPG binding site BPG effectively crosslinks the 2 chains It only fits in T (deoxy) form! 11/06/2012 Regulation; Molecular Motors I P. 47 of 65

BPG and physiology n n n p. O 2 is too high (40 Torr) for efficient release of O 2 in many cells in absence of BPG With BPG around, T-state is stabilized enough to facilitate O 2 release Big animals (e. g. sheep) have lower O 2 affinity but their Hb is less influenced by BPG 11/06/2012 Regulation; Molecular Motors I P. 48 of 65

Fetal hemoglobin n n Higher oxygen affinity because the type of hemoglobin found there has a lower affinity for BPG Fetal Hb is 2 2; doesn’t bind BPG as much as . Fetal Hb has more pronounced Bohr effect That helps ensure that plenty of O 2 gets from mother to fetus across the placenta Human fetal hemoglobin 65 k. Da heterotetramer 2. 5Å 11/06/2012 Regulation; Molecular Motors I P. 49 of 65

Sickle-cell anemia n n Genetic disorder: Hb residue 6 mutated from glu to val. This variant is called Hb. S. Results in intermolecular interaction between neighboring Hb tetramers that can cause chainlike polymerization Polymerized hemoglobin will partially fall out of solution and tug on the erythrocyte structure, resulting in misshapen (sickle-shaped) cells Oxygen affinity is lower because of insolubility 11/06/2012 Regulation; Molecular Motors I P. 50 of 65
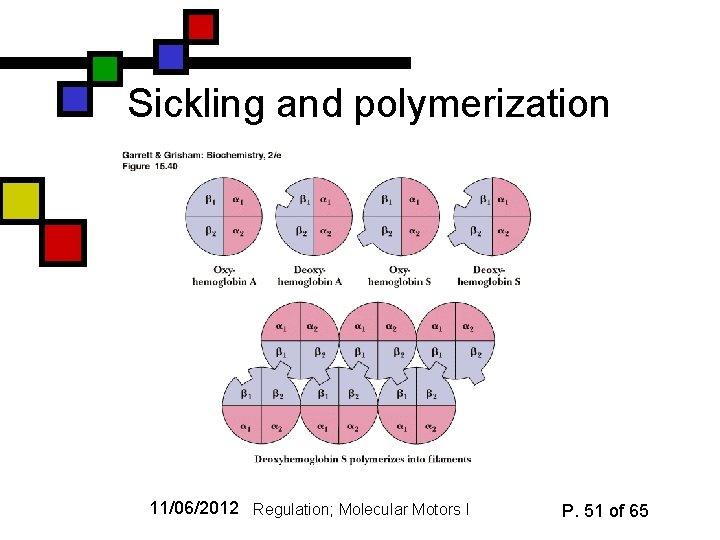
Sickling and polymerization 11/06/2012 Regulation; Molecular Motors I P. 51 of 65

Why has this mutation survived? n n n Homozygotes don’t generally Deoxy Hb. S survive to produce progeny; 2. 05 Å but heterozygotes do PDB 2 HBS Heterozygotes do have modestly reduced oxygen-carrying capacity in their blood because some erythrocytes are sickled BUT heterozygotes are somewhat resistant to malaria, so the gene survives in tropical places where malaria is a severe problem 11/06/2012 Regulation; Molecular Motors I P. 52 of 65

How is sickling related to malaria? n n n Malaria parasite (Plasmodium spp. ) infects erythrocytes They’re unable to infect sickled cells So a partially affected cell might survive the infection better than a nonsickled cell Still some argument about all of this Note that most tropical environments have plenty of oxygen around (not a lot of malaria at 2000 meters elevation) 11/06/2012 Regulation; Molecular Motors I Plasmodium falciparum from A. Dove (2001) Nature Medicine 7: 389 P. 53 of 65

Other hemoglobin mutants n n n Because it’s easy to get human blood, dozens of hemoglobin mutants have been characterized Many are asymptomatic Some have moderate to severe effects on oxygen carrying capacity or erythrocyte physiology 11/06/2012 Regulation; Molecular Motors I P. 54 of 65

