Registration of Veterinary Medicinal Products by ONSSA in

Registration of Veterinary Medicinal Products by ONSSA in Kingdom of Morocco Bruxelles 24– 25 juin 2014 Dr. GRINI Ahmed

P L A N Kingdom of Morocco. The Veterinary Pharmaceutical Sector in Morocco. Regulatory bases related to veterinary medicine products. Integration of the medicinal products in the veterinary control. Division of Pharmacy and Veterinary Inputs (DPIV). Dual regulatory authorities: Agriculture and Health. Organization’s chart of the Direction of Veterinary Services (DSV). Missions and Human resources of the DPIV. Control strategy : Value chain approach. Registration of Veterinary Pharmaceutical Establishments. Registration of Veterinary Medicinal Products. § Integration of the VICH requirement in the registration process. § VICH Guidelines already used in Morocco.
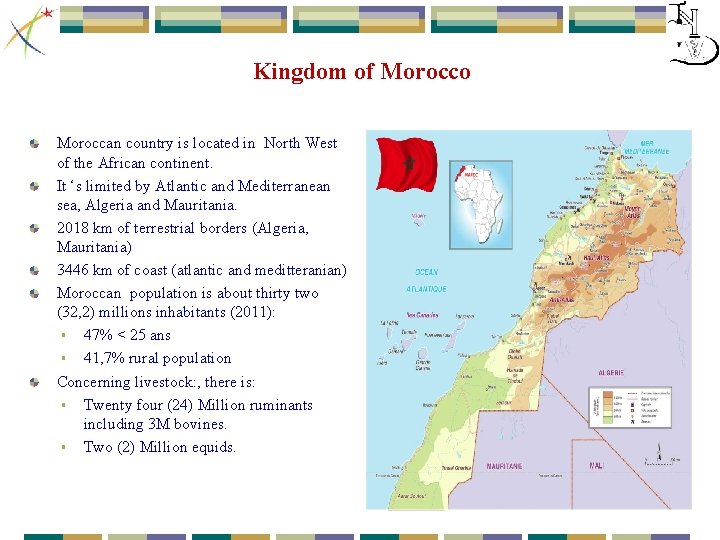
Kingdom of Morocco Moroccan country is located in North West of the African continent. It ‘s limited by Atlantic and Mediterranean sea, Algeria and Mauritania. 2018 km of terrestrial borders (Algeria, Mauritania) 3446 km of coast (atlantic and meditteranian) Moroccan population is about thirty two (32, 2) millions inhabitants (2011): § 47% < 25 ans § 41, 7% rural population Concerning livestock: , there is: § Twenty four (24) Million ruminants including 3 M bovines. § Two (2) Million equids.
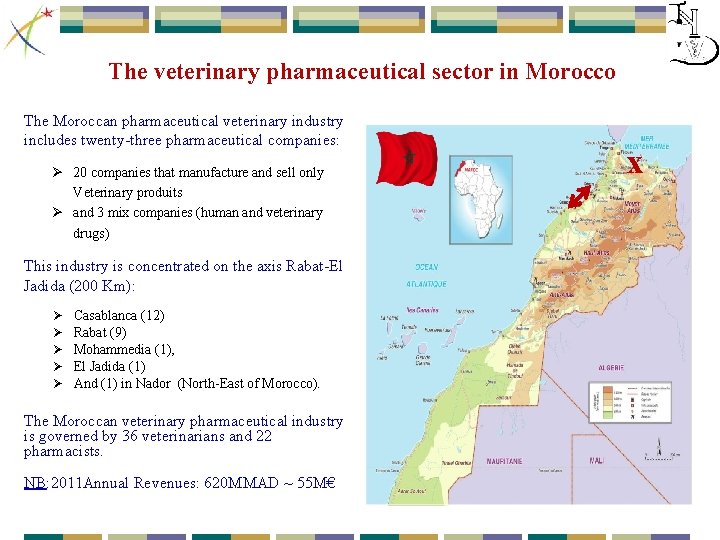
The veterinary pharmaceutical sector in Morocco The Moroccan pharmaceutical veterinary industry includes twenty-three pharmaceutical companies: Ø 20 companies that manufacture and sell only Veterinary produits Ø and 3 mix companies (human and veterinary drugs) This industry is concentrated on the axis Rabat-El Jadida (200 Km): Ø Ø Ø Casablanca (12) Rabat (9) Mohammedia (1), El Jadida (1) And (1) in Nador (North-East of Morocco). The Moroccan veterinary pharmaceutical industry is governed by 36 veterinarians and 22 pharmacists. NB: 2011 Annual Revenues: 620 MMAD ~ 55 M€ x

