Recognising materials substances and elements DATE Keywords Learning

Recognising materials, substances and elements DATE Keywords: Learning Objectives • Recognise the difference between materials, substances and elements • Identify elements by their names and symbols • Explain what is meant by a chemically pure substance DO IT NOW : Recap from primary school: 1. List 5 different materials 2. Next to each material, write at least ONE property of that material (e. g. strong, light, shiny)

Task: Key terms Materials: Combinations of different substances (e. g. brick, wool, plastic) Substance: something which is made of matter / particles (e. g. carbon dioxide, glass, sugar) Elements: the simplest form of a chemical. Elements are made of only one type of particle (e. g. iron, gold, oxygen)

Task: Key terms The Elements Game Take out your planners, and turn to your periodic table. Your teacher will read out either: a. the element NAME (e. g. hydrogen), or b. the element SYMBOL (e. g. Fe) You need to find the element in your periodic table, and then write down the name and symbol in your books (e. g. Hydrogen – H)
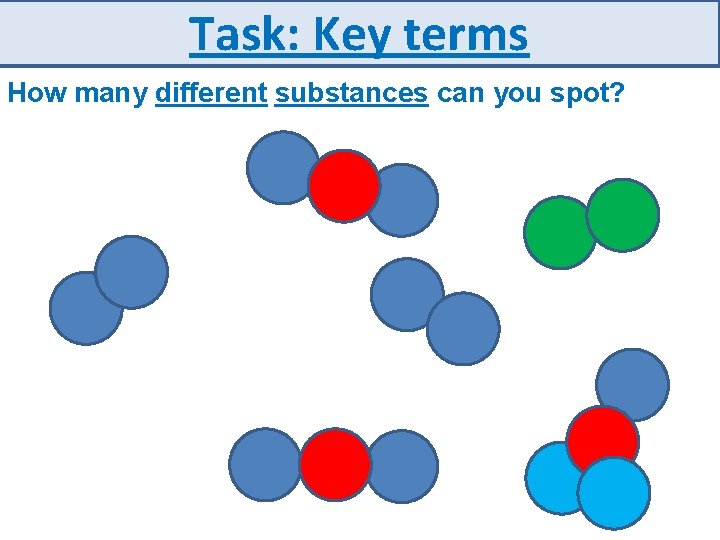
Task: Key terms How many different substances can you spot?
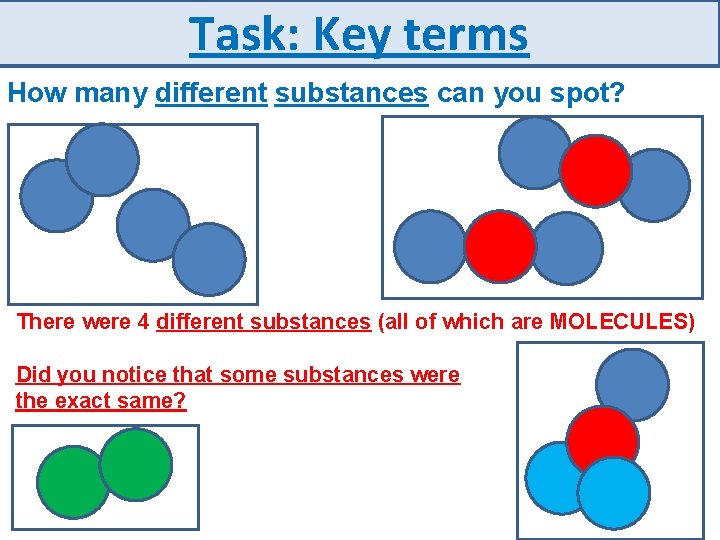
Task: Key terms How many different substances can you spot? There were 4 different substances (all of which are MOLECULES) Did you notice that some substances were the exact same?
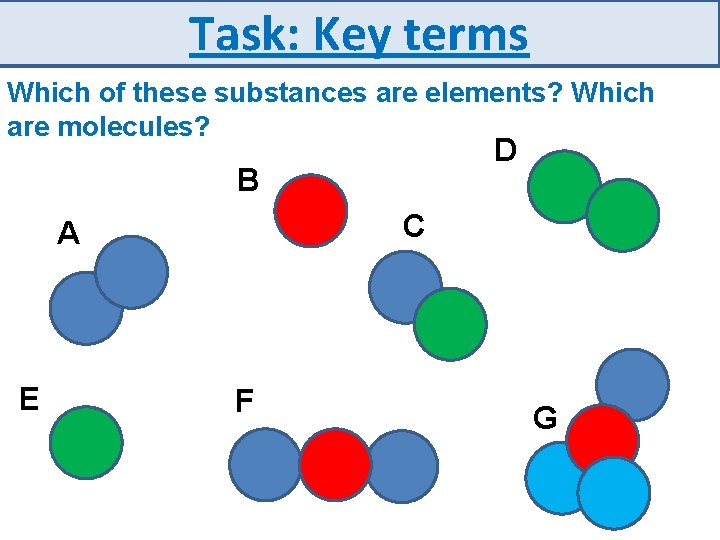
Task: Key terms Which of these substances are elements? Which are molecules? D B C A E F G
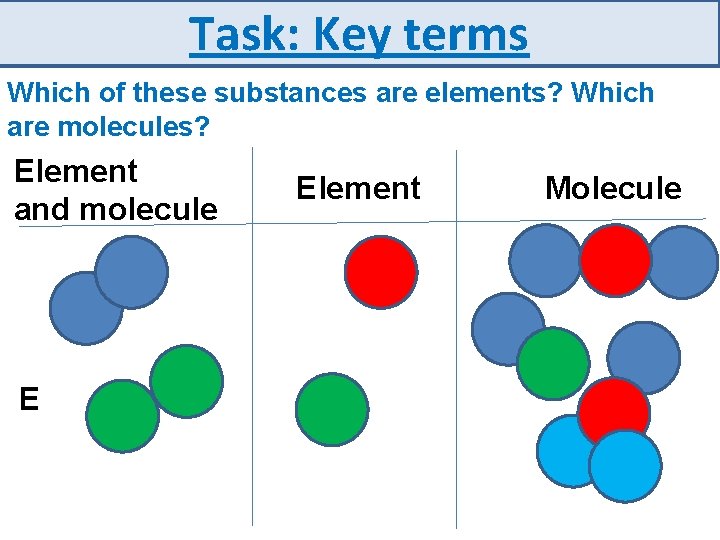
Task: Key terms Which of these substances are elements? Which are molecules? Element and molecule E Element Molecule
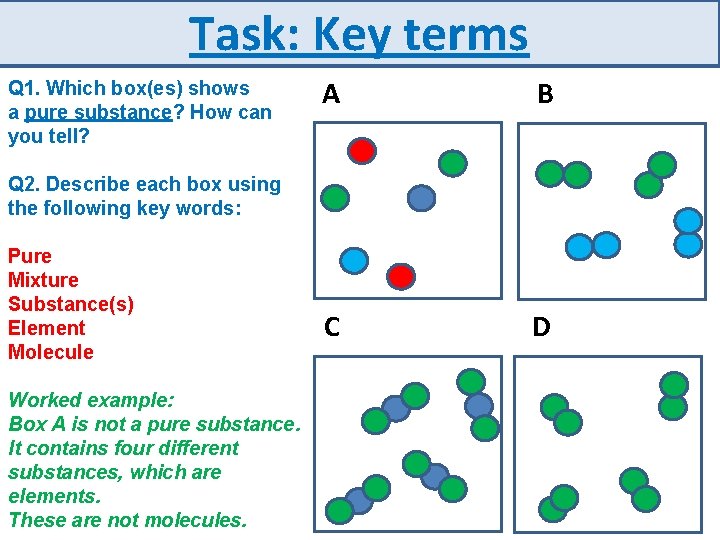
Task: Key terms Q 1. Which box(es) shows a pure substance? How can you tell? A B C D Q 2. Describe each box using the following key words: Pure Mixture Substance(s) Element Molecule Worked example: Box A is not a pure substance. It contains four different substances, which are elements. These are not molecules.