What is a molecular motor? A protein-based system that interconverts chemical energy and mechanical work n We’ll discuss several molecular motors today, and then Tuesday we’ll look at what might be the most important one: the vertebrate muscle. n 11/06/2012 Regulation; Molecular Motors I P. 55 of 65

Microtubules (G&G § 16. 3) n 30 -nm structures composed of repeating units of a heterodimeric protein, tubulin n -tubulin: 55 k. Da also Structure of microtubule itself: polymer in which the heterodimers wrap around in a staggered way to produce a tube 11/06/2012 Regulation; Molecular Motors I P. 56 of 65
Tubulin structure n n n and are similar but not identical Structure determined by electron diffraction, not X-ray diffraction Some NMR structures available too Two GTP binding sites per monomer Heterodimer is stable if Ca 2+ present 11/06/2012 Regulation; Molecular Motors I P. 57 of 65

i. Clicker quiz question 1 n Why might you expect crystallization of tubulin to be difficult? n n n (a) It is too big to crystallize (b) It is too small to crystallize (c) Proteins that naturally form complex but non-crystalline 3 -D structures are resistant to crystallization (d) It is membrane-bound (e) none of the above 11/06/2012 Regulation; Molecular Motors I P. 58 of 65
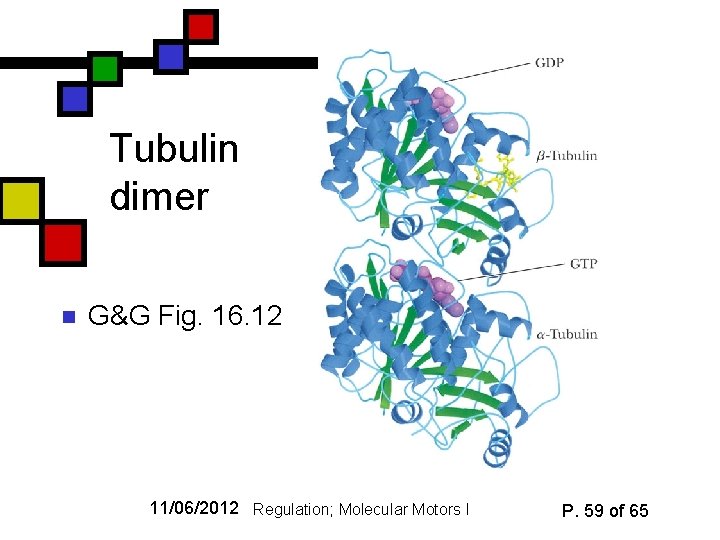
Tubulin dimer n G&G Fig. 16. 12 11/06/2012 Regulation; Molecular Motors I P. 59 of 65
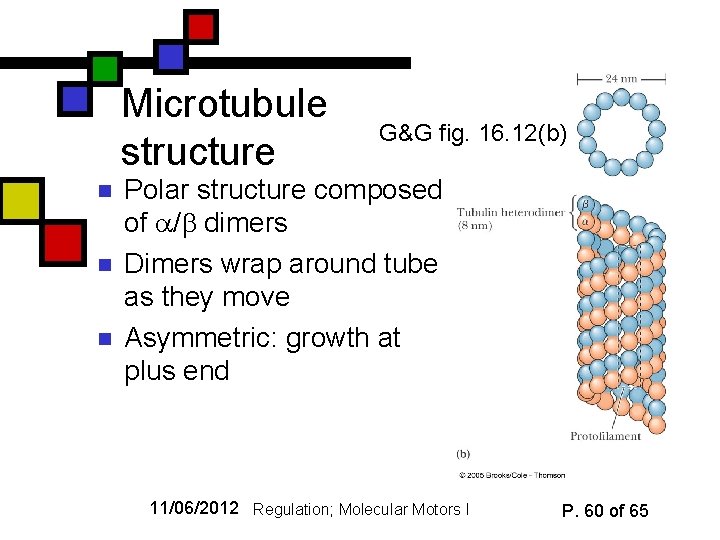
Microtubule structure n n n G&G fig. 16. 12(b) Polar structure composed of / dimers Dimers wrap around tube as they move Asymmetric: growth at plus end 11/06/2012 Regulation; Molecular Motors I P. 60 of 65
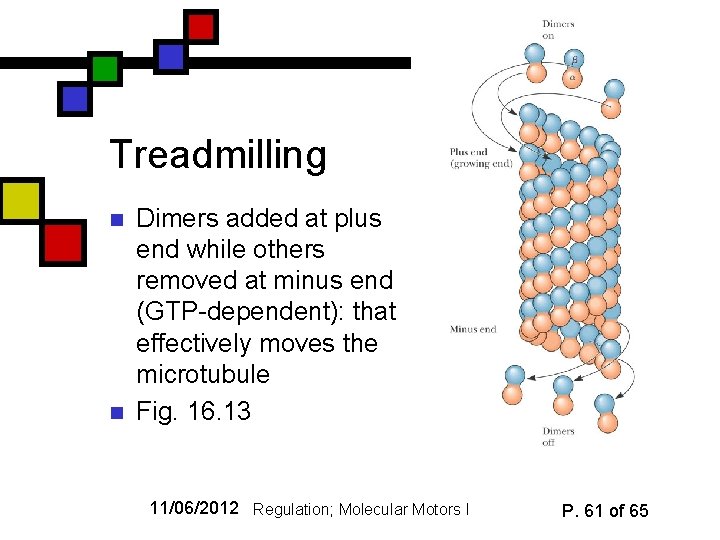
Treadmilling n n Dimers added at plus end while others removed at minus end (GTP-dependent): that effectively moves the microtubule Fig. 16. 13 11/06/2012 Regulation; Molecular Motors I P. 61 of 65