Regulatory bases related to veterinary medicine products q The Law 21 -80 on private practice as veterinarian, surgery and veterinary pharmacy. q The application Decree of the Law 21 -80. q The Joint Circular (Agriculture and Health ministries) on marketing authorization procedures for veterinary drugs. q The Joint Circular on Inspection of Pharmaceuticals veterinary plants according to GMP and Good Distribution Practice standards. q Circular Note concerning the Veterinary Pharmacovigilance.

Dual regulatory authorities: Agriculture and Health q The registration of veterinary medicinal products in Morocco is subject to dual regulatory authorities: the Ministry of Agriculture and the Ministry of Health. q The Ministry of Agriculture plays a key-role in the process of registration of veterinary drugs.

Integrated of veterinary medicinal products control q q In Morocco, integration of veterinary medicinal products control in the global animal health and food hygiene control aims to ensure: q Health and productivity of livestock; q Consumer safety; q Preservation of the environment; The Division of Pharmacy and Veterinary Inputs (DPIV/ONSSA) was created to ensure the quality of veterinary medicinal products (efficacy, safety) for imported and locally manufactured products.
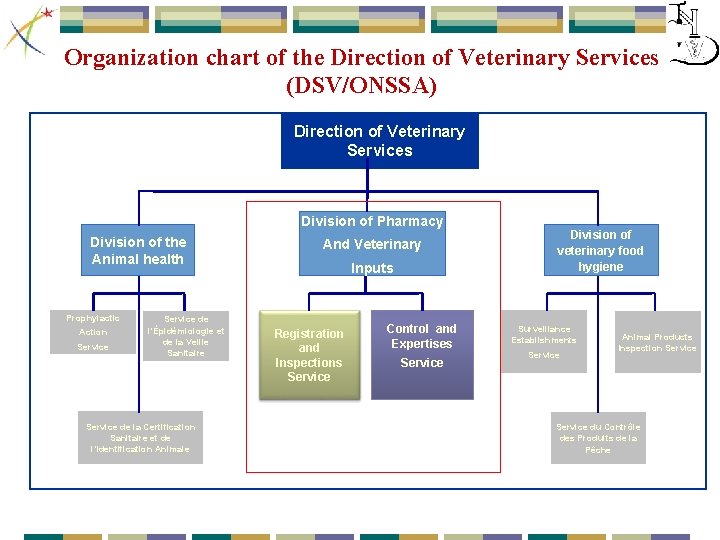
Organization chart of the Direction of Veterinary Services (DSV/ONSSA) Direction of Veterinary Services Division of Pharmacy Division of the Animal health Prophylactic Action Service de l’Épidémiologie et de la Veille Sanitaire Service de la Certification Sanitaire et de l’Identification Animale And Veterinary Inputs Registration and Inspections Service Control and Expertises Service Division of veterinary food hygiene Surveillance Establishments Service Animal Products Inspection Service du Contrôle des Produits de la Pêche

Missions of the DPIV (ONSSA) DPIV has the following missions: q Registration products of medicine and veterinary surgery (medicinal products, biocides, feed additives, . . . ) ; q Inspection of veterinary pharmaceutical industry; q Veterinary pharmacovigilance; q Monitoring of veterinary pharmaceutical sector; q Pharmaceutical veterinary expertise of veterinary drugs and their residues; q Development of regulatory texts for veterinary pharmacy.