Plenary The Elementary Inspector Find the names of these symbols: V_____ Mg______ B_____ Complete the gaps in the following passage using the elements names and symbols from the Periodic Table: The burglar had been caught red-handed with the (Ag)____ and (Au) ____. He had been caught after a high speed (Carbon + Argon)_______ chase. The Inspector was anxious to get this one (Fe)______ed out before the end of the night. The victim arrived at the station shouting (holmium + tungsten) _______ dare someone (sulphur + tellurium + aluminium)____ from me. The (cobalt) ____nstable (potassium + neon + tungsten)_______ this was going to be an easy (calcium + selenium) ______ to solve.

Dissolving 25/9/2015 Keywords: Learning Objectives • Explain the terms solvent, solution, solute and soluble • Identify factors that affect dissolving • Explain the difference between a dilute solution and a concentrated solution DO IT NOW: Think, Pair, Share: What do you think ‘dissolving’ means? Give an example of when something dissolves.

Key words Word Solute Solvent Solution Saturated Soluble Insoluble Dissolving Definition A solid that can dissolve A liquid that dissolves a solid A clear liquid that contains a dissolved solid A solution that cannot dissolve any more solid The word for a solid that can dissolve to form a solution The word for a solid that does not dissolve The physical process where a solid mixes into a solvent to make a clear solution
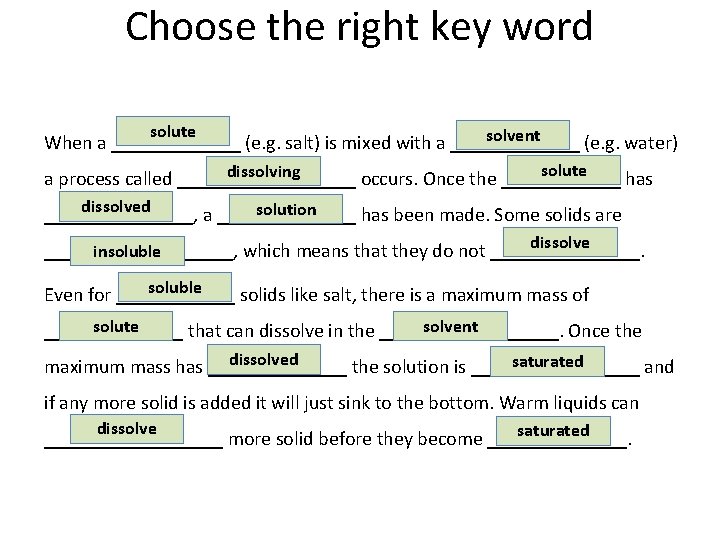
Choose the right key word solute solvent When a _______ (e. g. salt) is mixed with a _______ (e. g. water) solute dissolving a process called _________ occurs. Once the ______ has dissolved solution ________, a _______ has been made. Some solids are dissolve insoluble __________, which means that they do not ________. soluble Even for ______ solids like salt, there is a maximum mass of solute solvent _______ that can dissolve in the _________. Once the dissolved saturated maximum mass has _______ the solution is _________ and if any more solid is added it will just sink to the bottom. Warm liquids can dissolve saturated _________ more solid before they become _______.

Task: Dissolving animation Watch the following animation, then answer the questions: http: //group. chem. iastate. edu/Greenbowe/sections/projectfolder/flashfiles/thermochem/so lution. Salt. swf Q 1. In the animation, what was the: a. Solvent b. Solute c. Solution Q 2. Using the animation to help, draw THREE diagrams in your book, to show three steps in dissolving. Step 1: The salt and water BEFORE dissolving happens Step 2: The water molecules surrounding the salt Step 3: The solution, after the water dissolves the salt.

Solubility • Soluble: Dissolves. More soluble things dissolve more easily • Insoluble: Doesn’t dissolve Ways to speed up dissolving? Name TWO

Task: Dissolving animation Watch the following animation, then answer the questions: http: //group. chem. iastate. edu/Greenbowe/sections/projectfolder/flashfiles/thermochem/so lution. Salt. swf Q 1. In the animation, what was the: a. Solvent b. Solute c. Solution Q 2. Using the animation to help, draw THREE diagrams in your book, to show three steps in dissolving. Step 1: The salt and water BEFORE dissolving happens Step 2: The water molecules surrounding the salt Step 3: The solution, after the water dissolves the salt.

TASK: Draw and label a cup of black coffee. Your diagram should include the following: a. Diagrams of the particles in each ingredient of the cup of coffee (water, sugar, coffee) b. Labels showing each ingredient c. Labels showing whether the ingredient is a SOLUTE, a SOLVENT, or a SOLUTION

The solubility of sodium chloride (Na. Cl, which is table salt) is 36 g/100 ml. This means that only 36 g of sodium chloride can be dissolved in 100 ml of water. If any more is added, this will sink to the bottom, and not dissolve.

Solubility depends on the temperature of the solvent. The warmer the temperature, the faster the particles move. This means that more of the solute can be dissolved and separated. Q. Look at the diagram above. Which person will find it easier to make their tea? Explain why [hint: higher-level students would mention the difference in how the particles move]

Plenary Spray paint can be removed by a special ‘graffiti remover’. This is often sprayed at high temperature to remove graffiti. Q 1. Name the solvent and the solution above Q 2. Suggest why graffiti remover is sprayed at a high temperature

Separating Mixtures 23/02/2021 Keywords: filter, mixture, filtration, insoluble, immiscible Learning Objectives Recognise the differences between substances and use these to separate them DO IT NOW: It is very important for us to be able to separate different mixtures. 1. How do we know this is a mixture? 2. How can the components be separated?

What is the difference between a pure substance and a mixture? “A pure substance contains only one element or compound. ” Food and drink may be advertised as ‘pure’. For example, you may see cartons of ‘pure orange juice’ or ‘pure mineral water’. This means that nothing else was added to the orange juice or mineral water during manufacture Are these pure substances?

A mixture is made from molecules of elements and compounds that are simply mixed together, without chemical bonds. Mixtures can be separated using techniques such as filtration, chromatography, evaporation and distillation.