Role in cytoskeleton n Microtubules have a role apart from their role in molecular motor operations: They are responsible for much of the rigidity of the cytoskeleton Cytoskeleton contains: n n n Microtubules (made from tubulin) Intermediate fibers (7 -12 nm; made from keratins and other proteins) Microfilaments (8 nm diameter: made from actin) 11/06/2012 Regulation; Molecular Motors I P. 62 of 65
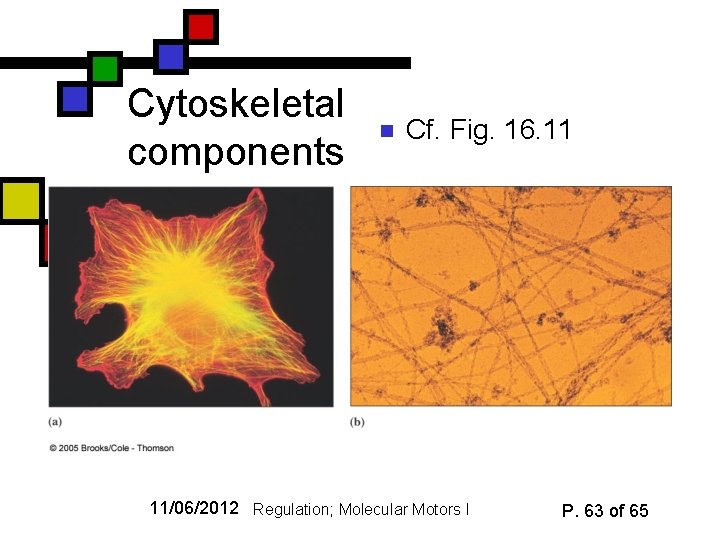
Cytoskeletal components n Cf. Fig. 16. 11 11/06/2012 Regulation; Molecular Motors I P. 63 of 65

Cilia and flagella n n Both are microtubule-based structures used in movement Cilia: n n n short, hairlike projections, found on many animal and lower-plant cells beating motion moves cells or helps move extracellular fluid over surface Flagella n n Longer, found singly or a few at a time Propel cells through fluids 11/06/2012 Regulation; Molecular Motors I P. 64 of 65

Axonemes n Bundle of microtubule fibers: n n n Two central microtubules Nine pairs of joined microtubules Often described as a 9+2 arrangement Surrounded by plasma membrane that connects to the cell’s PM If we remove the PM and add a lot of salt, the axoneme will release a protein called dynein 11/06/2012 Regulation; Molecular Motors I P. 65 of 65
- Slides: 65