Registration of a vaporizer as a MEDICAL DEVICE













- Slides: 20

Registration of a vaporizer as a MEDICAL DEVICE Jorge Fernández

index 1. Medicines to evaporate 2. Medical vaporizer 3. Toxicology and safety 4. Our work

Adventages fast effect 30 to 120 seconds : O easy, not aggressive even pleasant : D decarboxilation improving the action ; ) Non-active ingredients + heat = Activate

Disadventages: users not used how do I breath? : O dosig control when do I stop inhaling? : ( different absortion? : ?

Standardizing the medicine syntesis: active ingredients + excipients (taste, vapor. . ) directly herbal: standardize growing, drying/curing. .

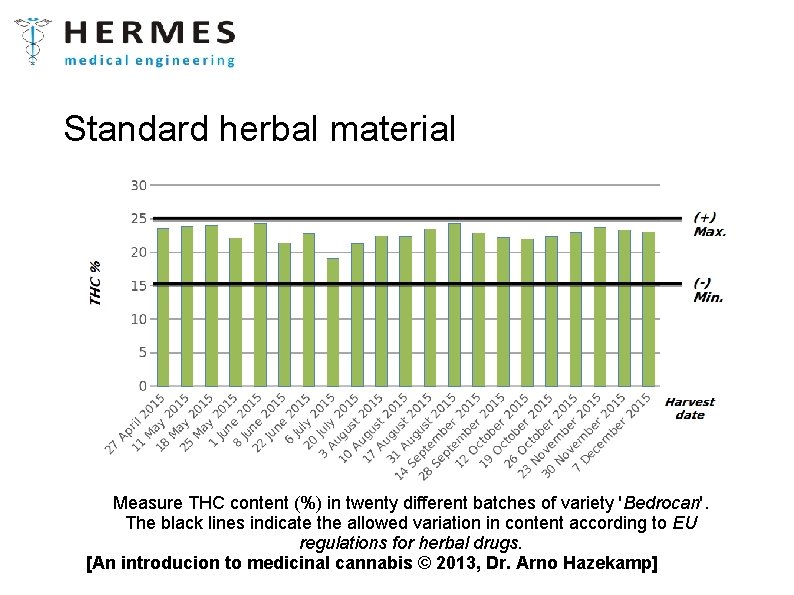
Standard herbal material Measure THC content (%) in twenty different batches of variety 'Bedrocan'. The black lines indicate the allowed variation in content according to EU regulations for herbal drugs. [An introducion to medicinal cannabis © 2013, Dr. Arno Hazekamp]

index 1. Medicines to evaporate 2. Medical vaporizer 3. Toxicology and safety 4. Our work

Standard medicines need: STANDARD administration 1. Protocol to inhale (duration: 5 seconds / number of inhalations: 10 each 30 seconds) 2. Standard hardware settings (temperature stability, type of airflow, conduction/convection heating… ) 3. Standard human parameters (ranges of use: force of inhalation, pressure drop…)

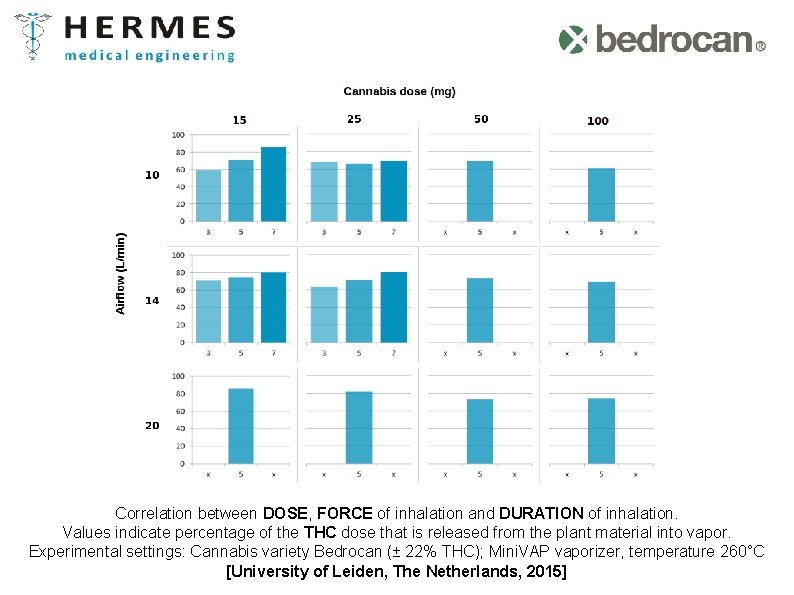
Correlation between DOSE, FORCE of inhalation and DURATION of inhalation. Values indicate percentage of the THC dose that is released from the plant material into vapor. Experimental settings: Cannabis variety Bedrocan (± 22% THC); Mini. VAP vaporizer, temperature 260°C [University of Leiden, The Netherlands, 2015]