Sort the images on the next slide into elements, compounds and mixtures! Elements Compounds Mixtures
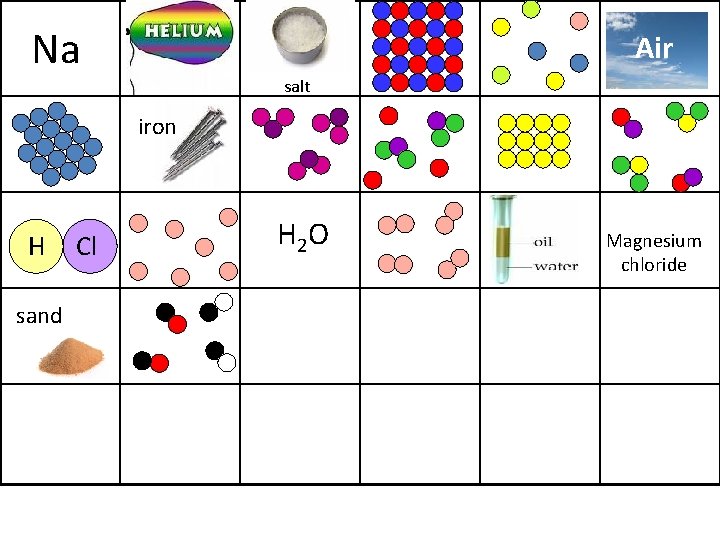
Na Air salt iron H sand Cl H 2 O Magnesium chloride

Task: Learning about separation Go through the following animation, then answer the questions: http: //www. mheducation. ca/school/applets/bcscience 7/mixtures/bcscience 7_mixtures. swf 1. Explain how a magnet could be used to separate a mixture 2. What could we use instead of a ‘desert tent apparatus’ in the laboratory, to separate substances? 3. Why can’t we use a filter and funnel to separate salt from salty water?
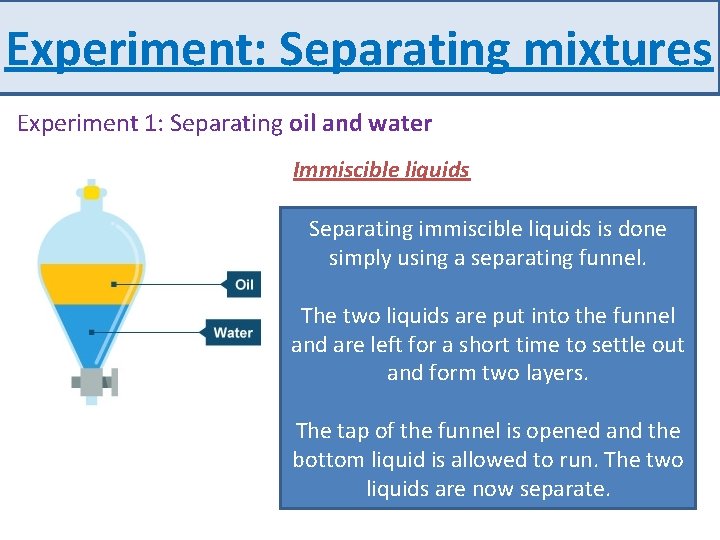
Experiment: Separating mixtures Experiment 1: Separating oil and water Immiscible liquids Separating and waterliquids in a separating Separatingoilimmiscible is done funnel simply using a separating funnel. Oil and water can be separated using a funnel The two liquids are put into the funnel and are left for a short time to settle out Immiscibleand means liquids don't formthat twothe layers. dissolve in each other – oil and water are an Theexample. tap of the funnel is opened and the bottom liquid is allowed to run. The two liquids are now separate.
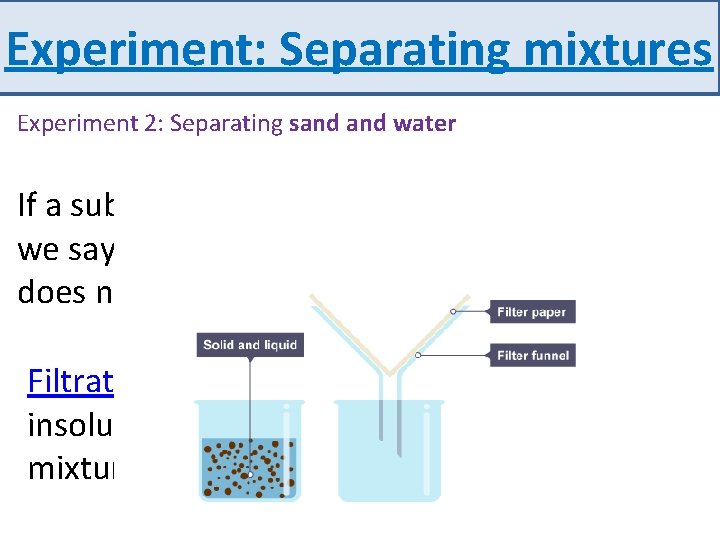
Experiment: Separating mixtures Experiment 2: Separating sand water If a substance does not dissolve in a solvent, we say that it is insoluble. For example, sand does not dissolve in water – it is insoluble. Filtration is a method for separating an insoluble solid from a liquid. When a mixture of sand water is filtered

