Reduction of a Reducible Representation Given a Reducible

Reduction of a Reducible Representation. Given a Reducible Rep how do we find what Irreducible reps it contains? Irreducible reps may be regarded as orthogonal vectors. The magnitude of the vector is h-1/2 Any representation may be regarded as a vector which is a linear combination of the irreducible representations, i = 1, 2…. Irred. Reps Reducible Rep = S (ai * Irreducible. Repi) i The Irreducible reps are orthogonal. Hence for the reducible rep and a particular irreducible rep j Sym ops S(character of Reducible Rep)(character of Irreducible Repi) = aj * h Or Sym ops aj = S(character of Reducible Rep)(character of Irreducible Repi) / h
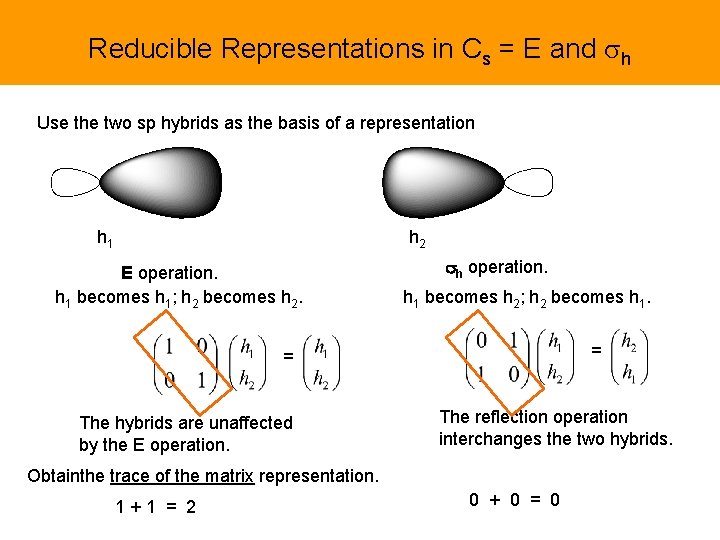
Reducible Representations in Cs = E and sh Use the two sp hybrids as the basis of a representation h 1 h 2 E operation. h 1 becomes h 1; h 2 becomes h 2. sh operation. h 1 becomes h 2; h 2 becomes h 1. = = The hybrids are unaffected by the E operation. The reflection operation interchanges the two hybrids. Obtainthe trace of the matrix representation. 1+1 = 2 0 + 0 = 0

Let’s observe one helpful thing here. Only the objects (hybrids) that remain themselves, appear on the diagonal of the transformation of the symmetry operation, contribute to the trace. They commonly contribute +1 or -1 to the trace depending whether or not they are multiplied by -1. h 1 h 2; do not become themselves, interchange h 1 , h 1; become themselves = = The hybrids are unaffected by the E operation. The reflection operation interchanges the two hybrids. Proceed using the trace of the matrix representation. 1+1 = 2 0 + 0 = 0

The Irreducible Representations for Cs. Cs E sh A’ A” 1 1 1 x, y, Rz x 2, y 2, z 2, xy -1 z, Rx, Ry yz, xz The reducible representation derived from the two hybrids can be attached to the table. G 2 0 (h 1, h 2) Note that G = A’ + A”

The Irreducible Representations for Cs. Cs E sh A’ A” 1 1 1 x, y, Rz x 2, y 2, z 2, xy -1 z, Rx, Ry yz, xz Let’s verify some things. Order of the group = # sym operations = 2 A’ and A’’ are orthogonal: 1*1 + 1+(-1) = 0 Sum of the squares over sym operations = order of group = h The magnitude of the A’ and A’’ vectors are each (2) 1/2: magnitude 2 = ( 12 + (+/- 1)2)

The Irreducible Representations for Cs. Cs E sh A’ A” 1 1 1 x, y, Rz x 2, y 2, z 2, xy -1 z, Rx, Ry yz, xz The reducible representation derived from the two hybrids can be attached to the table. G 2 0 (h 1, h 2)

Now let’s do the reduction. We assume that the reducible rep G can be expressed as a linear combination of A’ and A’’ G = a. A’ A’ + a. A’’; our task is to find out the coefficients a. A’ and a. A’’ Cs E sh A’ A” 1 1 1 -1 x, y, Rz z, Rx, Ry G 2 0 (h 1, h 2) x 2, y 2, z 2, xy yz, xz a. A’ = (1 * 2 + 1 *0)/2 = 1 a. A’’ = (1 * 2 + 1 *0)/2 = 1 Or again G = 1*A’ + 1*A’’. Note that this holds for any reducible rep G as above and not limited to the hybrids in any way.

Another Example of Reduction… Sulfur Difluoride belongs to the C 2 v point group. What this means is that the molecular structure behaves at A 1 for C 2 v This means that the molecular structure is transformed into itself by all the sym operations. We can apply these symmetry operations to any kind of object and see how the object behaves. Additional objects may be needed to show what the symmetry operations do.
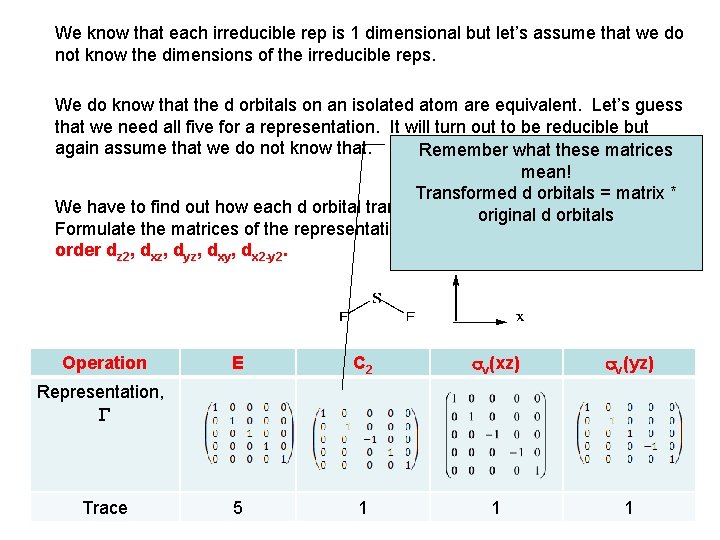
We know that each irreducible rep is 1 dimensional but let’s assume that we do not know the dimensions of the irreducible reps. We do know that the d orbitals on an isolated atom are equivalent. Let’s guess that we need all five for a representation. It will turn out to be reducible but again assume that we do not know that. Remember what these matrices mean! Transformed d orbitals = matrix * We have to find out how each d orbital transforms under the C operations. original d 2 vorbitals Formulate the matrices of the representation based on the d orbitals in this order dz 2, dxz, dyz, dxy, dx 2 -y 2. Operation E C 2 sv(xz) sv(yz) 5 1 1 1 Representation, G Trace