Spirocan vaporizer portable, safety, reliable (Hermes Medical core technology) easy, ergonomic, intuitive (thanks to the University of Delft) registration as a medical device (video)

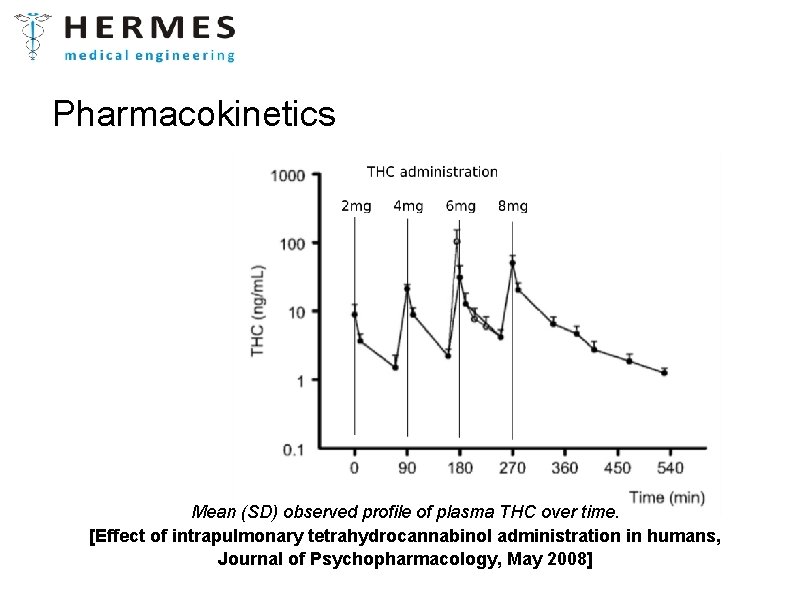
Pharmacokinetics Mean (SD) observed profile of plasma THC over time. [Effect of intrapulmonary tetrahydrocannabinol administration in humans, Journal of Psychopharmacology, May 2008]

index 1. Medicines to evaporate 2. Medical device 3. Toxicology and safety 4. Our work

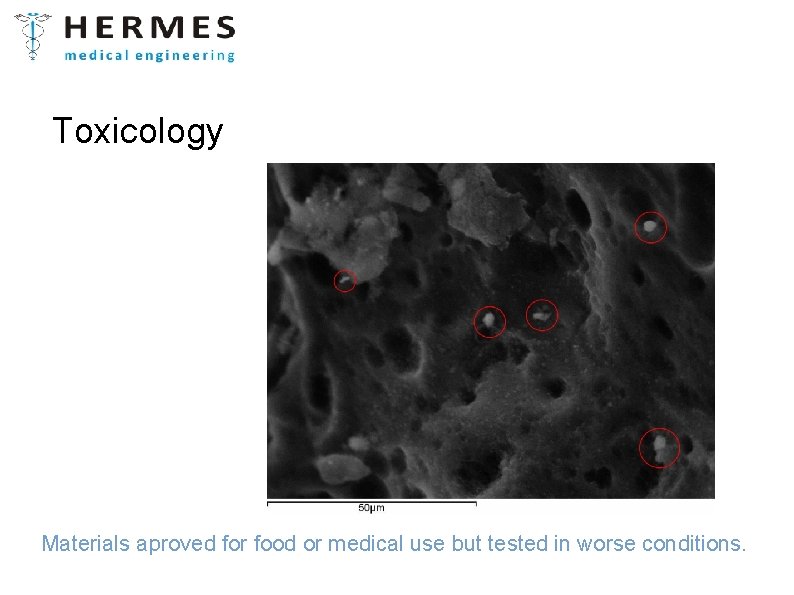
Toxicology Materials aproved for food or medical use but tested in worse conditions.

