Recrystallization Impure benzoic acid Benzoic acid after recrystallization

Recrystallization Impure benzoic acid Benzoic acid after recrystallization
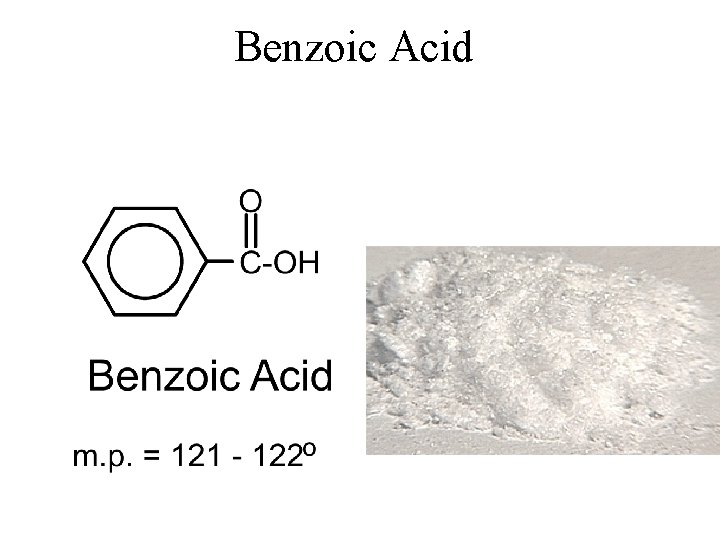
Benzoic Acid

Purification of Organic Compounds Solids: Recrystallization Liquids: Distillation Solids and Liquids: Gas or liquid chromotography

Types of impurities Soluble Insoluble Partly Soluble
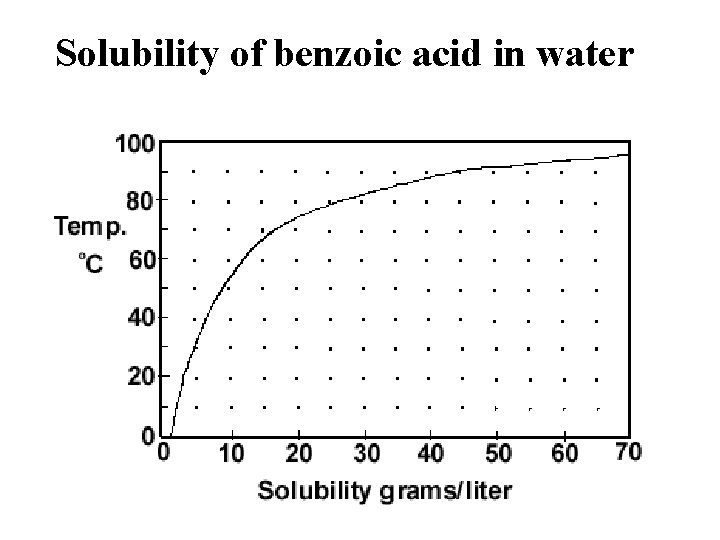
Solubility of benzoic acid in water
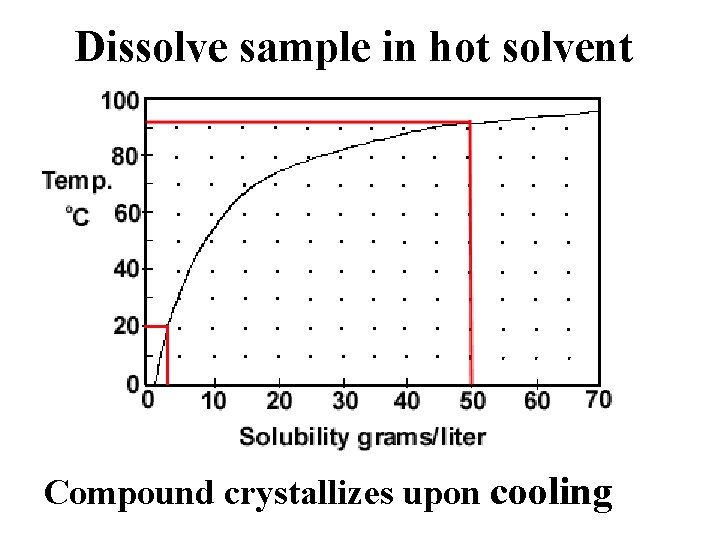
Dissolve sample in hot solvent Compound crystallizes upon cooling

Impure benzoic acid in hot water

Dissolve Add water and heat until all soluble material dissolves Insoluble material is removed by filtration

Filter Hot The hot solution is filtered to remove insoluble impurities

Fluted Filter Paper Maximize surface area.