Now do Reduction. We have four irreducible reps: A 1, A 2, B 1, B 2. The reducible rep, G, should be expressible as a linear combination of the irreducible reps. G = a. A 1 + a. A 2 + a. B 1 + a. B 2 Or using vector notation…. (5, 1, -1) = a. A 1 (1, 1, 1, 1) + a. A 2 (1, 1, -1) + a. B 1 (1, -1, 1, -1) + a. B 2 (1, -1, 1) Find one of the a coefficents, say, a. B 1, by multiplying everything by the elements of the B 1 rep and summing over sym elements. (1, -1, 1, -1) (1, -1, 1, -1) x x x (5, 1, 1, 1) = a. A 1 (1, 1, 1, 1) + a. A 2 (1, 1, -1) + a. B 1 (1, -1, 1, -1) + a. B 2 (1, -1, 1) 5 -1 +1 -1 = 4 0 0 a. B 1 4 a B 1 = 1 and similarly: a. A 1 = 2; a. A 2 = 1; a. B 2 = 1 Or the d orbitals transform as G = 2 A 1 + A 2 + B 1 + B 2 0

The d orbitals transform as G = 2 A 1 + A 2 + B 1 + B 2 Note that dz 2 and dx 2 -y 2 transform as A 1 dxy as A 2 dxz as B 1 dyz as B 2
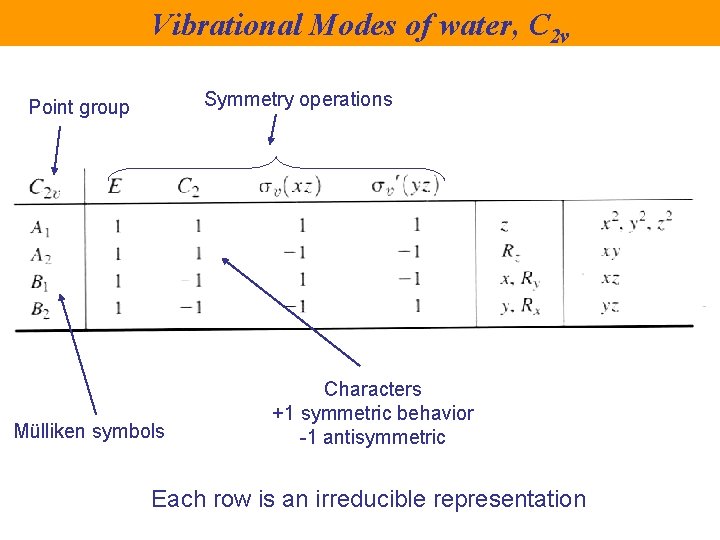
Vibrational Modes of water, C 2 v Symmetry operations Point group Mülliken symbols Characters +1 symmetric behavior -1 antisymmetric Each row is an irreducible representation
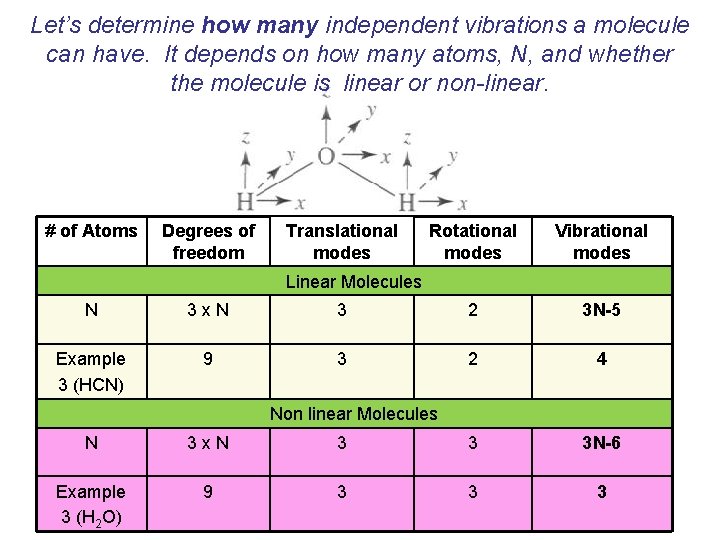
Let’s determine how many independent vibrations a molecule can have. It depends on how many atoms, N, and whether the molecule is linear or non-linear. # of Atoms Degrees of freedom Translational modes Rotational modes Vibrational modes Linear Molecules N 3 x. N 3 2 3 N-5 Example 3 (HCN) 9 3 2 4 Non linear Molecules N 3 x. N 3 3 3 N-6 Example 3 (H 2 O) 9 3 3 3

Symmetry and molecular vibrations A molecular vibration is IR active only if it results in a change in the dipole moment of the molecule A molecular vibration is Raman active only if it results in a change in the polarizability of the molecule In group theory terms: A vibrational mode is IR active if it corresponds to an irreducible representation with the same symmetry of a x, y, z coordinate (or function) and it is Raman active if the symmetry is the same as A quadratic function x 2, y 2, z 2, xy, xz, yz, x 2 -y 2 If the molecule has a center of inversion, no vibration can be both IR & Raman active
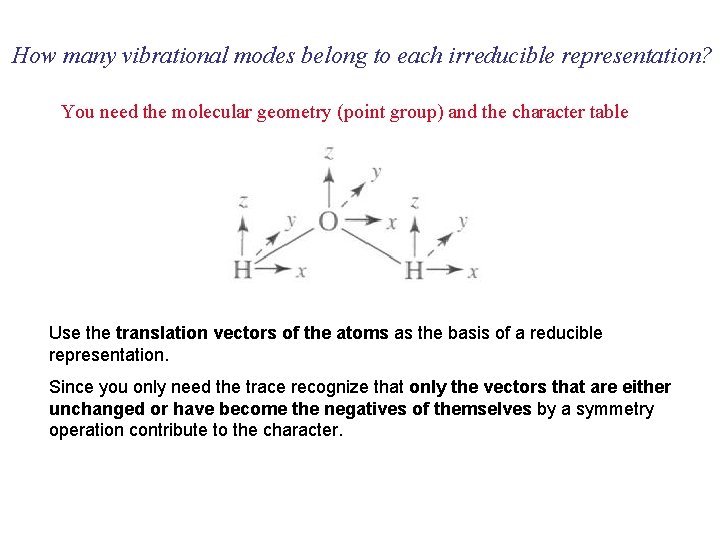
How many vibrational modes belong to each irreducible representation? You need the molecular geometry (point group) and the character table Use the translation vectors of the atoms as the basis of a reducible representation. Since you only need the trace recognize that only the vectors that are either unchanged or have become the negatives of themselves by a symmetry operation contribute to the character.

