Redox Geochemistry J Willard Gibbs Gibbs realized that

Redox Geochemistry

J. Willard Gibbs • Gibbs realized that for a reaction, a certain amount of energy goes to an increase in entropy of a system. • G = H –TS or DG 0 R = DH 0 R – TDS 0 R • Gibbs Free Energy (G) is a state variable, measured in KJ/mol or Cal/mol • Tabulated values of DG 0 R available…

Equilibrium Constant • for a. A + b. B c. C + d. D: • Restate the equation as: DGR = DG 0 R + RT ln Q • DGR= available metabolic energy (when negative = exergonic process as opposed to endergonic process for + energy) for a particular reaction whose components exist in a particular concentration

Activity • Activity, a, is the term which relates Gibbs Free Energy to chemical potential: mi-G 0 i = RT ln ai • Why is there now a correction term you might ask… – Has to do with how things mix together – Relates an ideal solution to a non-ideal solution

Ions in solution • Ions in solutions are obviously nonideal states! • Use activities (ai) to apply thermodynamics and law of mass action a i = g i mi • The activity coefficient, gi, is found via some empirical foundations

Activity Coefficients • Extended Debye-Huckel approximation (valid for I up to 0. 5 M): • Where A and B are constants (tabulated), and a is a measure of the effective diameter of the ion (tabulated)

Speciation • Any element exists in a solution, solid, or gas as 1 to n ions, molecules, or solids • Example: Ca 2+ can exist in solution as: Ca++ Ca(H 3 Si. O 4)2 Ca(O-phth) Ca. B(OH)4+ Ca. CH 3 COO+ Ca. CO 30 Ca. Cl+ Ca. F+ Ca. H 2 Si. O 4 Ca. H 3 Si. O 4+ Ca. HCO 3+ Ca. NO 3+ Ca. OH+ Ca. PO 4 Ca. SO 4 Ca. HPO 40 • Plus more species gases and minerals!!
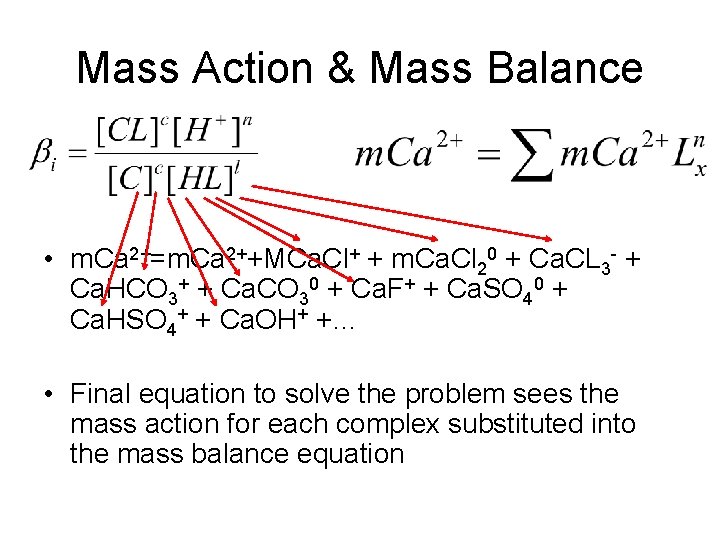
Mass Action & Mass Balance • m. Ca 2+=m. Ca 2++MCa. Cl+ + m. Ca. Cl 20 + Ca. CL 3 - + Ca. HCO 3+ + Ca. CO 30 + Ca. F+ + Ca. SO 40 + Ca. HSO 4+ + Ca. OH+ +… • Final equation to solve the problem sees the mass action for each complex substituted into the mass balance equation

Geochemical models • Hundreds of equations solved iteratively for speciation, solve for DGR • All programs work on same concept for speciation thermodynamics and calculations of mineral equilibrium – lots of variation in output, specific info…

Oxidation – Reduction Reactions • • R E Oxidation - a process involving loss of electrons. Reduction - a process involving gain of electrons. Reductant - a species that loses electrons. Oxidant - a species that gains electrons. G s y a s • Free electrons do not exist in solution. Any electron lost from one species in solution must be immediately gained by another. Ox 1 + Red 2 Red 1 + Ox 2 L O E

