Rectal Microbicide Protocol Status Need for Rectal Microbicides

Rectal Microbicide Protocol Status

Need for Rectal Microbicides • Oral Pr. EP is not for everyone – Just as there are choices for contraception based on needs and lifestyles, options are needed for HIV prevention • Could be used around the time of sex (instead of daily) • Wide range of formulations being developed & tested – Gels (with/without an applicator) – Enemas – Rectal inserts (fast-dissolving rectal tablets) – Suppositories – Other non-antiretroviral-based products

The Rectal Road • 7 drugs and 4 formulations being tested as rectal microbicides (RMs) by MTN and others • 2 MTN RM studies underway, 1 expecting results and 2 more launching in 2019
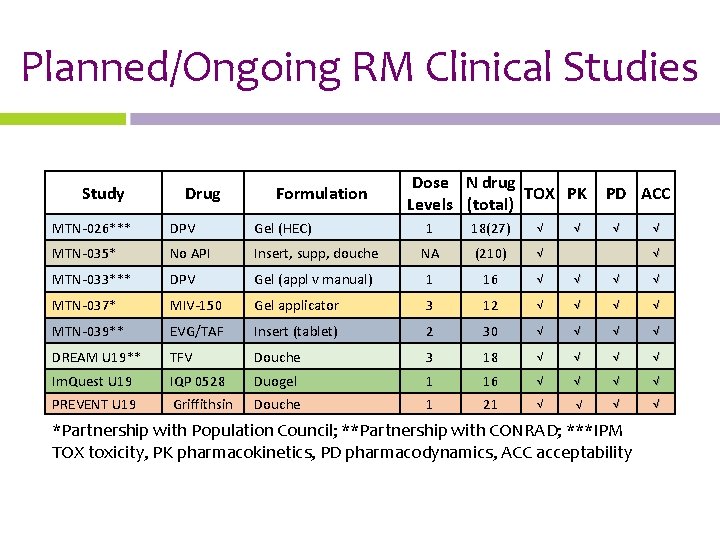
Planned/Ongoing RM Clinical Studies Study Drug Formulation MTN-026*** DPV Gel (HEC) MTN-035* No API MTN-033*** Dose N drug TOX PK Levels (total) PD ACC 1 18(27) √ Insert, supp, douche NA (210) √ DPV Gel (appl v manual) 1 16 √ √ MTN-037* MIV-150 Gel applicator 3 12 √ √ MTN-039** EVG/TAF Insert (tablet) 2 30 √ √ DREAM U 19** TFV Douche 3 18 √ √ Im. Quest U 19 PREVENT U 19 IQP 0528 Griffithsin Duogel Douche 1 1 16 21 √ √ √ *Partnership with Population Council; **Partnership with CONRAD; ***IPM TOX toxicity, PK pharmacokinetics, PD pharmacodynamics, ACC acceptability
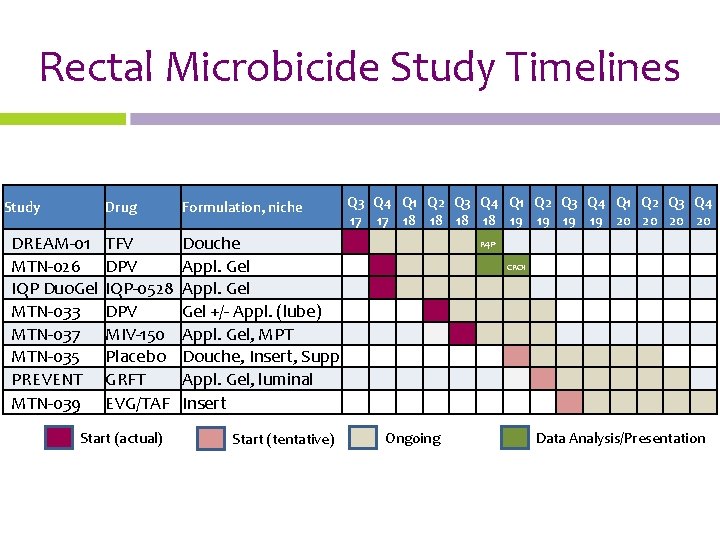
Rectal Microbicide Study Timelines Study DREAM-01 MTN-026 IQP Duo. Gel MTN-033 MTN-037 MTN-035 PREVENT MTN-039 Drug Formulation, niche TFV DPV IQP-0528 DPV MIV-150 Placebo GRFT EVG/TAF Douche Appl. Gel Gel +/- Appl. (lube) Appl. Gel, MPT Douche, Insert, Supp Appl. Gel, luminal Insert Start (actual) Start (tentative) Q 3 Q 4 Q 1 Q 2 Q 3 Q 4 17 17 18 18 19 19 20 20 R 4 P CROI Ongoing Data Analysis/Presentation

MTN-037 Study • Will multi-purpose antiviral achieve target concentrations? • • • HIV Phase I MIV-150 / Carrageenan / Zinc gel Blocks HIV, HSV, & HPV in lab & animals HSV HPV Men & women (cis & transgender) (N=24) Single dose, volume escalation 4 m. L to 32 m. L Safety, acceptability, drug conc’n, tissue protection Population Council Collaboration (also vaginal product)

MTN-039 Study • Is a rectal insert feasible as rectal microbicide? Phase 1 Elvitegravir/TAF insert Men & women (cis & transgender) (N = 20) Single dose at two dose levels Safety, acceptability, drug concentration, biopsy susceptibility to infection • CONRAD collaboration (also vaginal product) • •

Thank You!
- Slides: 8