Experiment: Separating mixtures Experiment 3: Separating sand pebbles

Experiment: Separating mixtures Experiment 4: Separating iron and sand
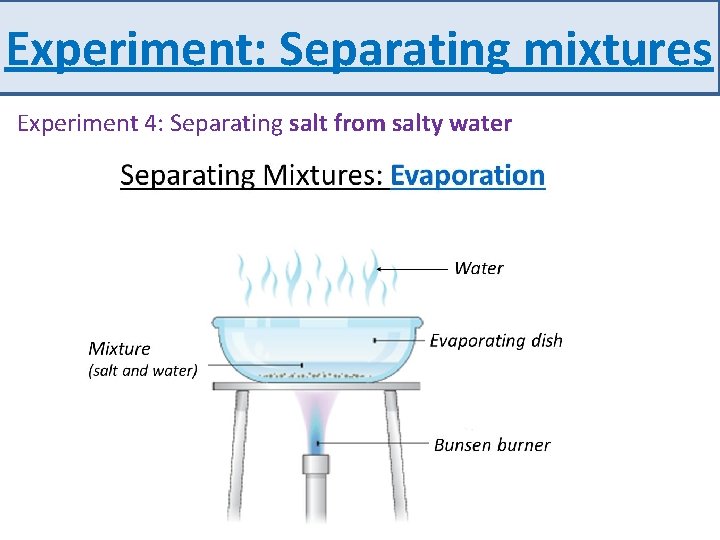
Experiment: Separating mixtures Experiment 4: Separating salt from salty water
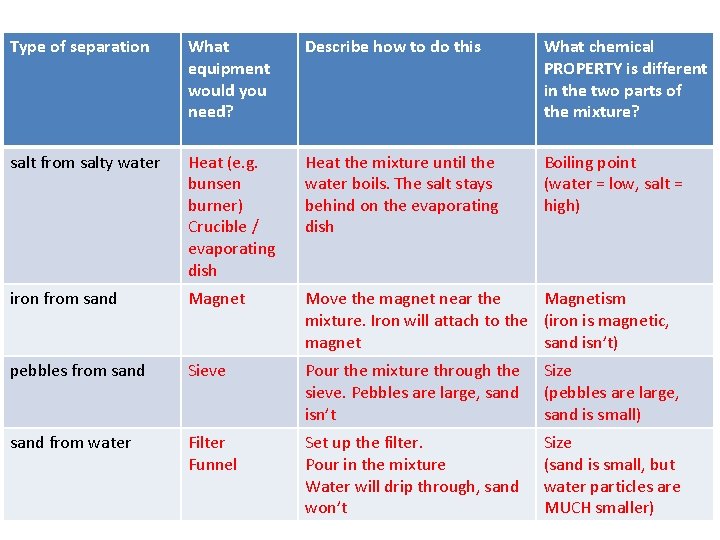
Type of separation What equipment would you need? Describe how to do this What chemical PROPERTY is different in the two parts of the mixture? salt from salty water Heat (e. g. bunsen burner) Crucible / evaporating dish Heat the mixture until the water boils. The salt stays behind on the evaporating dish Boiling point (water = low, salt = high) iron from sand Magnet Move the magnet near the Magnetism mixture. Iron will attach to the (iron is magnetic, magnet sand isn’t) pebbles from sand Sieve Pour the mixture through the sieve. Pebbles are large, sand isn’t Size (pebbles are large, sand is small) sand from water Filter Funnel Set up the filter. Pour in the mixture Water will drip through, sand won’t Size (sand is small, but water particles are MUCH smaller)

