Regents Chemistry Whats a Mole One mole of


























- Slides: 29

Regents Chemistry



What’s a Mole? ? ? One mole of ANY specified entity contains 6. 022 x 1023 of that entity. For example: l l l One mole of donuts contains 6. 022 x 1023 donuts One mole of H 2 O contains 6. 022 x 1023 molecules One mole of nails contains 6. 022 x 1023 nails One mole of Fe contains 6. 022 x 1023 atoms One mole of dogs contains 6. 022 x 1023 dogs One mole of electrons contains 6. 022 x 1023 electrons

(Covalently bonded)

These are Formula units!

How can we measure a mole? A mole = 6. 022 x 1023 particles l Could be a mole of an ionic compound l – contains 6. 022 x 1023 formula units (made of ions) l Could be a mole of a molecular compound – contains 6. 022 x 1023 molecules (made of elements) A mole of an element = the average atomic mass listed on the periodic table l A mole of a compound = total mass of the moles in the compound l

Finding the Mass of a Mole of a Compound

Example #1

Example #2


Summary Formula mass - mass of the smallest unit of an ionic compound; sum of atomic masses in amus l Gram Formula Mass - same as formula mass except in grams l Molecular Mass – same as FM except for used for molecular substances l Mole – 6. 022 x 1023 particles of anything! Just know it is also the GFM of any substance l

Regents Chemistry l Mass, Mole and Particle conversions

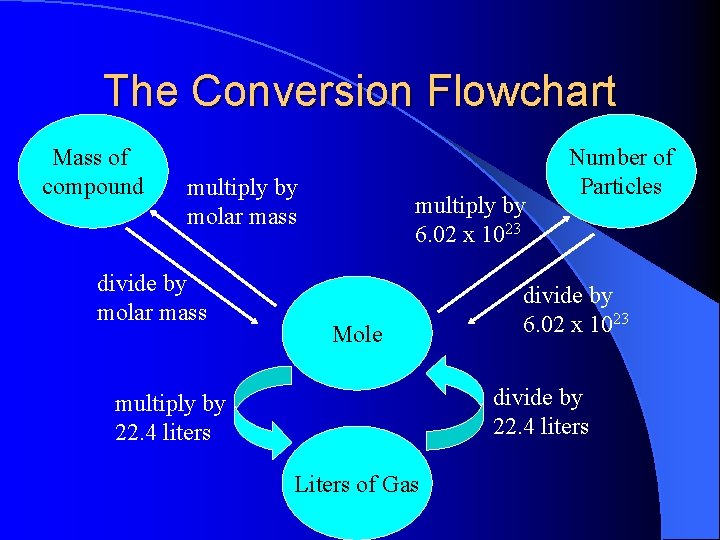
The Conversion Flowchart Mass of compound multiply by molar mass divide by molar mass multiply by 6. 02 x 1023 Mole Number of Particles divide by 6. 02 x 1023 divide by 22. 4 liters multiply by 22. 4 liters Liters of Gas

Practice mole to mass l How many grams are in 2 moles of Cu. Cl 2? 2 moles Cu. Cl 2 x 134 g per mole = 268 g l How many grams are in 3. 5 moles of H 2 O 3. 5 moles H 2 O x 18 g per mole = 63 g

Mass to Mole l How many moles of NO are in 67. 0 g of NO? 67. 0 g NO / 30. 0 g per mole = 2. 2 moles NO l How many moles of KCl are in 125. 0 g of KCl? 125. 0 g KCl / 74. 5 g per mole = 1. 68 moles KCl

Regents Chemistry l Finding Percent Composition

What is Percent Composition? l Formulas represent the composition of a substance l If we use the subscripts and atomic masses of the elements, we can find the percent by mass of each element in a substance l Essentially, Percent Composition finds how much of each element is in a substance

How do we do this? ? l 1. We can add up the total molar mass of all elements involved in the substance l 2. We next simply divide the total of the element in question by the total molar mass and multiply by 100%

Example #1 l Find the percent composition (in grams) of oxygen in potassium chlorate 1. Find the molar mass KCl. O 3 K - 1 mole x 39. 1 grams = 39. 1 g Cl - 1 mole x 35. 5 grams = 35. 5 g O - 3 moles x 16. 0 grams = 48. 0 g Molar mass = 122. 6 g 2. Divide and multiply by 100% %O = 48. 0 g x 100% 122. 6 g %O = 39. 2%

Moles and molecules/formula units l Moles - measured in grams l molecules / formula units - measured in amu (atomic mass units) l So… grams make up moles of a substance l and amu (atomic mass units) make up individual molecules/formula units l Use the same numbers from the PT!

