Regulating Nanomaterials under CEPA CIELAP Workshop February 22

Regulating Nanomaterials under CEPA CIELAP Workshop February 22, 2008 Bernard Madé, New Substances Program Environment Canada

Objective of the Presentation • To provide information on current thinking on how to address nanomaterials under CEPA 1999 • Overview – Background – Current views in the following areas: • Regulatory Aspects • Alignment with Other Countries • Research and Other Scientific/Technical Activities – Conclusions 2

Background • Nanomaterials are being developed and are entering the Canadian market – Industry Canada indicates about 80 Canadian companies advertise about 150 products containing 88 distinct nanomaterials – Some nanomaterials have been on the market for a long time • There is a concern that certain nanomaterials could have negative impacts on human health or the environment as a result of their new properties • As part of their responsibilities under CEPA, EC and HC need to ensure human health and environmental concerns are addressed appropriately – Need to explore concerns – Need to determine if some nanomaterials present risks – Need to address any significant risk (within CEPA jurisdiction) 3

Substances Assessment and Management under CEPA • CEPA 1999, Part 5: Controlling Toxic Substances – Assessments conducted jointly by Environment Canada (EC) and Health Canada (HC) to determine if a substance is toxic or capable of becoming toxic according to CEPA – Toxic is defined by whether a substance constitutes or may pause a risk to: • The environment or biodiversity • The environment upon which life depends • Human life or health – Risk Management: • If there is a risk, how can risks be reduced or eliminated? • Two programs: new and existing substances – A “new” substance under CEPA is a substance not appearing on the Domestic Substances List (DSL). – All new substances must be notified to EC before the are commercialized in Canada under the New Substances Notification Regulations (NSNR) 4

Some nanomaterials will be considered “new” and others will be considered “existing” under CEPA • Nanomaterials with unique structures and not listed on the DSL are considered “new” – New Substances regime apply • Nanomaterials that have the same structure as substances listed on the DSL are considered “existing” – Currently no special notification or assessment scheme for these substances 5

What have we done so far? • In September 2007, EC and HC held a multistakeholder consultation on a proposed regulatory framework – Overall, stakeholders are supportive of efforts – Interest is high and they want to be engaged in developing the specifics • Our objective: Protect human health and the environment and enhance public confidence by enabling the responsible introduction and use of nanotechnology through a program that scientifically assesses and appropriately manages the potential risks. 6

Regulatory Challenges • Ideally, all nanomaterials which exhibit new properties should be assessed as new substances, However, at the moment it is not the case • There are important knowledge gaps on properties, fate, and effects – i. e. , we may not know all we need to know to properly assess and manage risks • Current NSNR were not designed for nanomaterials – Issues with notification triggers, information requirements • Information on nanomaterials currently on the market is incomplete • There is a need to further inform manufacturers and importers about current and future requirements 7

Proposed Approach: Regulatory Aspects • Phase 1 (Short term): – Further inform companies of their obligations under CEPA – Gather information on materials already in commerce through a CEPA, section 71 survey and a voluntary challenge – Consider whether changes to CEPA (and NSNR) are needed • Objective: ensure assessment of nanomaterials of interest – Use Significant New Activity (SNAc) provisions for new nanomaterials, where applicable 8