DPIV Human ressources Profiles Veterinarians (10) Biologists (10) Technicians (4) Support staff (12) Jobs 6 Vet. instructors 9 Vet. inspectors 11 analysts
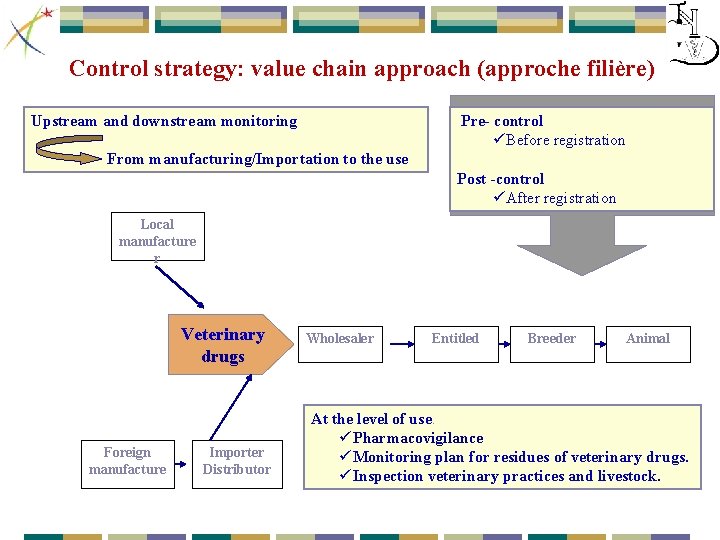
Control strategy: value chain approach (approche filière) Upstream and downstream monitoring Pre- control üBefore registration From manufacturing/Importation to the use Post -control üAfter registration Local manufacture r Veterinary drugs Foreign manufacture Importer Distributor Wholesaler Entitled Breeder Animal At the level of use. ü Pharmacovigilance ü Monitoring plan for residues of veterinary drugs. ü Inspection veterinary practices and livestock.