A shorter method can be devised. Recognize that a vector is unchanged or becomes the negative of itself if the atom does not move. A reflection will leave two vectors unchanged and multiply the other by -1 contributing +1. For a rotation leaving the position of an atom unchanged will invert the direction of two vectors, leaving the third unchanged. Etc. Apply each symmetry operation in that point group to the molecule and determine how many atoms are not moved by the symmetry operation. Multiply that number by the character contribution of that operation: E=3 s=1 C 2 = -1 i = -3 C 3 = 0 That will give you the reducible representation

Finding the reducible representation E=3 s=1 C 2 = -1 i = -3 C 3 = 0 G 3 x 3 9 1 x-1 -1 3 x 1 3 1 x 1 (# atoms not moving x char. contrib. ) 1

Now separate the reducible representation into irreducible ones to see how many there are of each type S G 9 -1 3 1 A 1 = 1/4 (1 x 9 x 1 + 1 x(-1)x 1 + 1 x 3 x 1 + 1 x 1 x 1) = 3 A 2 = 1/4 (1 x 9 x 1 + 1 x(-1)x 1 + 1 x 3 x(-1) + 1 x 1 x(-1)) = 1
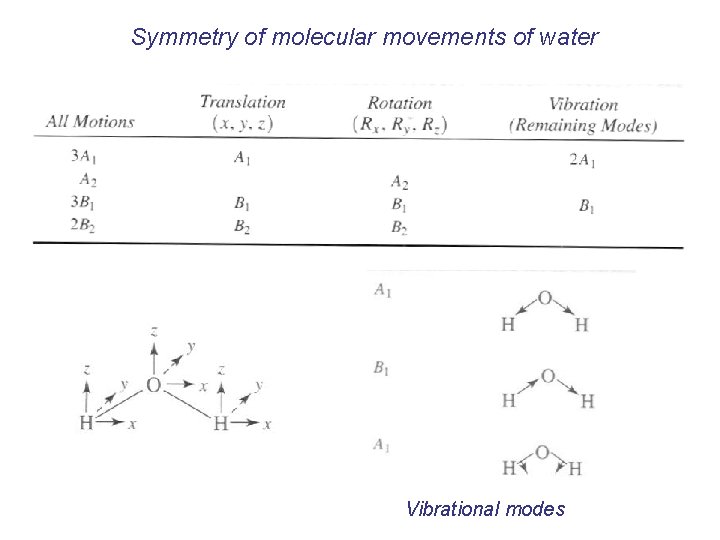
Symmetry of molecular movements of water Vibrational modes

Which of these vibrations having A 1 and B 1 symmetry are IR or Raman active? Raman active IR active
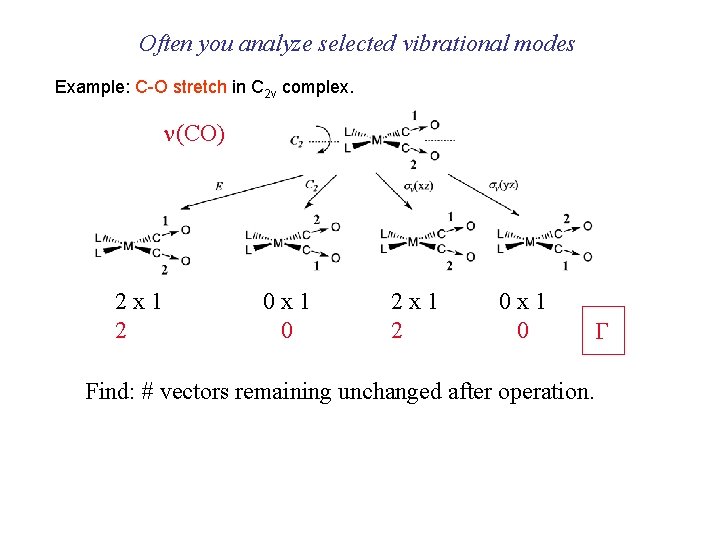
Often you analyze selected vibrational modes Example: C-O stretch in C 2 v complex. n(CO) 2 x 1 2 0 x 1 0 Find: # vectors remaining unchanged after operation. G
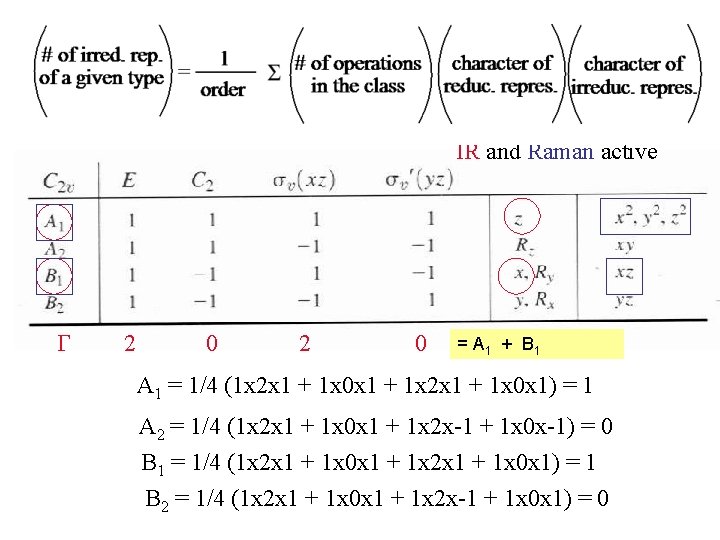
Both A 1 and B 1 are IR and Raman active G 2 0 = A 1 + B 1 A 1 = 1/4 (1 x 2 x 1 + 1 x 0 x 1 + 1 x 2 x 1 + 1 x 0 x 1) = 1 A 2 = 1/4 (1 x 2 x 1 + 1 x 0 x 1 + 1 x 2 x-1 + 1 x 0 x-1) = 0 B 1 = 1/4 (1 x 2 x 1 + 1 x 0 x 1 + 1 x 2 x 1 + 1 x 0 x 1) = 1 B 2 = 1/4 (1 x 2 x 1 + 1 x 0 x 1 + 1 x 2 x-1 + 1 x 0 x 1) = 0
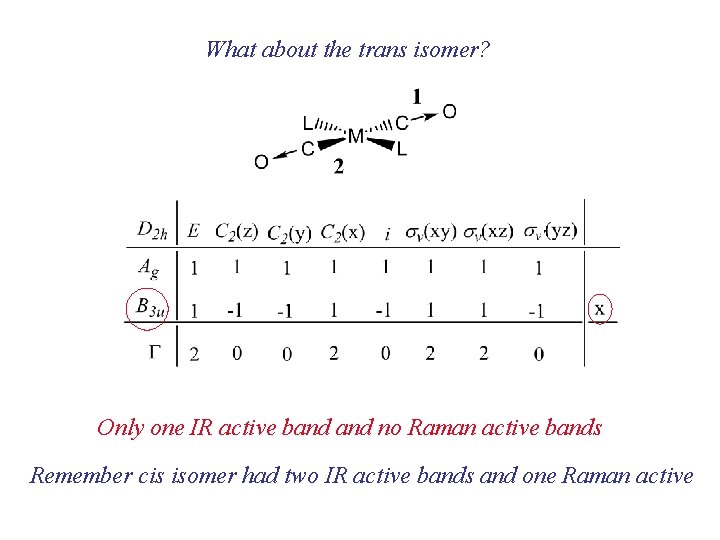
What about the trans isomer? Only one IR active band no Raman active bands Remember cis isomer had two IR active bands and one Raman active
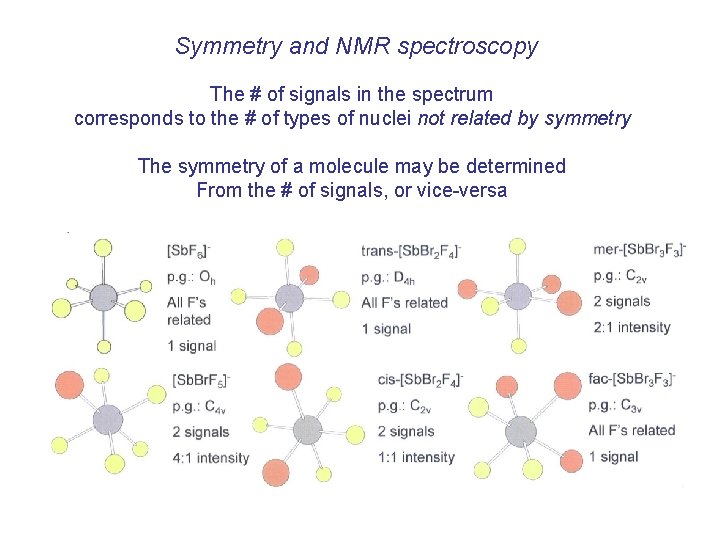
Symmetry and NMR spectroscopy The # of signals in the spectrum corresponds to the # of types of nuclei not related by symmetry The symmetry of a molecule may be determined From the # of signals, or vice-versa