Plenary Describe how you would separate: a. Salt from salty water b. Iron filings from a mixture of iron and sand c. Different coloured inks found in a black felt-tip pen d. Small stones from large rocks (2 minutes to discuss/write down each method – 8 minutes total)
Understanding Distillation 22 nd May 2018 Keywords: vapour, condense, distillation, Liebig condenser, purify Learning Objectives Describe distillation. Explain the physical processes involved in distillation. Identify the advantages of distillation. DO IT NOW : Match the Solute to the solvent and the solution Paint Liquid Tea Dissolved Nail Varnish Water Sugary Tea Sugar White Spirit Dissolved Paint Salt Acetone Salt Water
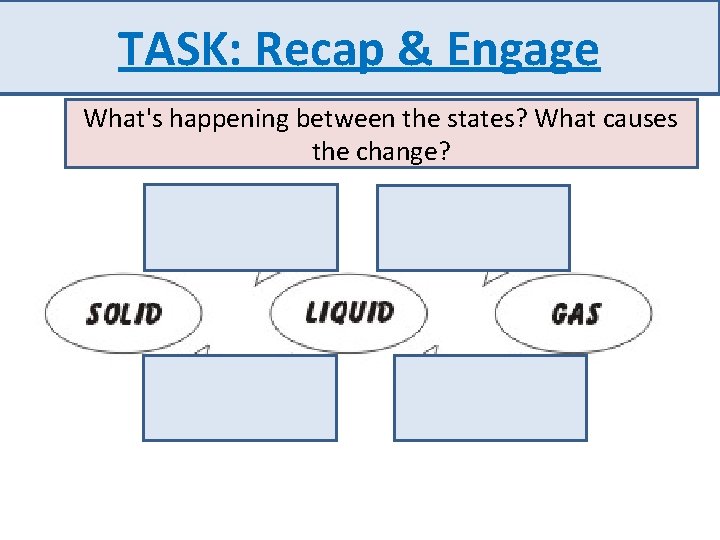
TASK: Recap & Engage What's happening between the states? What causes the change?
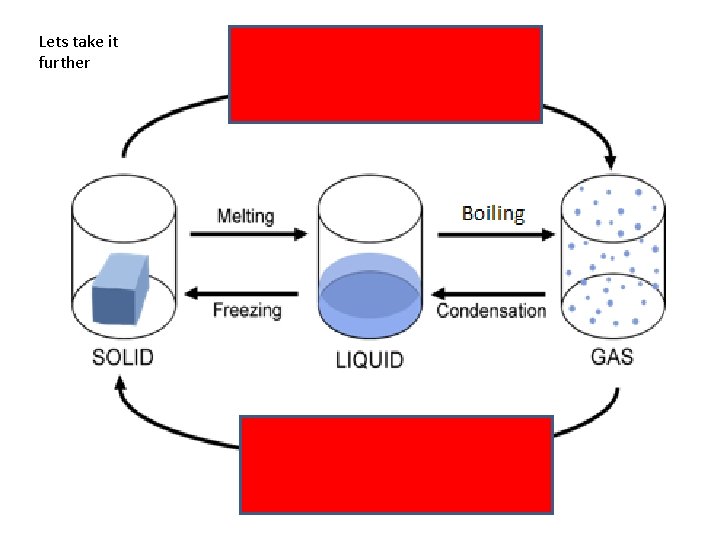
Lets take it further
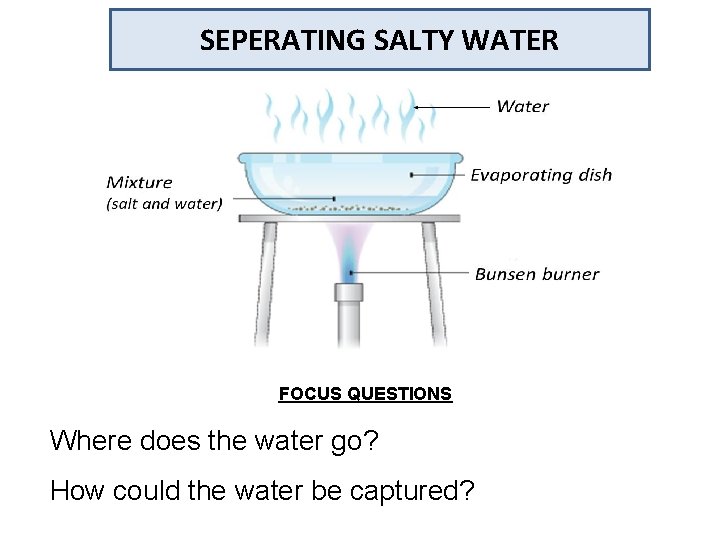
SEPERATING SALTY WATER FOCUS QUESTIONS Where does the water go? How could the water be captured?
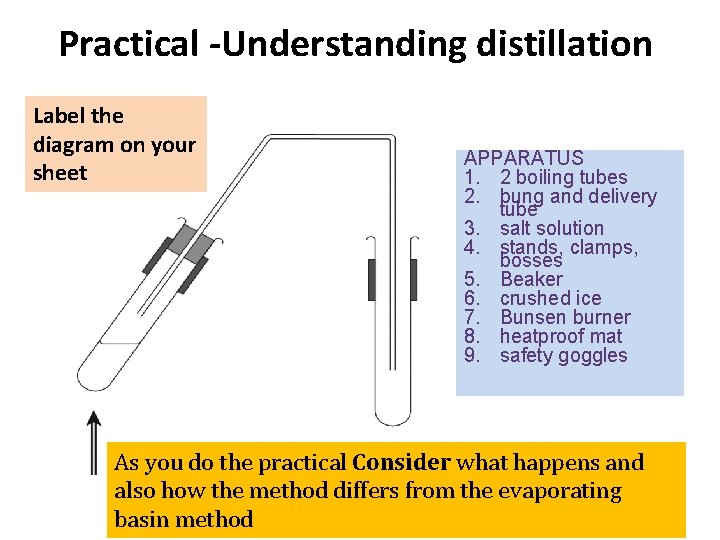
Practical -Understanding distillation Label the diagram on your sheet APPARATUS 1. 2 boiling tubes 2. bung and delivery tube 3. salt solution 4. stands, clamps, bosses 5. Beaker 6. crushed ice 7. Bunsen burner 8. heatproof mat 9. safety goggles As you do the practical Consider what happens and also how the method differs from the evaporating basin method
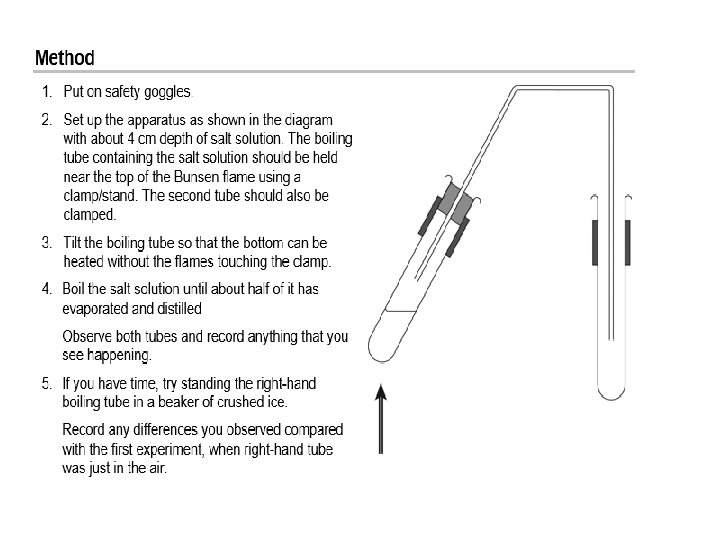