Half Reactions • Often split redox reactions in two: – oxidation half rxn e- leaves left, goes right • Fe 2+ Fe 3+ + e- – Reduction half rxn e- leaves left, goes right • O 2 + 4 e - 2 H 2 O • SUM of the half reactions yields the total redox reaction 4 Fe 2+ 4 Fe 3+ + 4 e. O 2 + 4 e - 2 H 2 O 4 Fe 2+ + O 2 4 Fe 3+ + 2 H 2 O

Half-reaction vocabulary part II • Anodic Reaction – an oxidation reaction • Cathodic Reaction – a reduction reaction • Relates the direction of the half reaction: • A A+ + e- == anodic • B + e- B- == cathodic

ELECTRON ACTIVITY • Although no free electrons exist in solution, it is useful to define a quantity called the electron activity: • The pe indicates the tendency of a solution to donate or accept a electron. • If pe is low, there is a strong tendency for the solution to donate electron - the solution is reducing. • If pe is high, there is a strong tendency for the solution to accept electron - the solution is oxidizing.

THE pe OF A HALF REACTION - I Consider the half reaction Mn. O 2(s) + 4 H+ + 2 e- Mn 2+ + 2 H 2 O(l) The equilibrium constant is Solving for the electron activity

DEFINITION OF Eh Eh - the potential of a solution relative to the SHE. Both pe and Eh measure essentially the same thing. They may be converted via the relationship: Where = 96. 42 k. J volt-1 eq-1 (Faraday’s constant). At 25°C, this becomes or

Free Energy and Electropotential • Talked about electropotential (aka emf, Eh) driving force for e- transfer • How does this relate to driving force for any reaction defined by DGr ? ? DGr = - n E – Where n is the # of e-’s in the rxn, is Faraday’s constant (23. 06 cal V-1), and E is electropotential (V) • pe for an electron transfer between a redox couple analagous to p. K between conjugate acid-base pair

Electropotentials • E 0 is standard electropotential, also standard reduction potential (write rxn as a reduction ½ rxn) – EH is relative to SHE (Std Hydrogen Electrode) At non-standard conditions: At 25° C:

