Reducing Catheter Related Bloodstream Infections APIC Utah August

Reducing Catheter Related Bloodstream Infections: APIC Utah August 18, 2016 Melissa Steffee RN Stacey Nieporte, VACC Industry Partner 1

• 1. Understanding Scope and Magnitude of catheter-related Bloodstream Infections Objectives • 2. Understanding the Changing Healthcare Landscape • 3. Standardizing The Care of Peripheral Intravenous Catheters • 4. Partnering for Best Patient Outcomes 2
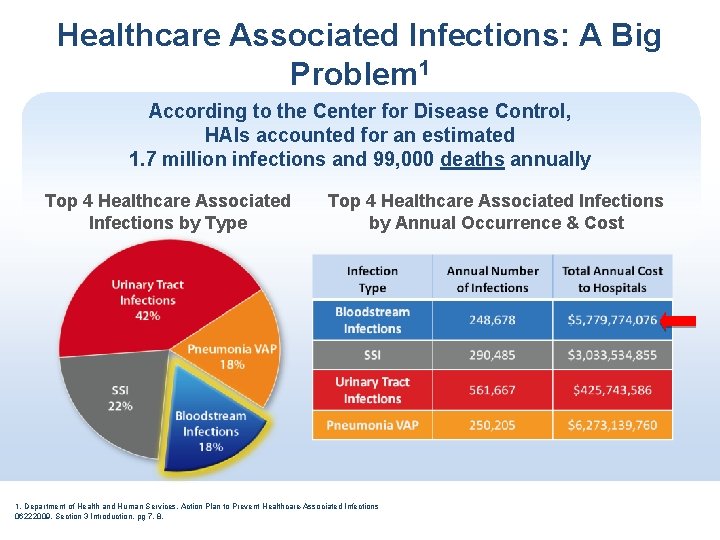
Healthcare Associated Infections: A Big Problem 1 According to the Center for Disease Control, HAIs accounted for an estimated 1. 7 million infections and 99, 000 deaths annually Top 4 Healthcare Associated Infections by Type Top 4 Healthcare Associated Infections by Annual Occurrence & Cost 1. Department of Health and Human Services, Action Plan to Prevent Healthcare-Associated Infections 06222009, Section 3 Introduction, pg 7, 8.