Molecular Orbitals

Atomic orbitals interact to form molecular orbitals Electrons are placed in molecular orbitals following the same rules as for atomic orbitals In terms of approximate solutions to the Scrödinger equation Molecular Orbitals are linear combinations of atomic orbitals (LCAO) Y = caya + cbyb (for diatomic molecules) Interactions depend on the symmetry properties and the relative energies of the atomic orbitals
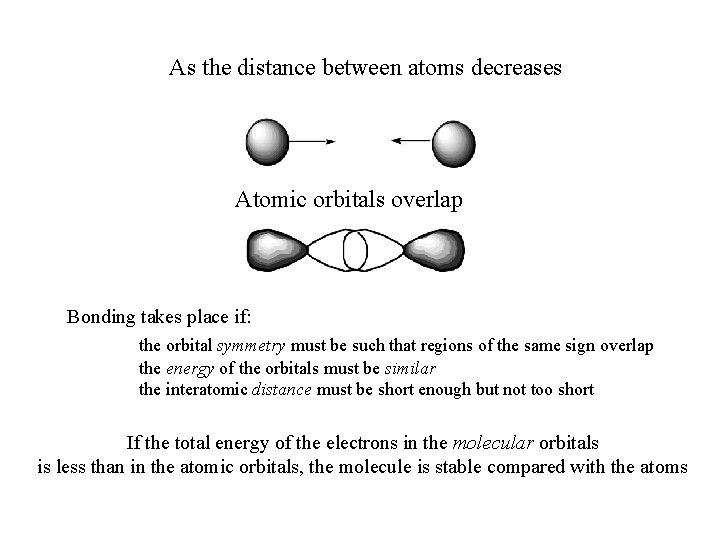
As the distance between atoms decreases Atomic orbitals overlap Bonding takes place if: the orbital symmetry must be such that regions of the same sign overlap the energy of the orbitals must be similar the interatomic distance must be short enough but not too short If the total energy of the electrons in the molecular orbitals is less than in the atomic orbitals, the molecule is stable compared with the atoms
