Regents Chemistry 11032008 Mrs Nephew Physical Behavior of

Regents Chemistry 11/03/2008 Mrs. Nephew

Physical Behavior of Matter Phases of Matter Solidmatter held in rigid form definite volume, definite shape particles held in place by strong attractive forces. crystalline structure

Liquid. Particles not held together rigidly Particles can move past one another Lack definite shape Have definite volume

Gas. Minimal attractive forces holding them together Lack definite shape Lack definite volume Expand to fill container
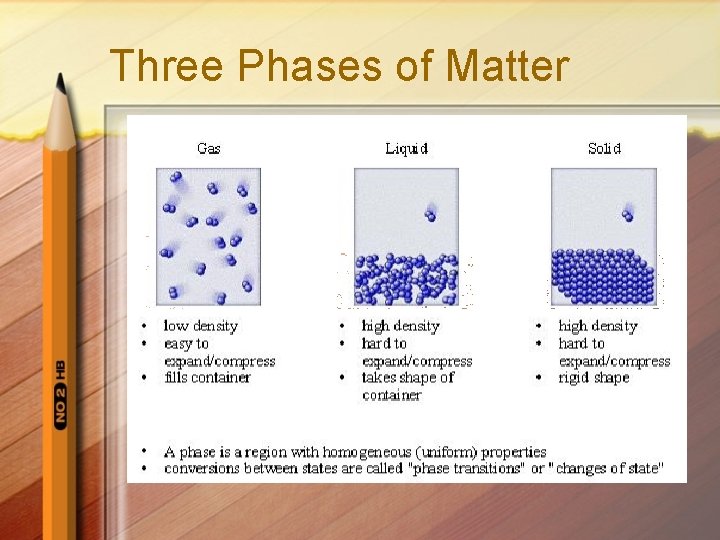
Three Phases of Matter

Potential Energy l The energy associated with the position of the object; the “stored” form of energy.

Kinetic Energy l The energy associated with the motion of an object.

Heating and Cooling Curves l Show temperature of a sample varies over time as it is heated.

Heating Curves A solid is heated (1) then it melts (2). Once it is totally melted, heat the liquid to the boiling point (3), boil it (4), and then raise the temperature of the gas (5).

Heating Curves l l Sections (1), (3), (5) of this graph are showing an increase in kinetic energy as the temperature increases. Sections (2), (4) of this graph are showing an increase in potential energy through the phase changes.

6 phase changes you need to know l l l Boiling/vaporization: increase in potential energy, changing a liquid to a gas Condensation: decrease in potential energy, changing a gas to a liquid Melting/fusion: increase in potential energy, changing a solid to a liquid Freezing/solidification: decrease in potential energy, changing a liquid to a solid Sublimation: Increase in potential energy, changing a solid to a gas Deposition: Decrease in potential energy, changing a gas to a solid

REMEMBER… l l Endothermic reactions absorb heat energy (heat is a reactant). Exothermic reactions release heat energy (heat is a product). ***Heating curve steps are all endothermic (absorb heat energy).

Fusion: Also known as melting. When the particles in the substance eventually possess enough kinetic energy to break the bonds holding them in the solid phase. Temperature remains constant even though heat is added at a constant rate. Heat is absorbed by the substance as potential energy. Kinetic energy remains constant. Liquid and solid phases are present. (Liquid and solid over time).

Heat of Fusion l Amount of heat needed to convert a solid at its melting point to a liquid.

Vaporization: l l Also known as boiling. Once all the solid is melted, only liquid is present, temperature begins to rise again. Particles in the liquid have enough kinetic energy to break the attractive forces holding them in the liquid phase. Temp. remains constant, objects PE increases as heat is absorbed. (Gas has more PE than liquid phase)

Cooling Curve If a gas at high temp is allowed to cool at a constant rate, a cooling curve results. Phase changes are not accompanied by temperature changes.

l l l The reverse of boiling is condensation. The reverse of melting is freezing (or solidification). The cooling curve steps are all EXOTHERMIC (release heat energy).

l Sublimation-solid goes directly to gas without passing through a noticeable liquid phase. (solid CO 2). l Deposition-reverse of sublimation, where a gas changes directly into a solid.

Homework l Worksheet on Physical Behavior of Matter Problems 1 -16. Due tomorrow in class.
- Slides: 19