Central Line-associated Bloodstream Infections (CLA-BSI) Impact CLA-BSIs are a significant cause of hospital morbidity and mortality • In the United States, approximately 250, 000 patients will acquire a CLABSI 1 • One third of Healthcareassociated infections (HAIs) were due to CLABSIs with an associated mortality rate of 12. 3%2 4
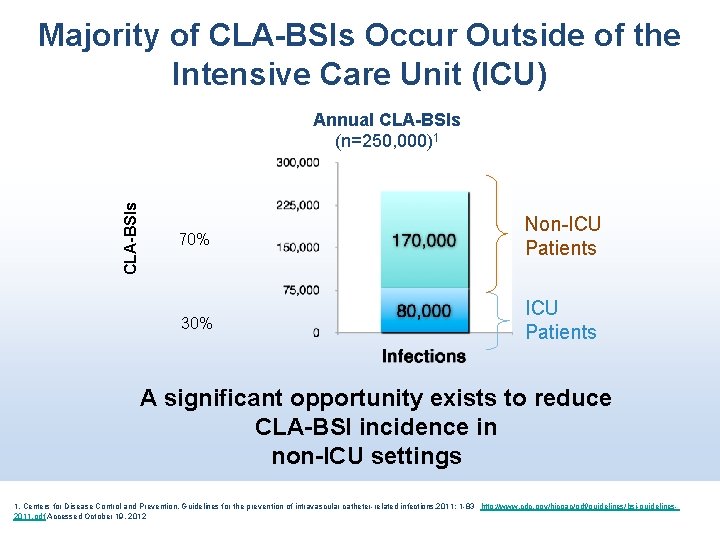
Majority of CLA-BSIs Occur Outside of the Intensive Care Unit (ICU) CLA-BSIs Annual CLA-BSIs (n=250, 000)1 70% Non-ICU Patients 30% ICU Patients A significant opportunity exists to reduce CLA-BSI incidence in non-ICU settings 1. Centers for Disease Control and Prevention. Guidelines for the prevention of intravascular catheter-related infections, 2011; 1 -83 http: //www. cdc. gov/hicpac/pdf/guidelines/bsi-guidelines 2011. pdf Accessed October 19, 2012
What Could Be Causing These Infections? Back To Basics 6
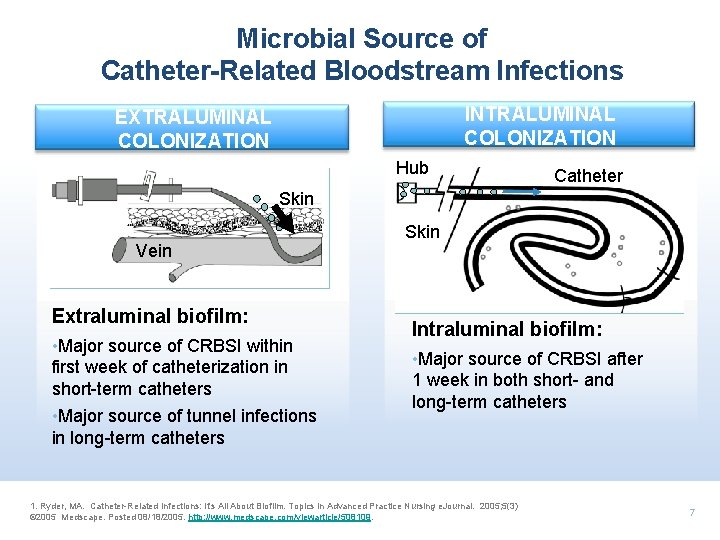
Microbial Source of Catheter-Related Bloodstream Infections INTRALUMINAL COLONIZATION EXTRALUMINAL COLONIZATION Hub Catheter Skin Vein Extraluminal biofilm: • Major source of CRBSI within first week of catheterization in short-term catheters • Major source of tunnel infections in long-term catheters Skin Intraluminal biofilm: • Major source of CRBSI after 1 week in both short- and long-term catheters 1. Ryder, MA. Catheter-Related Infections: It's All About Biofilm. Topics in Advanced Practice Nursing e. Journal. 2005; 5(3) © 2005 Medscape. Posted 08/18/2005. http: //www. medscape. com/viewarticle/508109. 7
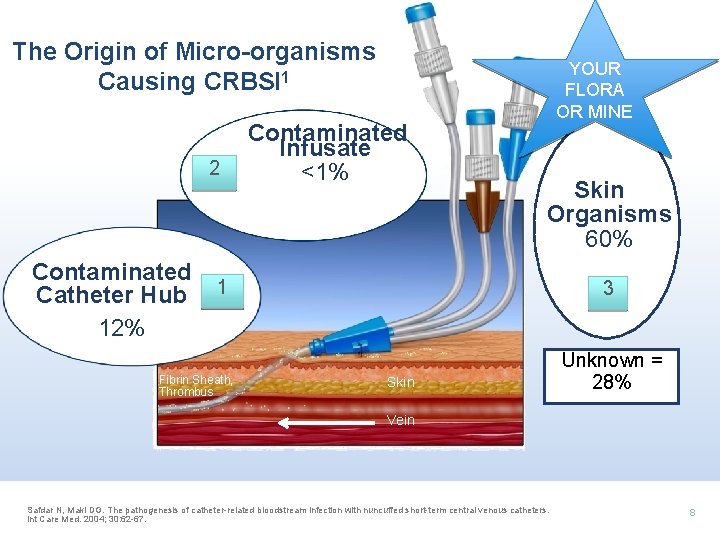
The Origin of Micro-organisms Causing CRBSI 1 YOUR FLORA OR MINE Contaminated 2 Contaminated Catheter Hub 12% Infusate <1% Skin Organisms 60% 1 Fibrin Sheath, Thrombus 3 Skin Unknown = 28% Vein Safdar N, Maki DG. The pathogenesis of catheter-related bloodstream infection with nuncuffed short-term central venous catheters. Int Care Med. 2004; 30: 62 -67. 8
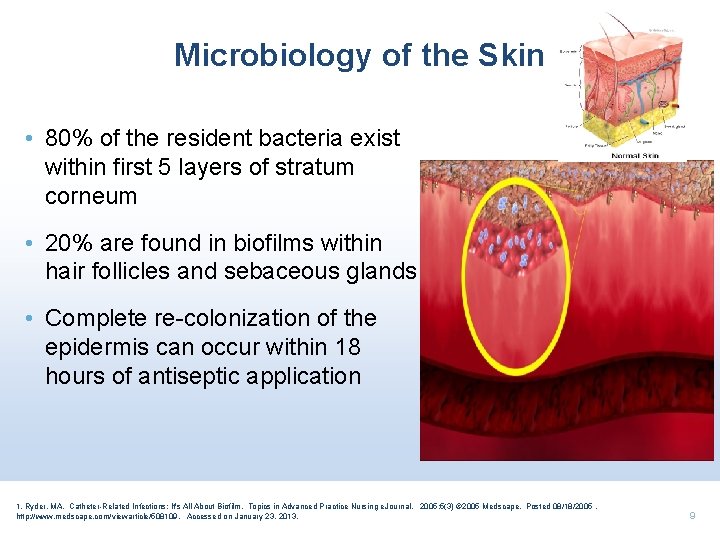
Microbiology of the Skin • 80% of the resident bacteria exist within first 5 layers of stratum corneum • 20% are found in biofilms within hair follicles and sebaceous glands • Complete re-colonization of the epidermis can occur within 18 hours of antiseptic application 1. Ryder, MA. Catheter-Related Infections: It's All About Biofilm. Topics in Advanced Practice Nursing e. Journal. 2005; 5(3) © 2005 Medscape. Posted 08/18/2005. http: //www. medscape. com/viewarticle/508109. Accessed on January 23, 2013. 9
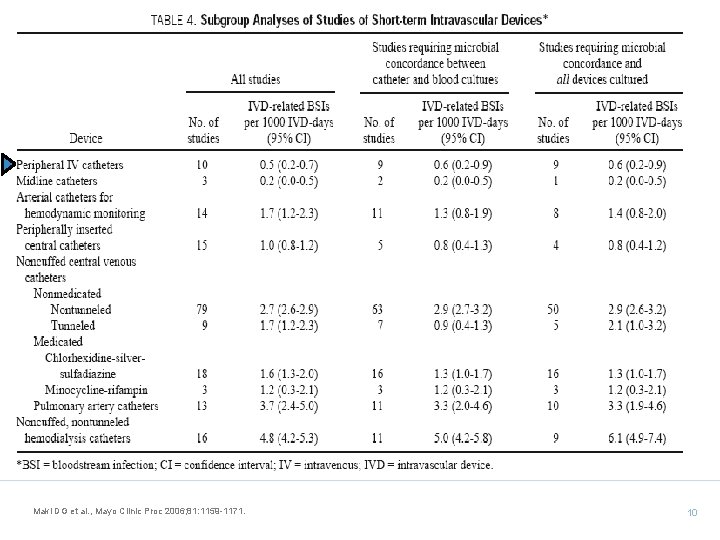
Maki DG et al. , Mayo Clinic Proc 2006; 81: 1159 -1171. 10