It’s the same process to find the % of amu in a 1 molecule or formula unit! l Find the percent composition (in amu) of oxygen in potassium chlorate 1. Find the mass in amu KCl. O 3 K - 1 atom x 39. 1 amu = 39. 1 amu Cl - 1 atom x 35. 5 amu = 35. 5 amu O - 3 atoms x 16. 0 amu = 48. 0 amu Formula mass = 122. 6 amu 2. Divide and multiply by 100% %O = 48. 0 amu x 100% 122. 6 amu %O = 39. 2%

Example #2 l Find the percent composition (in amu) of water in sodium carbonate crystals Na 2 CO 3 • 10 H 2 O 1. Formula Mass = 286. 0 amu 2. Total formula mass of H 2 O = 180. 0 amu 3. Divide %H 2 O = 180. 0 amu x 100% -------286. 0 amu %H 2 O = 62. 9% worksheet

Regents Chemistry l Mole Relations in Balanced Equations

RECAP l The Law of Conservation of Mass tells us: – Moles (and atoms) in equations must be balanced – Since moles (and atoms) have mass, mass is also balanced (we did this by finding missing mass) – So we have relationship between reactants and products – Thus if we change the moles of reactants (or number of atoms) we change the moles /# of atoms of products. . HOW?

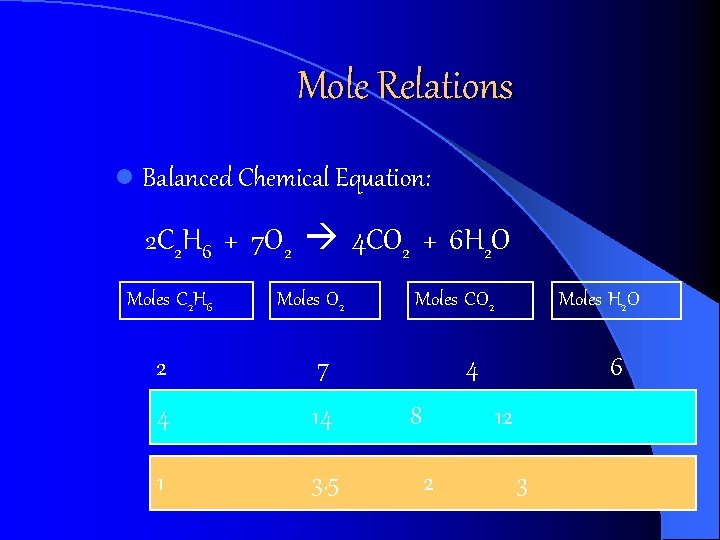
Mole Relations l Balanced Chemical Equation: 2 C 2 H 6 + 7 O 2 4 CO 2 + 6 H 2 O Moles C 2 H 6 Moles O 2 2 7 4 14 1 3. 5 Moles CO 2 Moles H 2 O 4 8 6 12 2 3

Sample Problem l How many moles of water will be produced from the complete combustion of 3. 0 mol of ethane? 2 C 2 H 6 + 7 O 2 4 CO 2 + 6 H 2 O moles ethane: moles water = 2: 6 3. 0 mole C 2 H 6 2. 0 mole C 2 H 6 x = 9. 0 mole H 2 O = x 6 mole H 2 O

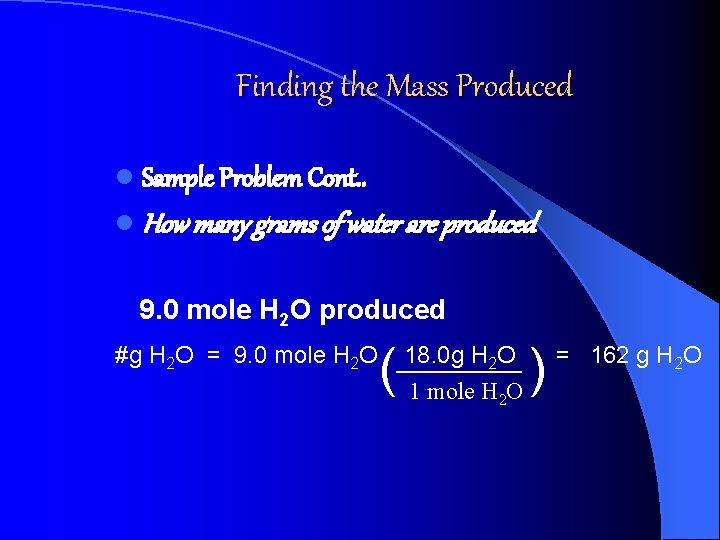
Finding the Mass Produced l Sample Problem Cont. . l How many grams of water are produced 9. 0 mole H 2 O produced #g H 2 O = 9. 0 mole H 2 O ( 18. 0 g H 2 O 1 mole H 2 O ) = 162 g H 2 O

Sample Problem #2 l How many moles of iron (III) oxide are produced from the combination of 6 moles of Fe ? How many grams? 4 Fe + 3 O 2 2 Fe 2 O 3 6 mole Fe 4 mole Fe x = = x mole Fe 2 O 3 2 mole Fe 2 O 3 3 mole Fe 2 O 3 equals 480 g Fe 2 O 3