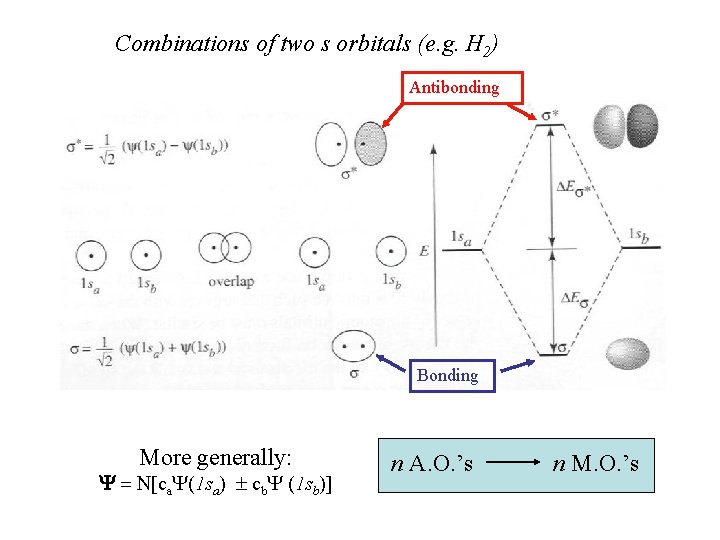
Combinations of two s orbitals (e. g. H 2) Antibonding Bonding More generally: Y = N[ca. Y(1 sa) ± cb. Y (1 sb)] n A. O. ’s n M. O. ’s
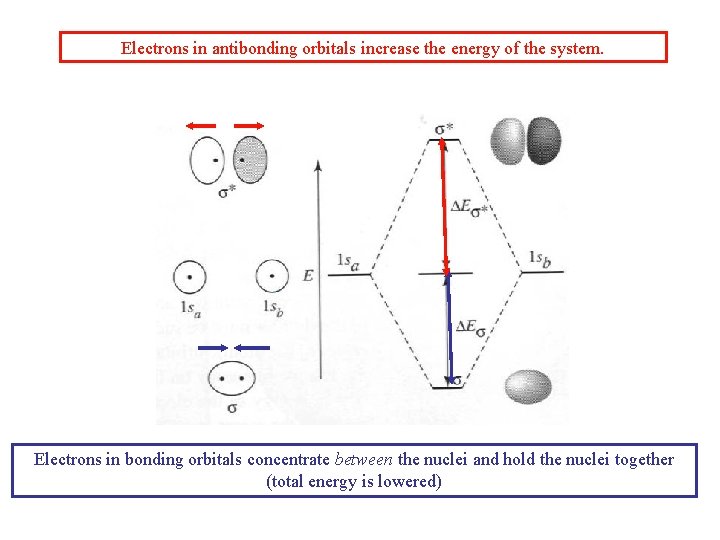
Electrons in antibonding orbitals increase the energy of the system. Electrons in bonding orbitals concentrate between the nuclei and hold the nuclei together (total energy is lowered)
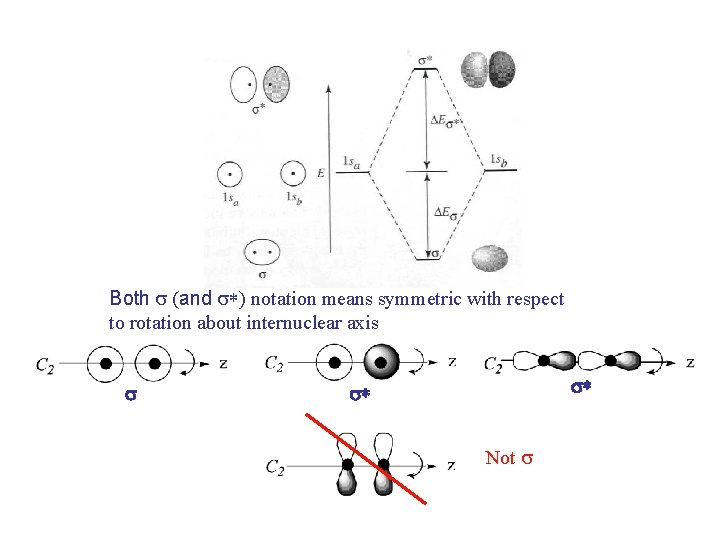
Both s (and s*) notation means symmetric with respect to rotation about internuclear axis s s* s* Not s
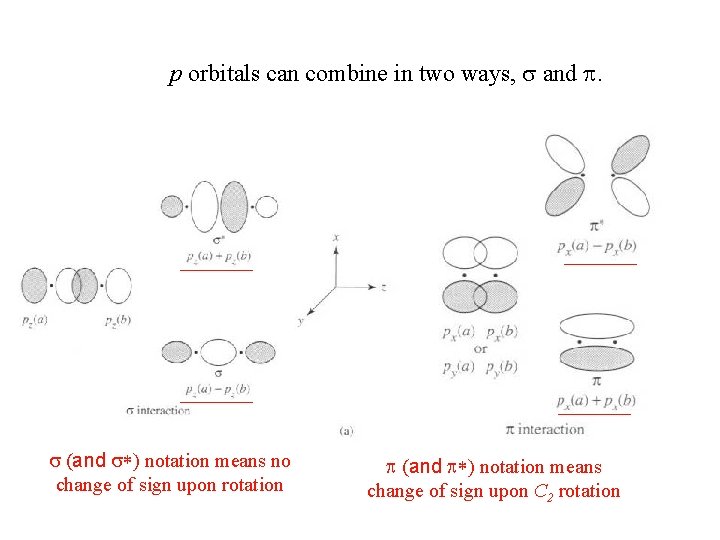
p orbitals can combine in two ways, s and p. s (and s*) notation means no change of sign upon rotation p (and p*) notation means change of sign upon C 2 rotation
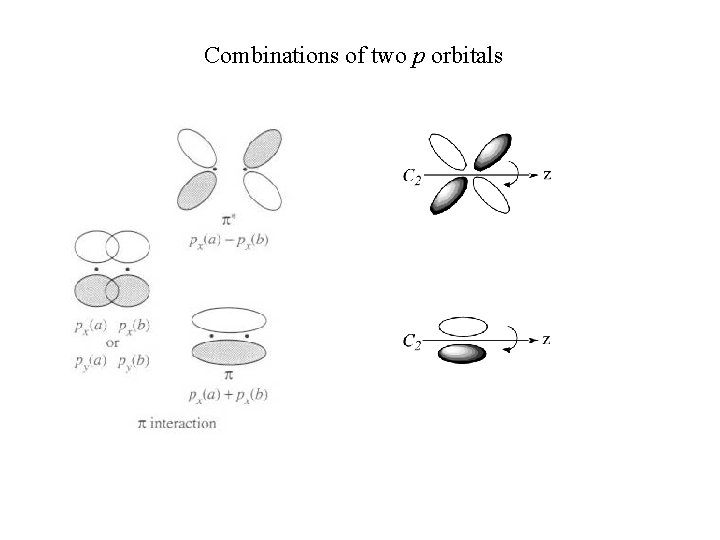
Combinations of two p orbitals