Hospital Acquired Staphylococcs aureus primary bloodstream infection: A comparison of events that do and do not meet the central line associated bloodstream infection definition Kovacs et al, 2016, Association for Professionals is Infection Control and Epidemiology

Are Peripheral IVs an Overlooked Source of Infection? 1 12

Right to the Point: PIV Scope and Magnitude • PIVs are the most frequently used invasive device in hospitals • Up to 70% of patients require a PIV during their hospital stay 1 • 330 M peripheral IV catheters are sold in the US each year 1. Zingg W. et al. , Int J Antimicrob Agents 2009; 34 Suppl 4: S 38 -42. 14

Nothing Routine About It: The Patient Experience • 60% of first attempts to insert are unsuccessful 1 • 27% of patients endure 3 or more attempts 1, 2 • 58% of patients report being dissatisfied with the skill of the person starting their peripheral IV 1, 3 • 57% of RNs report that they were not taught how to insert PIVs during nursing school 4 1. Kokotis K. Cost containment and infusion services. J Infusion Nurs. 2005; 28(3 S): S 22 -S 32 2. Barton AJ, Danek G, Johns P, Coons M. Improving patient outcomes through CQI: vascular access planning. J Nurs Care Qual. 1998; 13(2): 77 -85. 3. Wolosin RJ. Largest study of patient satisfaction ever conducted. The Press Ganey Satisfaction Report. August 2003; VII: 2 -4 4. Vizcarra, C. Recommendations for Improving Safety Practices with Short Peripheral Catheters (SPC) Think Safety, Insert Safely. INS Safety Practice Survey. 2013 15

What About Infection? BSI related to PIV • 0. 2 -0. 7 per 1, 000 device-days infection rate 1 • Population is so large that the number of patients potentially affected is actually quite significant • This risk exists with or without extended dwell times q Vascular Catheters are the single most common source of bacteremia and fungemia 2 q An estimated 5% to 25% of peripheral catheters were colonized with bacteria at the time of removal 3 q As many as 10, 000 Staphylococcus aureus bacteremias from peripheral catheters annually in the United States 3 1. Trinh, et al. Peripheral Venous Catheter-Related Staphylococcus aureus Bacteremia. Infect Control Hosp Epidemiol 2011; 32(6): 579 -583 2. Pien BC, Sundaram P, Raoof N, et al. The clinical and prognostic importance of positive blood cultures in adults. Am J Med. 2010; 123(9): 819 -828 3. Short Peripheral Intravenous Catheters and Infections Lynn Hadaway MEd, RN, BC, CRNI® Journal of Infusion Nursing, August 2012 Vol 35: 4 16