Information Gathering • There is a need to gather information on all current activities involving nanomaterials to inform the implementation of a regulatory framework: – Which nanomaterials are currently being developed or are on the market; – In which quantities; – For what applications; – Have there been any toxicological studies done 9
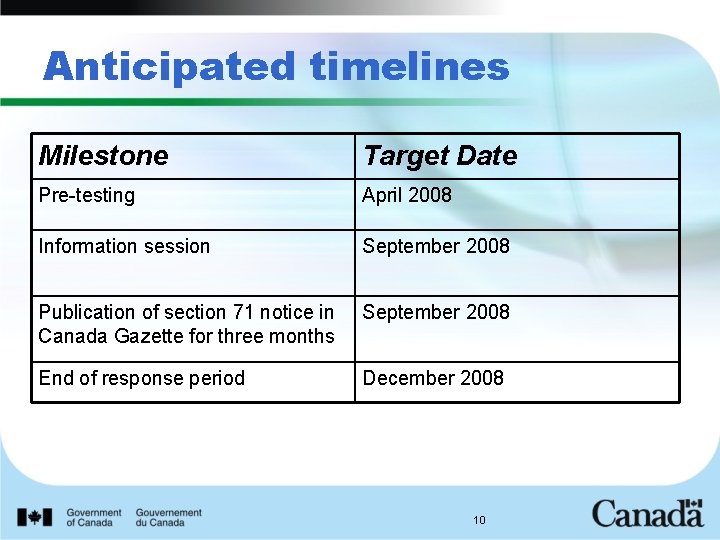
Anticipated timelines Milestone Target Date Pre-testing April 2008 Information session September 2008 Publication of section 71 notice in Canada Gazette for three months September 2008 End of response period December 2008 10

Proposed Approach: Regulatory Aspects (2) • Phase 2 (Longer term) – Consider more elaborate changes to NSNR to ensure information requirements are sufficient to assess and manage nanomaterials – Consider using SNAc provisions for existing nanomaterials 11

Alignment with Other Countries 12

Current Status • ISO has initiated work to develop standards for nanomaterials, including in terminology and nomenclature – A new nomenclature is likely necessary to provide a unique identifier for each nanomaterial; an essential requirement for the appropriate regulation of these substances. – EC is chairing the Technical Committee sub-group on nomenclature • Canada and the US are very active participants to the OECD Working Party on Manufactured Nanomaterials – This Working Party is developing a research strategy and testing program to address the substantial science gaps associated with nanomaterials • High level of cooperation between EC, HC, and the US EPA 13

Proposed Approach: Alignment with Other Countries • Support effort of the Canadian Standards Association (CSA) in the development of standard nomenclature and terminology through the ISO (2008 -2010) • Support the efforts of the OECD with regard to research that will be done on nanomaterials identified as priorities (2008 -2012) • Continue collaboration with US EPA 14

Research and Other Scientific/Technical Activities 15

Ecological Risk Assessment Needs Need to develop an understanding of: – properties and effects of NMs in the environment – transformation characteristics of NMs in the environment. – how does the size, shape and unique characteristics of these materials effect bioavailability. – the vector aspect of the nanomaterials wrt how NMs can increase the toxicity of other organisms. – the relationship between acute and chronic toxicity to aquatic organisms – the relationship between octanol water partition coefficient and bioaccumulation for nanomaterials. – baseline environmental levels and how to measure levels in the environment. 16

Proposed Approach: Research and Other Scientific/Technical Activities • Build up EC research capacity for NMs • Involve research funding agencies • Develop an EC research strategy which will contribute to the OECD testing program – Needs are beyond the scope of any one country’s research capacity. Partnering with others is essential • Consider compliance verification and monitoring needs related to NMs 17

Conclusions • Concerns with regard to nanomaterials must be addressed • This will not be done overnight: some important information needs to be developed in the coming years to help us regulate these new materials • EC and HC are taking steps to address concerns and assess nanomaterials 18

Questions? 19

Supplemental Slides 20

OECD Working Party on Manufactured Nanomaterials Eight Steering Groups working on specific issues: SG 1: SG 2: SG 3: SG 4: SG 5: SG 6: SG 7: SG 8: OECD database on EHS Research strategies on Manufactured Nanomaterials Safety testing of a representative set of Manufactured Nanomaterials and Test Guidelines Co-operation on Voluntary Schemes and Regulatory Programs Co-operation on Risk Assessment Role of Alternative Methods in Nano Toxicology Exposure Measurement and Exposure Mitigation 21

OECD representative nanomaterials • • Fullerenes (C 60) SWCNTs MWCNTs Silver nanoparticles Iron nanoparticles Carbon black Titanium dioxide • • Aluminum dioxide Cerium oxide Zinc oxide Silicon dioxide Polystyrene Dendrimers Nanoclays 22
- Slides: 22