To avoid crystallization in the funnel: Use GLASS funnel Put flask on hot plate.

Boil off excess solvent

Boiling Sticks

Hot solution after filtration: Cool Seed Scratch

Crystals form in the warm solution Next: Cool, filter, wash, dry, weigh, mp

Cool
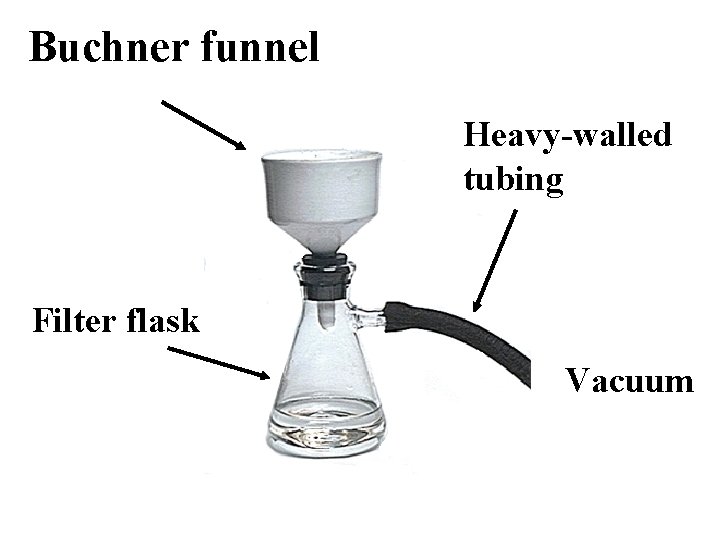
Buchner funnel Heavy-walled tubing Filter flask Vacuum

. . Put filter paper in Buchner funnel

Filter and wash with cold solvent
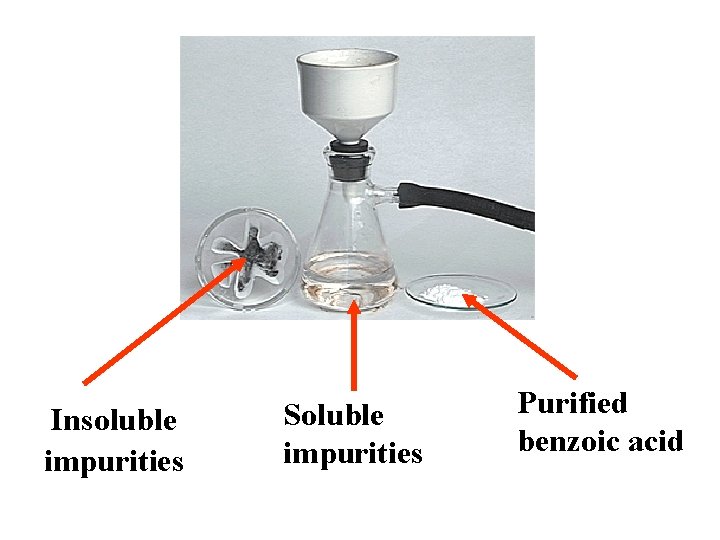
Insoluble impurities Soluble impurities Purified benzoic acid
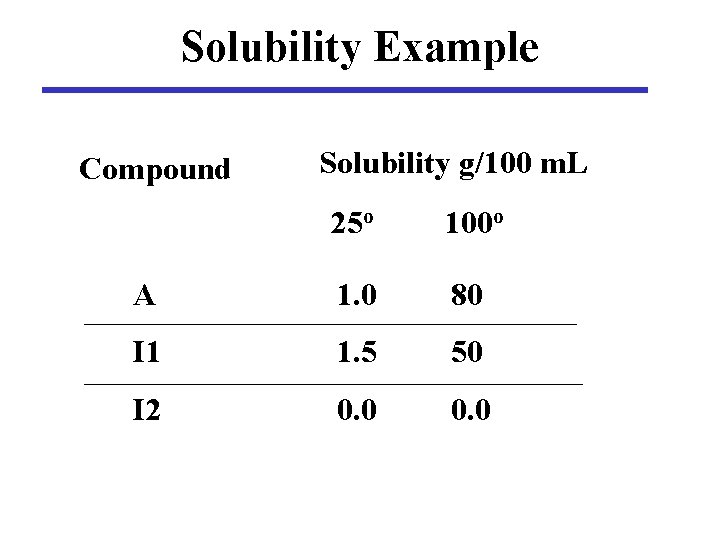
Solubility Example Compound Solubility g/100 m. L 25 o 100 o A 1. 0 80 I 1 1. 5 50 I 2 0. 0