CDC Recommendation Periodically assess knowledge of and adherence to guidelines for all personnel involved in the insertion and maintenance of intravascular catheters. Category IA • “There is no need to replace peripheral catheters more frequently than every 7296 hours to reduce risk of infection and phlebitis in adults [36, 140, 141]. Category 1 B” • “No recommendation is made regarding replacement of peripheral catheters in adults only when clinically indicated [142– 144]. Unresolved issue” • “Some studies have suggested that planned removal at 72 hours vs. removing as needed resulted in similar rates of phlebitis and catheter failure [142– 144]. However, these studies did not address the issue of CRBSI, and the risk of CRBSIs with this strategy is not well studied. ” O'Grady, N. P. , et al. Guidelines for the Prevention of Intravascular Catheter-Related Infections. American Journal of Infection Control. 2011; 39 (4 Suppl 1): S 1 -34. 22

INS Standards • Routine site care and dressing changes not performed on short peripheral catheters unless the dressing is soiled or no longer intact • The nurse should consider replacement of the short peripheral catheter when clinically indicated and when infusion treatment does not include peripheral parenteral nutrition • The nurse should not routinely replace short peripheral catheters in pediatric patients Infusion Nursing Standard of Practice, Journal of Infusion Nursing. 2011; (34) 1 S 23

Benefits of Longer Dwell Fewer Invasive Procedures • Improved patient experience • Fewer breaches in skin • Increased nursing efficiency • Reduction in material costs • Vein preservation Regardless of dwell time, risks are still associated with PIVs 24

Moving the Needle: One Standard of Care Central Line Care Peripheral IV Care Sterile Alcohol Scrub the Transparent Impregnated Hub Prior to Dressing Caps Access Chlorhexidine Solution Skin Prep Flushing Protocols BIOPATCH ® Protective Disk with CHG Solution Skin Prep Flushing Protocols BIOP A Protec TCH ® tive D isk CHG with 31

Evidence You Should Ask for… ion t a I c i S d B n I CR d f e r o a le on i C t c u ü ed R r o f of l e v Le t s ies e d h u t g i S ü H idence/ Ev ine l e d i u G l ns a o n i t o i a t a nd N e m ü m Reco 32

§ Products on the market will have an FDA Cleared Indication. § It is important to understand exactly what is the intended use for the product § Read the Product Insert, under “Indication for Use” FDA Cleared Indications

§ FDA MAUDE (Manufacturer & User Facility Device Experience) § http: //www. accessdata. fda. gov/scripts/cdrh/cfdocs/cf MAUDE/search. CFM § Search all of the FDA § http: //www. fda. gov/Medic al. Devices/Safety/Alertsand Notices/ucm 181502. htm FDA Resources
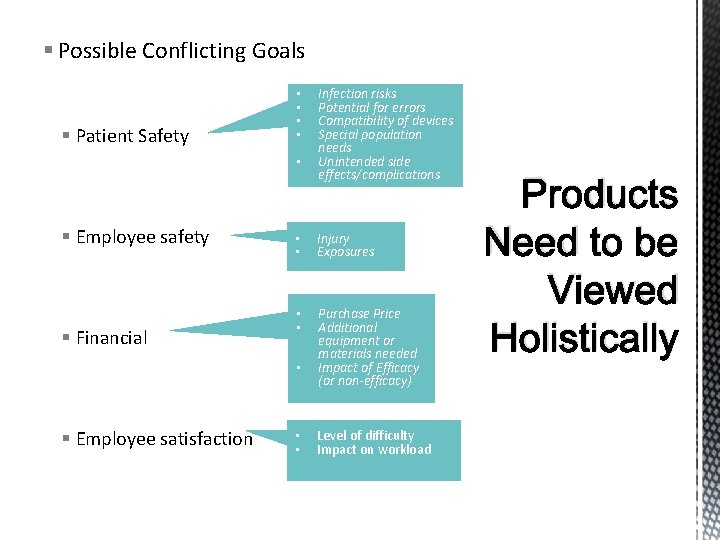
§ Possible Conflicting Goals § Patient Safety • • • § Employee safety § Financial • • Injury Exposures • • Purchase Price Additional equipment or materials needed Impact of Efficacy (or non-efficacy) • § Employee satisfaction Infection risks Potential for errors Compatibility of devices Special population needs Unintended side effects/complications • • Level of difficulty Impact on workload Products Need to be Viewed Holistically

§ What are the legal/safety/ quality implications for using/not using this product? § What is the greatest priority for my staff? § Is there an increased risk of exposure/harm to staff? § How is it better than what we use today? § Who else is using it? § Who has stopped using it and why? What are the educational needs for staff? § Where else will the patient go with the device? § Both in house (areas of hospital) and post discharge Impact on Facility/ Staff

§ What is the quality of the product? § FDA approved indication § Review recalls and FDA citations § Does this address the greatest threat of real harm to my patients? § Are there potential unintended side effects/complications of using this product? Impact on Patient Outcomes
- Slides: 25