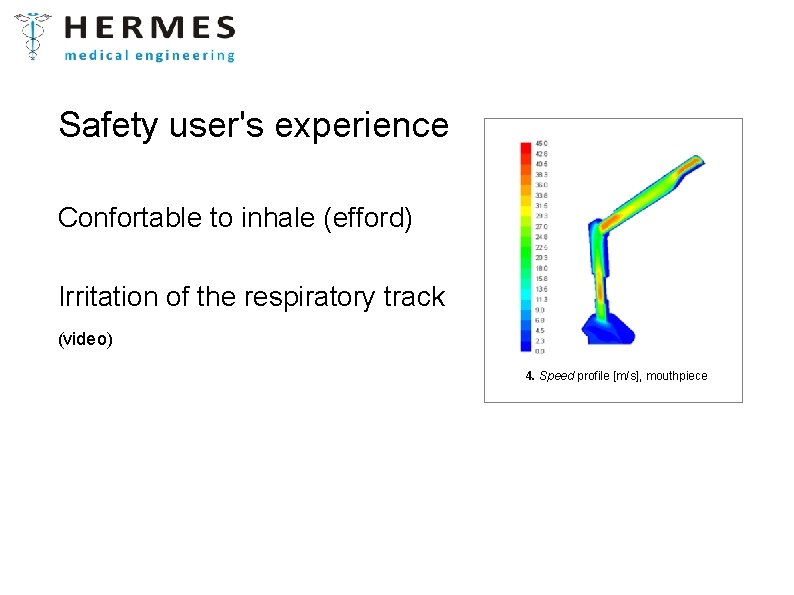
Safety user's experience Confortable to inhale (efford) Irritation of the respiratory track (video) 4. Speed profile [m/s], mouthpiece

Safety standards Intenational standards for: - interface/alarms - materials - ES and EMC safety - manufacturing, tracking, . .

index 1. Medicines to evaporate 2. Medical device 3. Toxicology and safety 4. Our work

Our products www. minivap. com - Looking for a real user experience Design + functionality Original taste & feeling of smoking Convection or mixed convection-conduction in a Portable vaporizer (video)

Our technology: heating cores Designing heating cores for different applications or standars: Neuroscience Recreational Medical. .

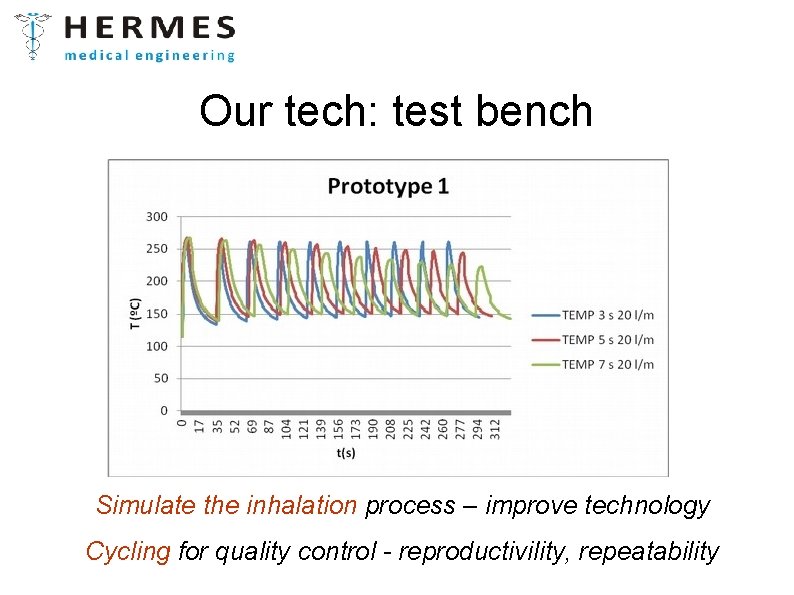
Our tech: test bench Simulate the inhalation process – improve technology Cycling for quality control - reproductivility, repeatability

Thank you www. minivap. com www. hermesmedical. es info@hermesmedical. es