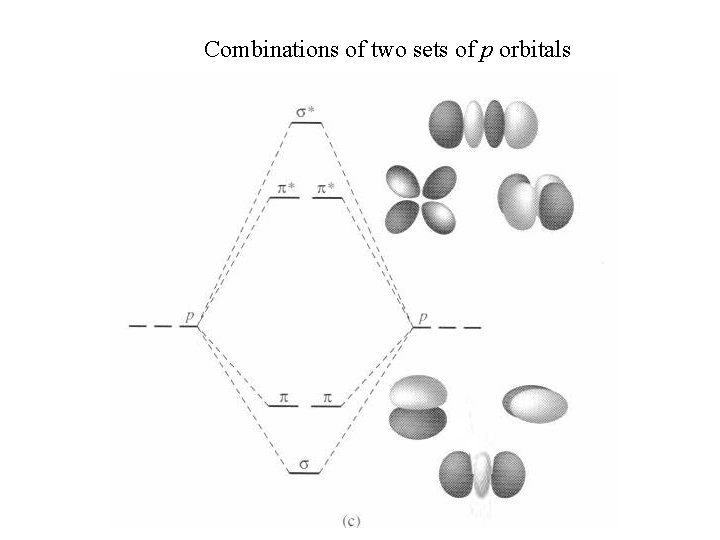
Combinations of two sets of p orbitals
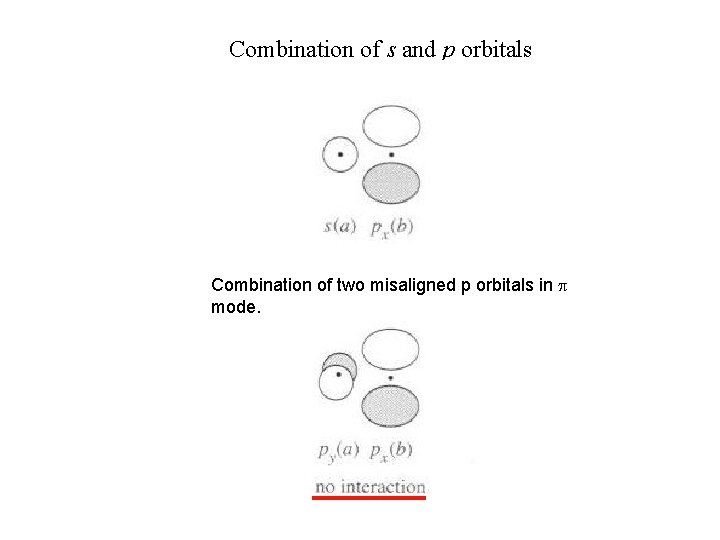
Combination of s and p orbitals Combination of two misaligned p orbitals in p mode.
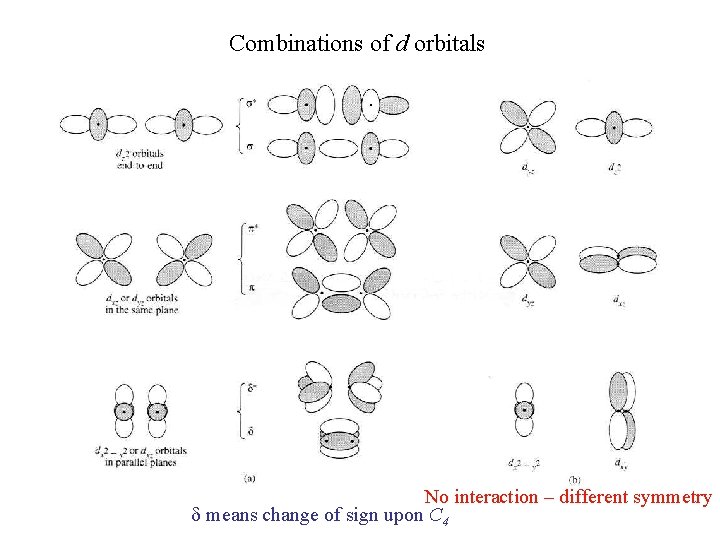
Combinations of d orbitals No interaction – different symmetry d means change of sign upon C 4
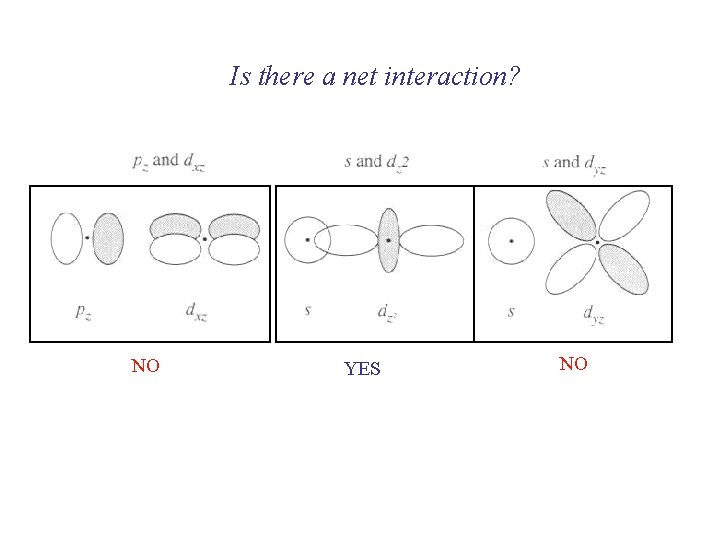
Is there a net interaction? NO YES NO
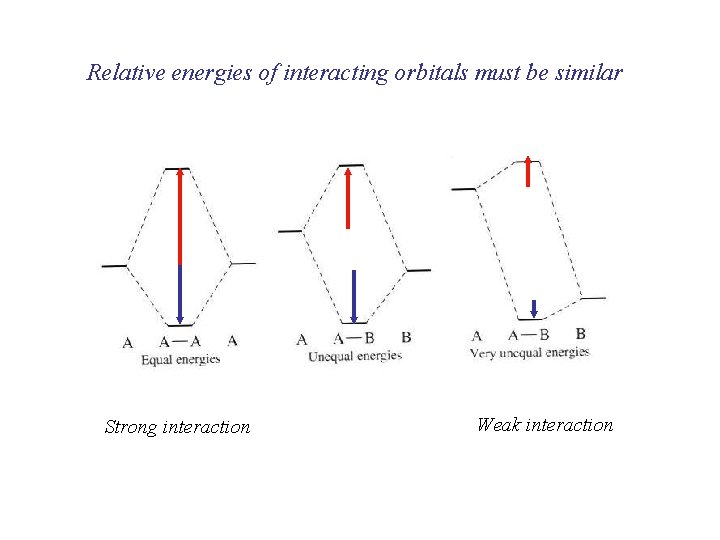
Relative energies of interacting orbitals must be similar Strong interaction Weak interaction
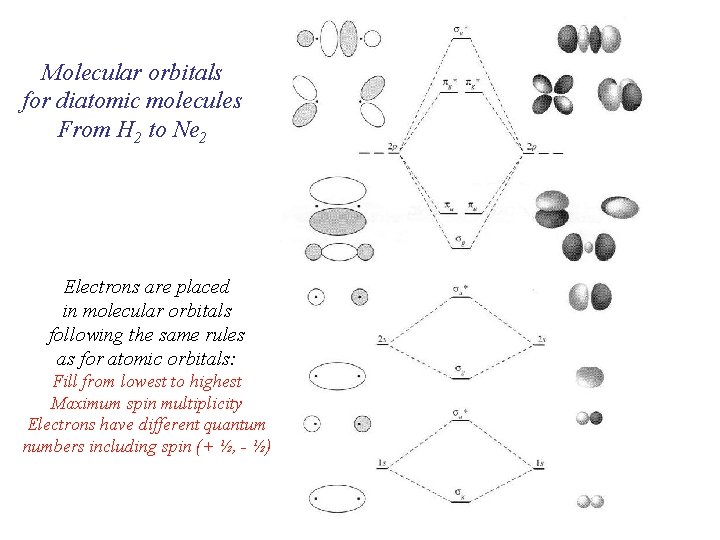
Molecular orbitals for diatomic molecules From H 2 to Ne 2 Electrons are placed in molecular orbitals following the same rules as for atomic orbitals: Fill from lowest to highest Maximum spin multiplicity Electrons have different quantum numbers including spin (+ ½, - ½)