Electromotive Series • When we put two redox species together, they will react towards equilibrium, i. e. , e- will move which ones move electrons from others better is the electromotive series • Measurement of this is through the electropotential for half-reactions of any redox couple (like Fe 2+ and Fe 3+) – Because DGr =-n E, combining two half reactions in a certain way will yield either a + or – electropotential (additive, remember to switch sign when reversing a rxn) +E - DGr, therefore spontaneous • In order of decreasing strength as a reducing agent strong reducing agents are better e- donors
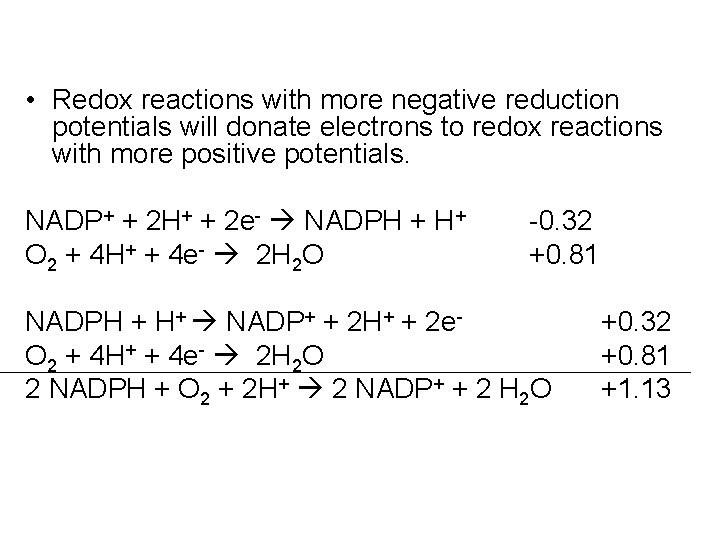
• Redox reactions with more negative reduction potentials will donate electrons to redox reactions with more positive potentials. NADP+ + 2 H+ + 2 e- NADPH + H+ O 2 + 4 H+ + 4 e- 2 H 2 O -0. 32 +0. 81 NADPH + H+ NADP+ + 2 H+ + 2 e. O 2 + 4 H+ + 4 e- 2 H 2 O 2 NADPH + O 2 + 2 H+ 2 NADP+ + 2 H 2 O +0. 32 +0. 81 +1. 13
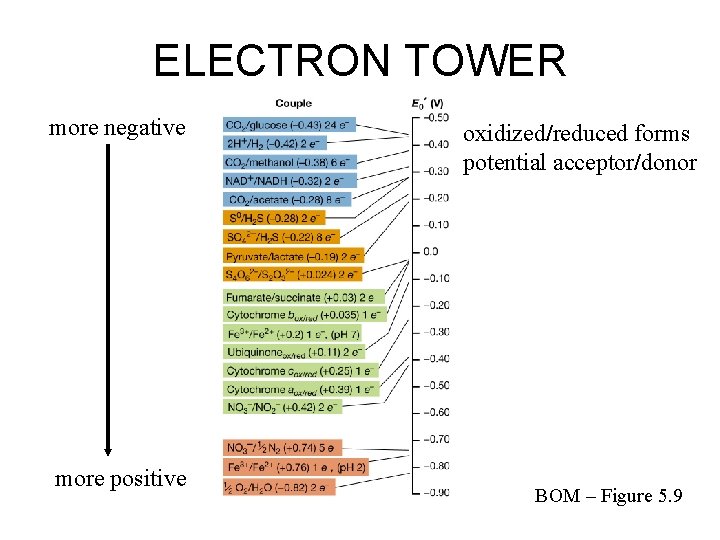
ELECTRON TOWER more negative more positive oxidized/reduced forms potential acceptor/donor BOM – Figure 5. 9
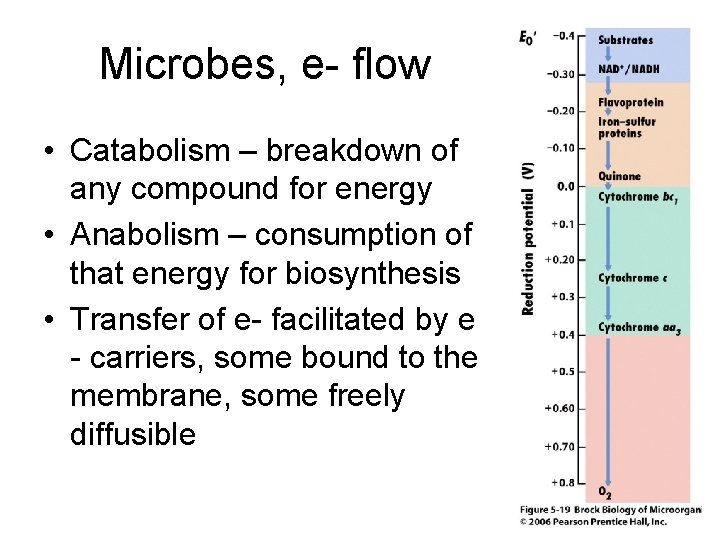
Microbes, e- flow • Catabolism – breakdown of any compound for energy • Anabolism – consumption of that energy for biosynthesis • Transfer of e- facilitated by e - carriers, some bound to the membrane, some freely diffusible
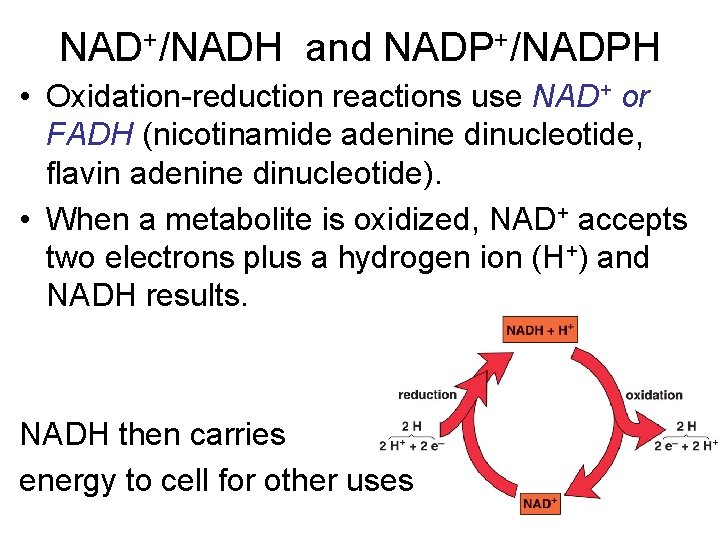
NAD+/NADH and NADP+/NADPH • Oxidation-reduction reactions use NAD+ or FADH (nicotinamide adenine dinucleotide, flavin adenine dinucleotide). • When a metabolite is oxidized, NAD+ accepts two electrons plus a hydrogen ion (H+) and NADH results. NADH then carries energy to cell for other uses
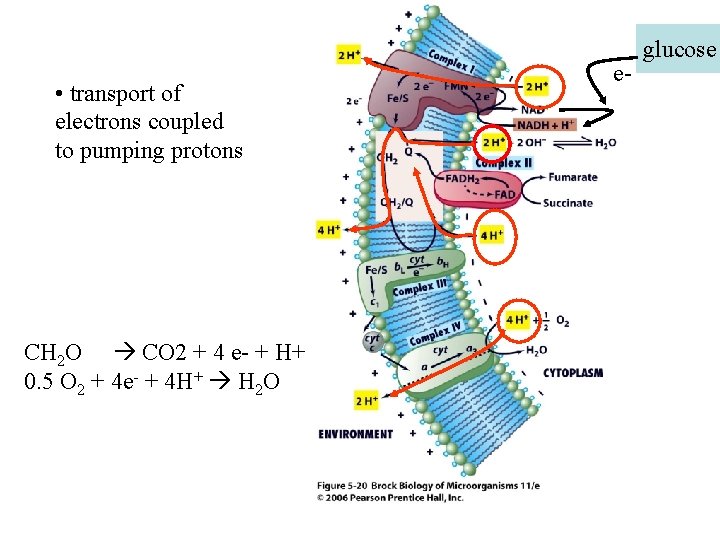
• transport of electrons coupled to pumping protons CH 2 O CO 2 + 4 e- + H+ 0. 5 O 2 + 4 e- + 4 H+ H 2 O e- glucose