Registration and Control of pharmaceutical and veterinary inputs processes q All registration activities are according to the standard NM ISO / CEI 9001: 2000. q Inspection of veterinary pharmaceutical plants are under the standard NM ISO / CEI 17020: 1998. (DPIV is accredited since 2012). q Laboratory activities are under NM ISO / CEI 17025: 2005. (DPIV is accredited since 2012). q Assessment technical standards for registration are: • European Pharmacopea (Ph. Eur. ) • United Sted Pharmacopea (USP) • Code Federal Regulatory (USA) • World Organisation for Animal Health (OIE). • VICH guidelines.
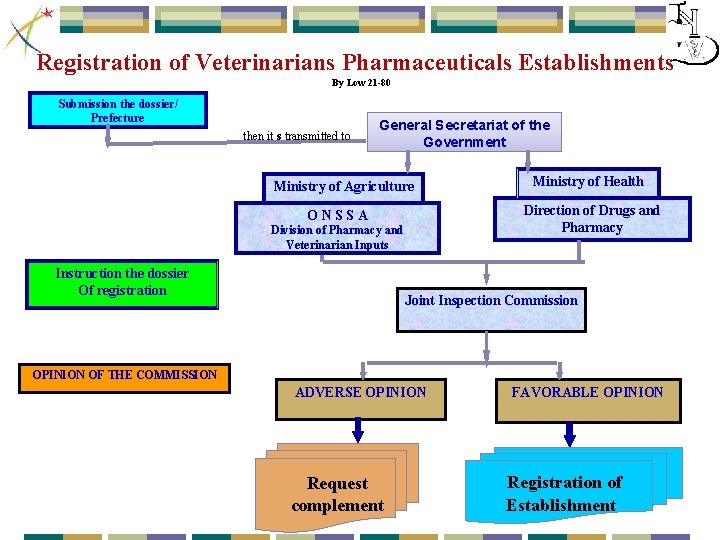
Registration of Veterinarians Pharmaceuticals Establishments By Low 21 -80 Submission the dossier/ Prefecture then it s transmitted to General Secretariat of the Government Ministry of Agriculture O N S S A Division of Pharmacy and Veterinarian Inputs Instruction the dossier Of registration Ministry of Health Direction of Drugs and Pharmacy Joint Inspection Commission OPINION OF THE COMMISSION ADVERSE OPINION Request complement FAVORABLE OPINION Registration of Establishment
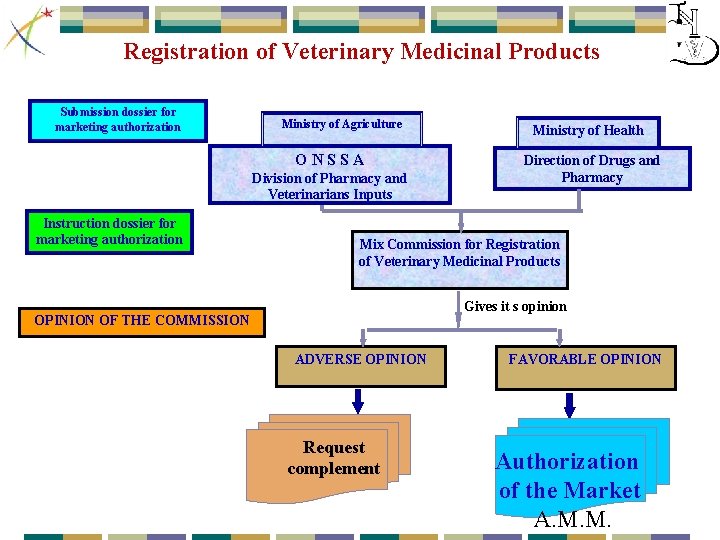
Registration of Veterinary Medicinal Products Submission dossier for marketing authorization Ministry of Agriculture O N S S A Division of Pharmacy and Veterinarians Inputs Instruction dossier for marketing authorization Ministry of Health Direction of Drugs and Pharmacy Mix Commission for Registration of Veterinary Medicinal Products Gives it s opinion OPINION OF THE COMMISSION ADVERSE OPINION Request complement FAVORABLE OPINION Authorization of the Market A. M. M.
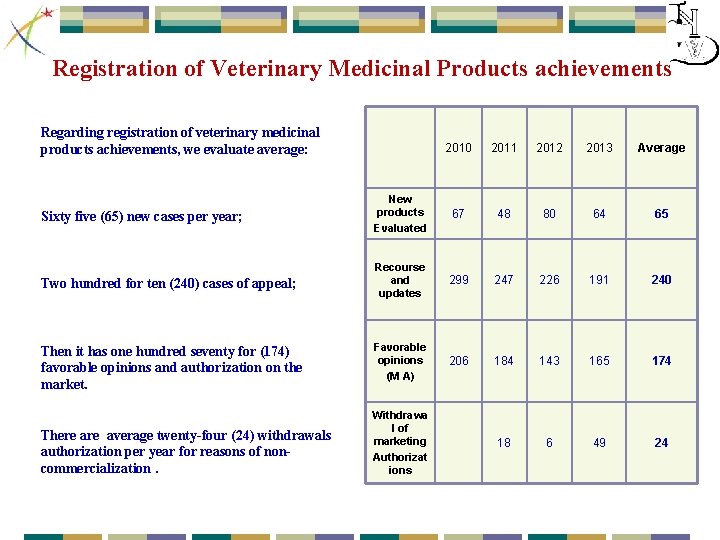
Registration of Veterinary Medicinal Products achievements Regarding registration of veterinary medicinal products achievements, we evaluate average: Sixty five (65) new cases per year; Two hundred for ten (240) cases of appeal; Then it has one hundred seventy for (174) favorable opinions and authorization on the market. There average twenty-four (24) withdrawals authorization per year for reasons of noncommercialization. 2010 2011 2012 2013 Average New products Evaluated 67 48 80 64 65 Recourse and updates 299 247 226 191 240 Favorable opinions (M A) 206 184 143 165 174 18 6 49 24 Withdrawa l of marketing Authorizat ions

Integration of VICH requirements in the registration process Objective: Integration of VICH requirements in the Moroccan registration process Planning: Working meetings were scheduled internally in DPIV since 2012, 2013 and 2014 Achievements: Study and commentary on draft guideline GL 34 Study and review of draft guidelines GL 50 and 51 Internal training and awareness of veterinary instructors for the requirements of GL 3, GL 8 and GL 17 And study on draft guideline GL 52. Perspectives: Integration of other VICH Guidelines in the Moroccan registration process Consultation meetings with the Ministry of Health and professionals Rational use of antibiotics monitoring And antibioresistance monitoring

VICH Guidelines already used in Morocco Nr (Reference) Title of Guideline VICH GL 3 (R) Stability: stability testing of new veterinary drug substances (revision). VICH GL 17 (Stability 4) Stability testing of new biotechnological / biological veterinary medicinal products. VICH GL 1 (Validation Definition) Validation of analytical procedures: definition and terminology. VICH GL 2 (Validation Methodology) Validation of analytical procedures : methodology.

Thank you for your attention
- Slides: 18