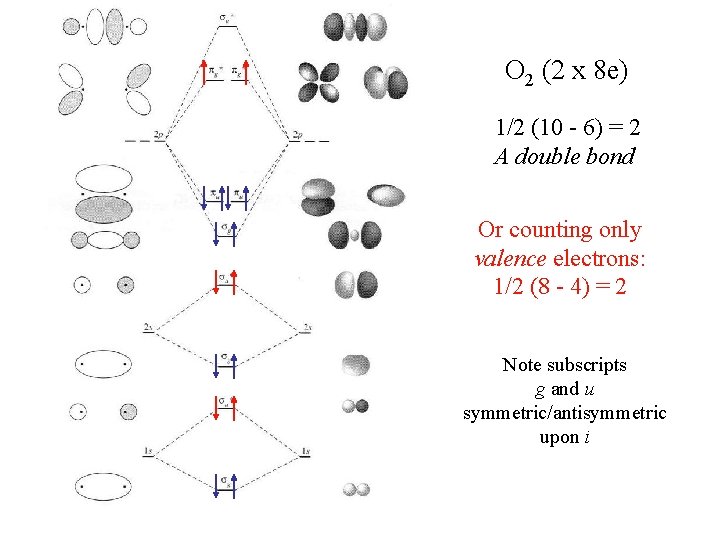
O 2 (2 x 8 e) 1/2 (10 - 6) = 2 A double bond Or counting only valence electrons: 1/2 (8 - 4) = 2 Note subscripts g and u symmetric/antisymmetric upon i
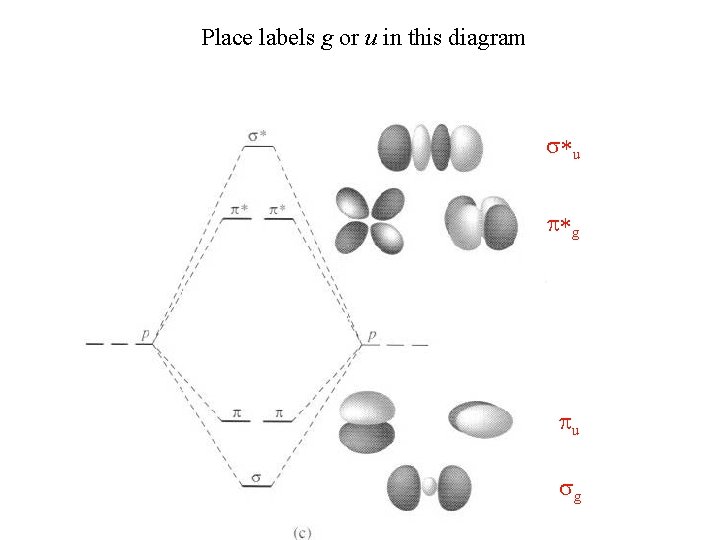
Place labels g or u in this diagram s*u p*g pu sg