Clear water in boiling tube 2 Test with p. H paper Not efficient, lots of heat energy used and some water lost as steam/vapour Put boiling tube two into a beaker with ice. Cold surface increases the rate of condensation Use a Liebig condenser

Using a Liebig condenser • https: //www. youtube. com/watch? v=N 0 f 73 tb. GCRE

TASK: Distillation Now try these questions… Q 1. What liquid drips into the beaker? Q 2 Explain why running cold water is needed to make the condenser work Q 3. Explain why a Liebig condenser purifies substances [HINT: Think about what you know from previous lessons on what purity means] Q 3. Describe how you would separate water from ethanol.

Plenary Define the following key words: A. Distillation B. Pure C. Boiling point
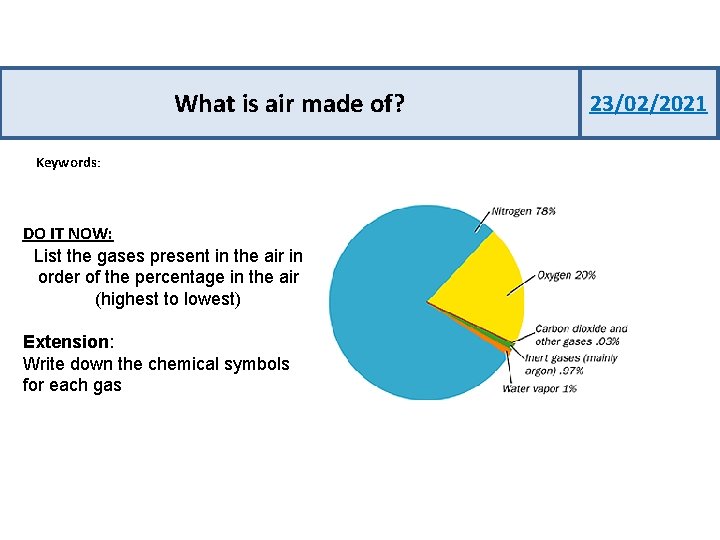
What is air made of? Keywords: DO IT NOW: List the gases present in the air in order of the percentage in the air (highest to lowest) Extension: Write down the chemical symbols for each gas 23/02/2021

TASK: Air composition varies! Carbon dioxide makes up roughly 0. 04% of our atmosphere. However this percentage can go up or down! Discuss and write down reasons to explain: a. Why carbon dioxide levels sometimes go above 0. 04% b. Why carbon dioxide levels sometimes go below 0. 04% [HINT: Think of natural, or man-made factors that change the levels of carbon dioxide]