Proton Motive Force (PMF) • Enzymatic reactions pump H+ outside the cell, there a number of membranebound enzymes which transfer e-s and pump H+ out of the cell • Develop a strong gradient of H+ across the membrane (remember this is 8 nm thick) • This gradient is CRITICAL to cell function because of how ATP is generated…
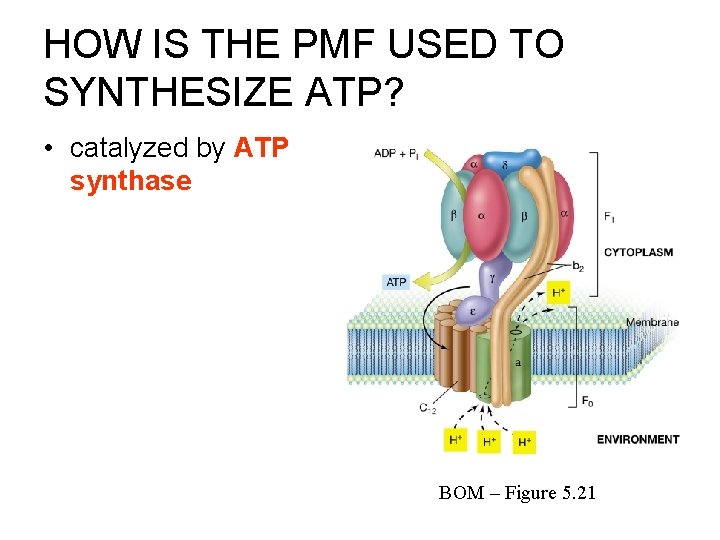
HOW IS THE PMF USED TO SYNTHESIZE ATP? • catalyzed by ATP synthase BOM – Figure 5. 21

ATP generation II • Alternative methods to form ATP: • Phosphorylation coupled to fermentation, low yield of ATP

ATP • Your book says ATP: “Drives thermodynamically unfavorable reactions” BULLSHIT, this is impossible • The de-phosphorylation of ATP into ADP provides free energy to drive reactions!

Minimum Free Energy for growth • Minimum free energy for growth = energy to make ATP? • What factors go into the energy budget of an organism? ?

Growth Efficiency • How much energy does it take to grow a new microbe? • How much energy does a microbe gain from any metabolic reaction? • How much energy is ‘wasted’, i. e. , how much energy does it ‘cost’ the microbe to hang out in it’s environment that is not directly attributed to the energy required for growth and division…
- Slides: 30