Compound Solubility g/100 m. L A 25 o 1. 0 100 o 80 I 1 1. 5 50 I 2 0. 0 A = 10 g I 1 = 1. 0 g I 2 = 1. 0 g Total = 12. 0 g 100 m. L water at 1000 A dissolves I 1 dissolves I 2 insoluble

Compound Solubility g/100 m. L A 25 o 1. 0 100 o 80 I 1 1. 5 50 I 2 0. 0 A = 10 g I 1 = 1. 0 g I 2 = 1. 0 g Total = 12. 0 g 100 m. L water at 1000 Cool to 25 o A dissolves A 9. 0 g crystallize I 1 dissolves I 1 0 g crystallize I 2 insoluble Yield = 100 * 9. 0/12. 0 = 75%

Compound Solubility g/100 m. L A 25 o 1. 0 100 o 80 I 1 1. 5 50 I 2 0. 0 A = 10 g I 1 = 1. 0 g I 2 = 1. 0 g Total = 12. 0 g 50 ml of water at 1000 A dissolves I 1 dissolves I 2 insoluble

Compound Solubility g/100 m. L A 25 o 1. 0 100 o 80 I 1 1. 5 50 I 2 0. 0 A = 10 g I 1 = 1. 0 g I 2 = 1. 0 g Total = 12. 0 g Cool to 25 o 50 m. L water at 1000 A dissolves A 9. 5 g crystallize I 1 dissolves I 1 0. 25 g crystallize I 2 insoluble Yield = 100 * 9. 75/12. 0 = 81% NOT PURE!

Maximize Yield What volume of solvent is needed to give the maximum yield of pure A? Solubility of I 1 = 1. 5 g/100 m. L 1. 5 g = 100 m. L 1. 0 g x m. L X = (100 m. L * 1. 0 g)/1. 5 g = 67 m. L

Maximize Yield What is the yield of pure A from 67 m. L solvent? Solubility of A = 1. 0 g/100 m. L 1. 0 g = 100 m. L Xg 67 m. L X = (67 m. L x 1. 0 g) / 100 m. L = 0. 67 g Yield = 10. 0 g - 0. 67 g = 9. 33 g % yield = 100 * 9. 33 / 12 = 78%

Second Crop Boil off some solvent Cool, filter, wash, dry, weigh, mp
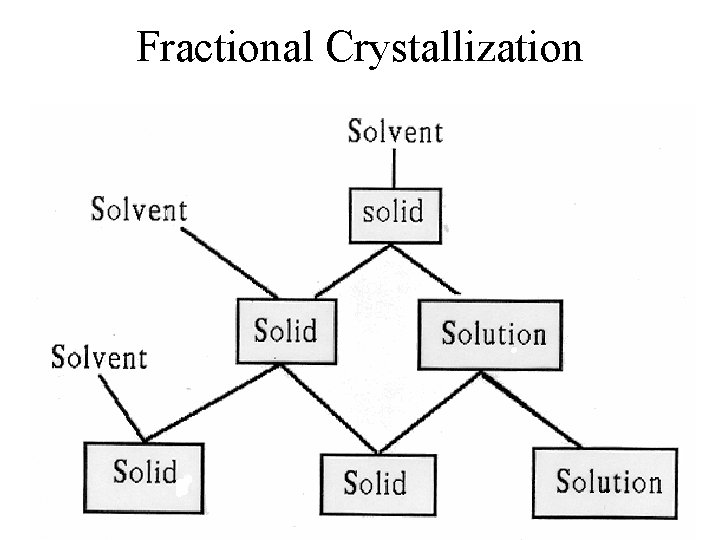
Fractional Crystallization
- Slides: 29