Separation by fractional distillation

Poster project • Make a labelled poster which shows the composition of different gases in air on earth (pie chart). • Particle diagram representing the different gases in the earth’s atmosphere • All the gases in the earth and their uses (use notes) • Pollutants… car fumes, industry, cows (see me if you pick this) • Title banner: The Earth’s Atmosphere

Plenary Exam-style question – MARK SCHEME Describe the process of separating gases in the air (6 marks = 6 points) Use the following key words in your answer: Compress Temperature Boil Liquid Cooled 1. Air is compressed, (so the particles are closer together. ) 2. The air is also cooled (to a very low temperature) 3. This (low temperature) means the air turns into a liquid 4. (The liquid is then) slowly heated 5. Different gases boil at different temperatures 6. This allows the different gases to be separated. Heated

Exploring chromatography 23 February 2021 Keywords: mixture, separate, dye, chromatogram, paper chromatography, soluble Learning Objectives 1. Use chromatography to separate dyes DO IT NOW: complete the match up activity B A Filtration D C Evaporation Distillation Extension: Explain the method of each technique Chromatography

So what is chromatography? Chromatography is a separating technique used to separate two or more soluble substances. It is an important method used to identify unknown substances. It is widely used by scientists to detect drugs, dyes, explosives and paints.

So what is chromatography? _____ is a separating technique used to separate two or more ______ substances. It is an important method used to identify ________. It is widely used by _______ to detect drugs, dyes, explosives and paints. Keywords: scientists, unknown substances, chromatography, soluble
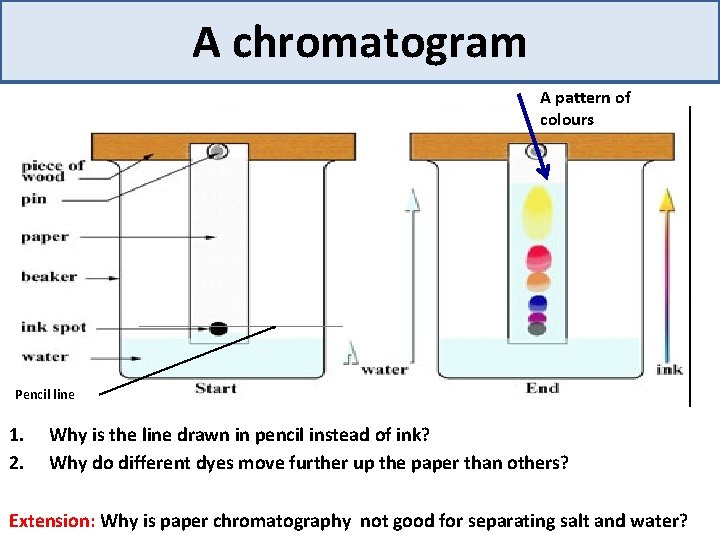
A chromatogram A pattern of colours Pencil line 1. 2. Why is the line drawn in pencil instead of ink? Why do different dyes move further up the paper than others? Extension: Why is paper chromatography not good for separating salt and water?

Method Put a spot of ____ on a piece of filter _____. Place the bottom of the filter paper in some ____. Wait for a few minutes while the ink separates into different ____. Different colours will move different _______ up the filter paper. Chromatography lets you find out what colours there are hidden in a paint or a pen.
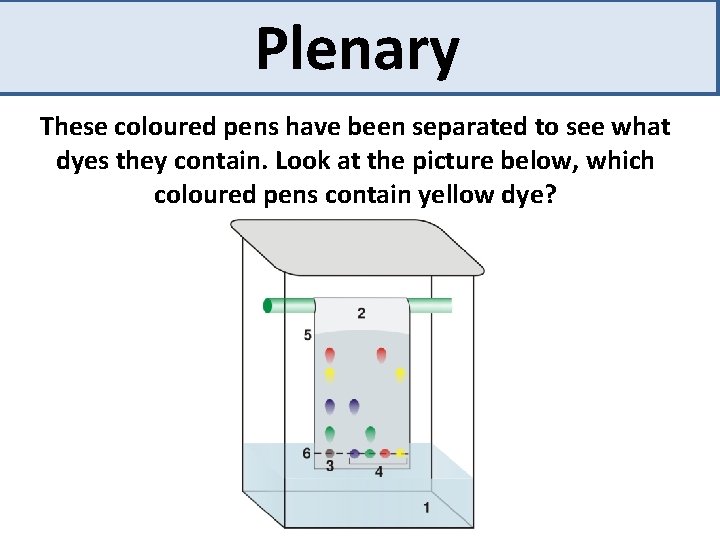
Plenary These coloured pens have been separated to see what dyes they contain. Look at the picture below, which coloured pens contain yellow dye?

Getting it right Make each one into a top tip…for example ‘don’t leave the paper in the water for too long because……. ’
- Slides: 55