Reduction Oxidation Chapters 20 21 Oxidation Numbers States

Reduction Oxidation Chapters 20 + 21

Oxidation Numbers (States) n Positive, negative or neutral values assigned to an atom to keep track of the number of electrons lost or gained.

Oxidation Number Rules n Elements alone (not in a compound) ¨ Example: n Cu, N 2 Monatomic ion (single atom) = charge ¨ Example: Cl- =0

Oxidation Number Rules n Compound ¨ Example: n H 2 O sum of all atoms = 0 H+H+O=0 Polyatomic ion ¨ sum of all atoms = charge ¨ Example: NO 3 - N + O + O = -1

Common Oxidation Numbers Group 1 +1 n Group 2 +2 n Group 13 +3 n Group 15 -3 n Group 16 -2 n Group 17 -1 n ¨ Some exceptions to each above


Redox Reactions n Reduction – Oxidation, or redox, involves the transfer of electrons n Reduction – gain of electrons n Oxidation – loss of electrons

Redox Reactions n LEO goes GER n Lose Electrons Oxidation n Gain Electrons Reduction

Redox Reaction 0 n Mg 0 +2 -1 + Cl 2 Mg. Cl 2 ¨Mg - lost electrons (oxidation) ¨Cl – gained electrons (reduction)

Redox Reaction 0 n 2 Al ¨Ni +2 +5 -2 0 +3 +5 -2 + 3 Ni(NO 3)2 3 Ni + 2 Al(NO 3)3 - lost electrons (oxidation) – gained electrons (reduction)


Redox Reaction n Zn + Cu. SO 4 Cu + Zn. SO 4 ¨ One element loses electrons (oxidation) ¨ One element gains electrons (reduction) ¨ All other ions are spectators

Net Ionic Equation n Shows only the ions involved in the redox reaction, not spectator ions ¨ Still shows conservation of mass and charge n Zn + Cu. SO 4 Cu + Zn. SO 4 n Zn + Cu+2 Cu + Zn+2

Net Ionic Example n Zn + 2 HCl H 2 + Zn. Cl 2 n Zn + 2 H+ H 2 + Zn 2+


Half Reactions Only shows one element and how many electrons are gained or lost n Zn + Cu. SO 4 Cu + Zn. SO 4 n ¨ Zn + Cu+2 Cu + Zn+2 ¨ Zn Zn+2 + 2 e- ¨ Cu 2+ + 2 e- Cu (Net Ionic) Oxidation Reduction

Oxidation n Loss of Electrons n Examples: ¨ Zn ¨ 2 Cl- Zn+2 + 2 e Cl 2 + 2 e-

Reduction n Gain of electrons n Examples: ¨ Ag+ ¨ Cl 2 + e- Ag + 2 e- 2 Cl-

Balancing Reactions n The number of electrons lost must equal the number of electrons gained Example: n 2 Na + Zn. Cl 2 Zn + 2 Na. Cl n ¨ Zn+2 + 2 e- Zn ¨ 2(Na Na + + e- )


Spontaneous Reactions n More active element does not want to be alone ¨ Table n J Metal being oxidized must be ABOVE metal being reduced for spontaneous reactions to occur ¨ Reversed for Nonmetals

Spontaneous Reactions Examples: n Zn + Cu. SO 4 Cu + Zn. SO 4 n YES n Ca. SO 4 + Mg Ca + Mg. SO 4 n Zn + 2 HCl H 2 + Zn. Cl 2 n F 2 + 2 Na. I I 2 + 2 Na. F NO YES


Electrochemical Cells n any device that converts chemical energy into electrical energy or electrical energy into chemical energy n Two types ¨ Voltaic (Chemical) ¨ Electrolytic

Electrochemical Cells n Electrode – conductor in an electrical circuit that carries electrons to or from a substance other than a metal. Cathode – electrode where reduction takes place n Anode – electrode where oxidation takes place n

Voltaic Cell n Flow of electrons is spontaneous based on electronegativity and ionization energy n Chemical energy is converted to electrical energy n Examples: Batteries

Voltaic Cell



Electrochemical Cell Components n Salt Bridge ¨ Allows n for the passage of ions, not electrons Switch ¨ Device that opens(turns off) and closes(turns on) circuit

Voltaic Cell

Electrolysis n Process in which electrical energy is converted to chemical energy n Example: ¨ 2 H 2 O 2 H 2 + O 2

Electrolytic Cells n Electrons are pushed by an outside power source n Electrical energy is converted to chemical energy n Examples: Electroplating, Electropolishing

Electrolytic Cell
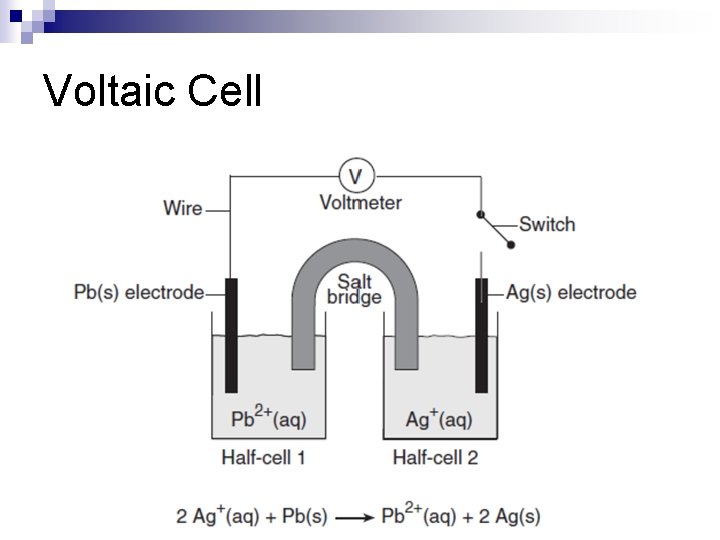
Voltaic Cell
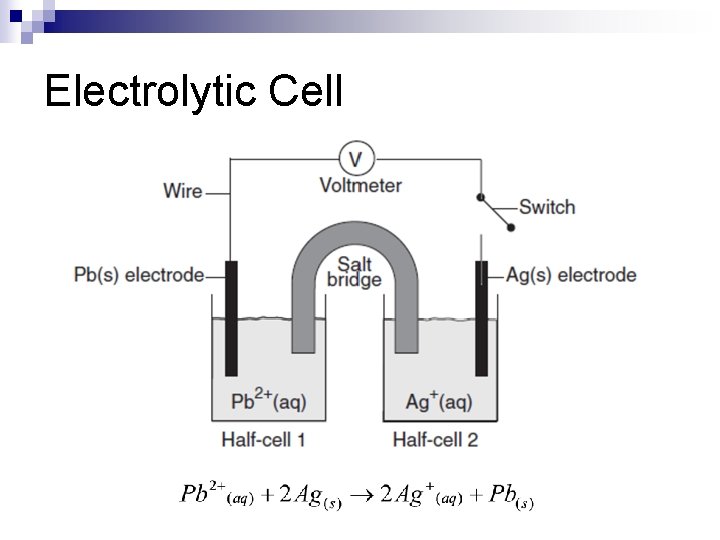
Electrolytic Cell

Voltaic or Electrolytic?

Voltaic or Electrolytic?
- Slides: 38