s*u g or u? sg p*g pu d*u dg
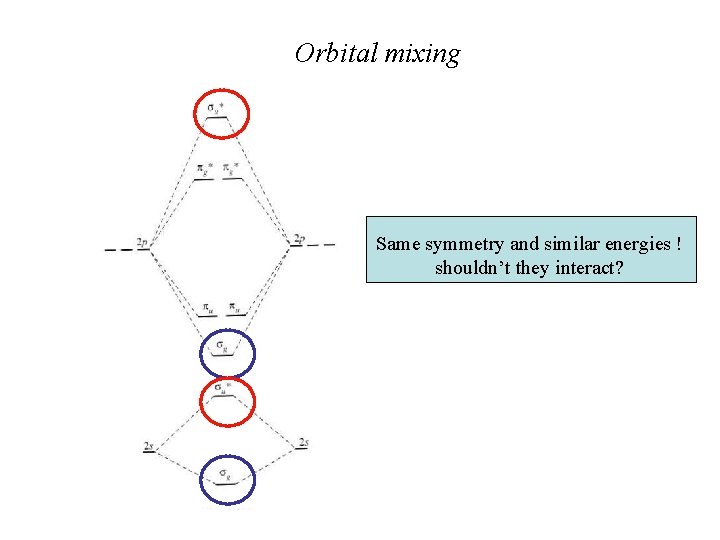
Orbital mixing Same symmetry and similar energies ! shouldn’t they interact?
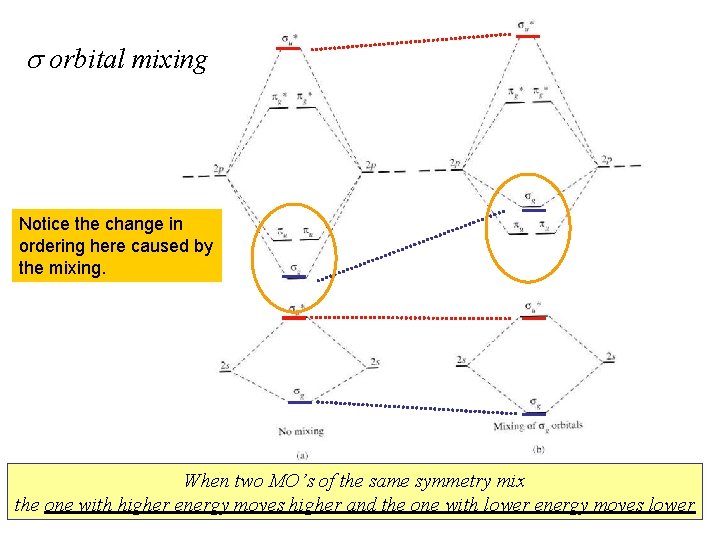
s orbital mixing Notice the change in ordering here caused by the mixing. When two MO’s of the same symmetry mix the one with higher energy moves higher and the one with lower energy moves lower
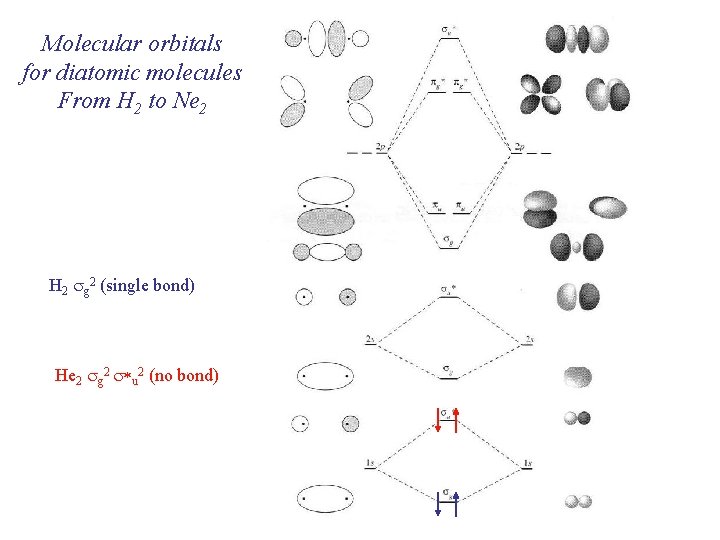
Molecular orbitals for diatomic molecules From H 2 to Ne 2 H 2 sg 2 (single bond) He 2 sg 2 s*u 2 (no bond)
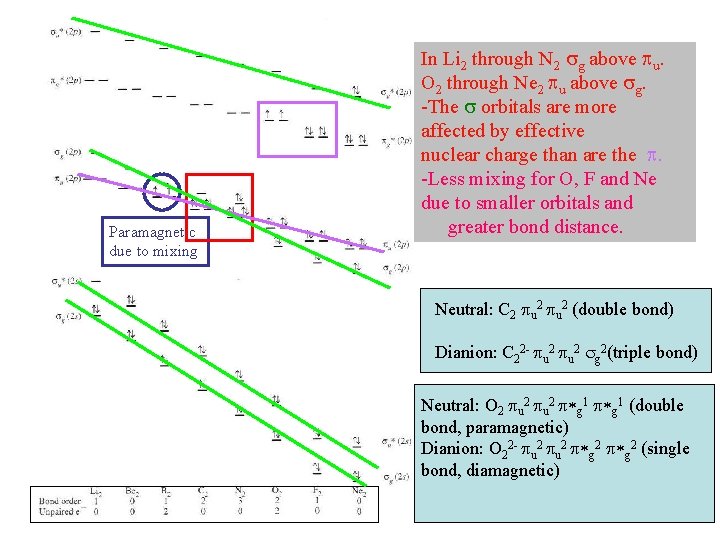
Paramagnetic due to mixing In Li 2 through N 2 sg above pu. O 2 through Ne 2 pu above sg. -The s orbitals are more affected by effective nuclear charge than are the p. -Less mixing for O, F and Ne due to smaller orbitals and greater bond distance. Neutral: C 2 pu 2 (double bond) Dianion: C 22 - pu 2 sg 2(triple bond) Neutral: O 2 pu 2 p*g 1 (double bond, paramagnetic) Dianion: O 22 - pu 2 p*g 2 (single bond, diamagnetic)
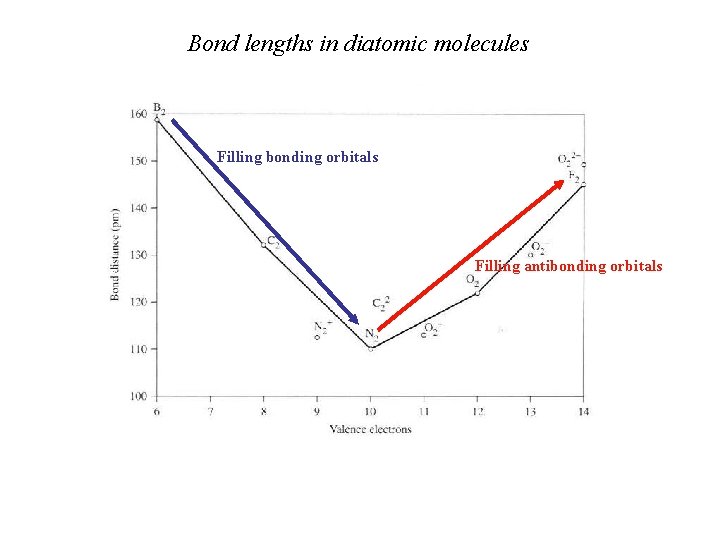
Bond lengths in diatomic molecules Filling bonding orbitals Filling antibonding orbitals
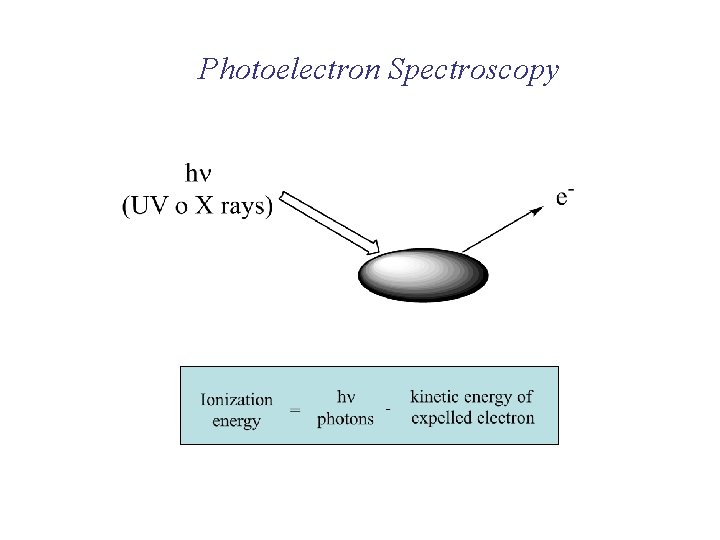
Photoelectron Spectroscopy
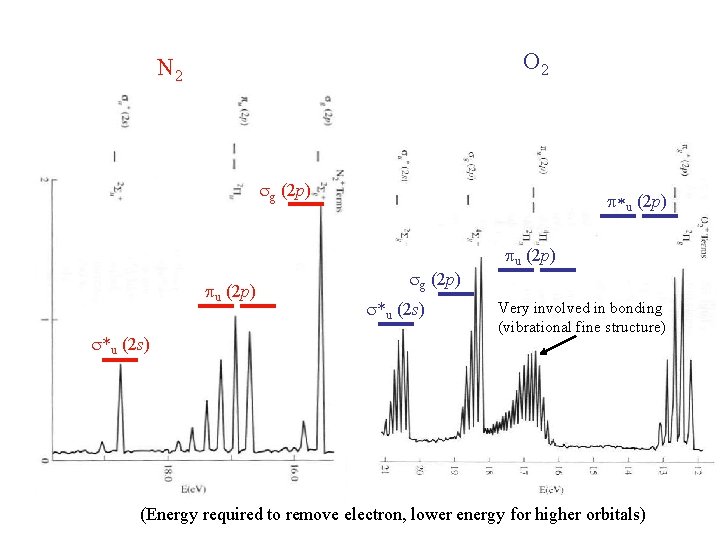
O 2 N 2 sg (2 p) pu (2 p) s*u (2 s) p*u (2 p) sg (2 p) s*u (2 s) pu (2 p) Very involved in bonding (vibrational fine structure) (Energy required to remove electron, lower energy for higher orbitals)
- Slides: 49