Regents Chemistry Nuclear Chemistry Stability of Isotopes Transmutation
Regents Chemistry Nuclear Chemistry Stability of Isotopes Transmutation • June 8 -10, 2020

The ratio of neutrons to protons in the nucleus determines the stability of a specific nucleus. Too many neutrons = unstable • If any element has an unstable isotope, it is radioactive and called a radioisotope. • All elements above atomic number 83 are radioactive

Transmutation • When the atomic nucleus of one element is changed into the nucleus of a different element
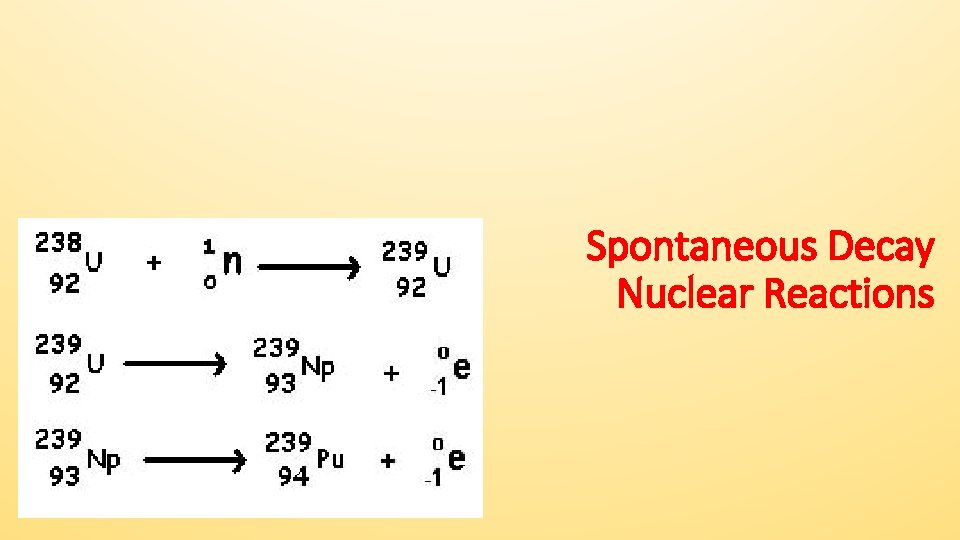
Spontaneous Decay Nuclear Reactions

Reference Tables N and O -Table N lists the type of decay mode for specific nuclides (radioisotopes) -Table O lists information about the most common types of radiation

Spontaneous Decay • Can involve the release of alpha particles, beta particles, positrons, and/or gamma radiation from the nucleus of an UNSTABLE atom. • These emissions are listed and described on Tables N and O. They have different masses, charges, and power.

You must be able to write nuclear equations for alpha and beta decay. • Alpha Decay- when an unstable nucleus emits an alpha particle, the atomic number decreases by 2, and the atomic mass decreases by 4

You must be able to write nuclear equations for alpha and beta decay. • Alpha Decay- when an unstable nucleus emits an alpha particle, the atomic number decreases by 2, and the atomic mass decreases by 4

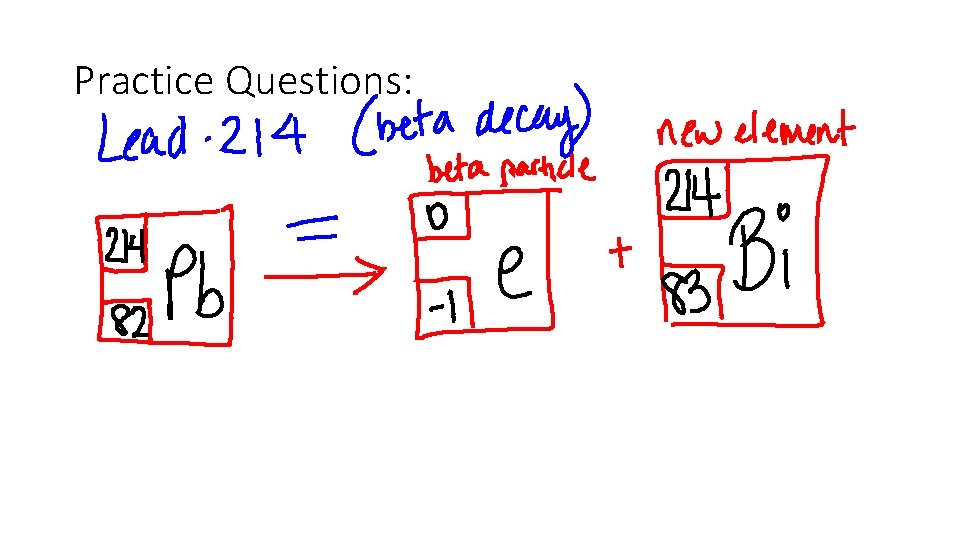
Beta Decay- when an unstable nucleus emits a beta particle, the atomic number INCREASES by 1, and the mass number stays the same. • What’s really going on? A neutron in the nucleus breaks up into an electron and a proton. The electron is emitted which changes that crazy neutron into a proton!

Write the beta decay equation for Lead-214
Practice Questions:
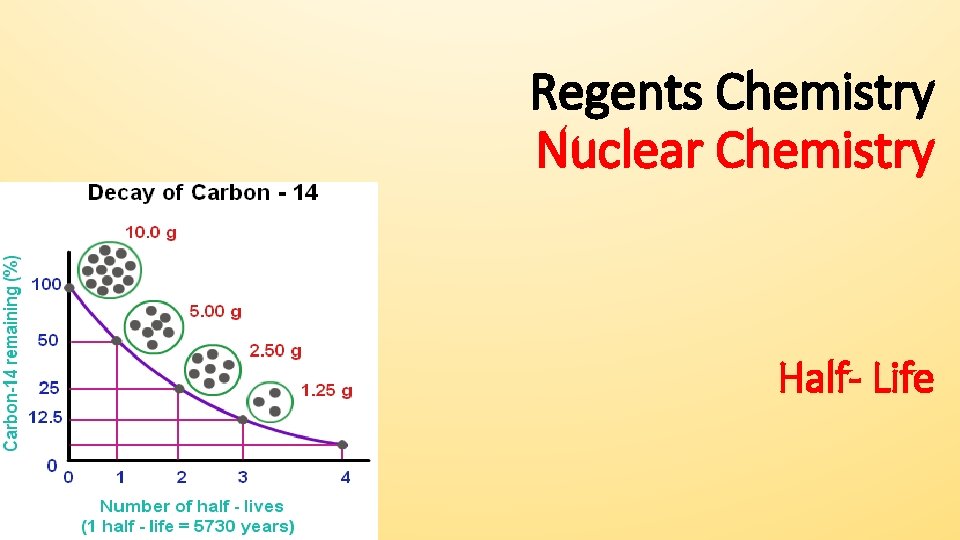
Regents Chemistry Nuclear Chemistry Half- Life

Every radioactive isotope has a specific mode and rate of decay (half-life)- see Table N • This constant rate is unaffected by temperature, pressure, or concentration • Half-life: the time it takes for half of the atoms in a given sample to decay • The shorter the half-life = less stable isotope

Half-life Calculations: • There are three potential questions that you need to be able to solve. The question will give you two of the three variables, and you must calculate the missing information • 1 - initial amount • 2 - fraction remaining/grams remaining • 3 - half-life of the isotope

To solve half-life problems, you will need lots of space to write and show your work… • Look at examples 1, 2 and 3 on pages 83 and 84 in the note packet
- Slides: 15