RECEPTOR MODELLING OF AIRBORNE PARTICULATE MATTER Roy M

RECEPTOR MODELLING OF AIRBORNE PARTICULATE MATTER Roy M Harrison Division of Environmental Health & Risk Management The University of Birmingham United Kingdom

RECEPTOR MODELLING OF PARTICULATE MATTER IN EUROPE BACKGROUND (1) • CAFE Position Paper reports only results of studies based upon major component composition • Historically there have been a number of studies which have used multi-elemental analyses and multivariate statistics to apportion PM mass to major source categories. These studies are mostly over five years old and the results reflect emissions at the time of air sampling

RECEPTOR MODELLING OF PARTICULATE MATTER IN EUROPE BACKGROUND (2) • The use of sophisticated receptor modelling methods is hampered by - a lack of multi-element datasets of sufficient length - adequate information on source profiles of inorganic constituents (for CMB) - the absence of a road traffic marker since the removal of lead from gasoline - a shortage of high quality EC/OC data - a lack of adequate information on the abundance of organic tracers in relevant source material

EUROPEAN STRENGTHS AND WEAKNESSES • Extensive high quality measurements of physical properties of airborne particles • Good international collaboration in data assessment • Scarcity of detailed information on aerosol chemical composition



RECEPTOR MODELLING OF PARTICULATE MATTER IN THE UNITED KINGDOM (1) • You can only use the data that you have! • QUALITY OF URBAN AIR REVIEW GROUP (1996) - estimated road traffic-derived particles from correlations with NOx and CO - estimated secondary inorganic PM from mass of sulphate and estimated nitrate/ sulphate ratios - assumed coarse particles (PM 2. 5 – 10) to be resuspended dust and sea salt

RECEPTOR MODELLING OF PARTICULATE MATTER IN THE UNITED KINGDOM (2) • THE AIRBORNE PARTICLES EXPERT GROUP (1999) developed a quantitative three component model PM 10 (TEOM) = a. BS + b. SO 42 - + c where BS = black smoke a and b are coefficients determined by regression; c is an intercept representing “other”, mainly coarse particles • The model was subsequently refined (John Stedman, AEA Technology) - to estimate a road traffic contribution from national maps of nitrogen oxides - to include dispersion model results for emissions from non-traffic point and area sources

RECEPTOR MODELLING OF PARTICULATE MATTER IN THE UNITED KINGDOM (3) • The “pragmatic mass closure model” of Harrison et al (2003) offers greater refinement. - nitrate is included as well as sulphate - measurements of OC and EC allow estimation of primary and secondary organic carbon - coarse dusts are quantified through analysis of Cl- (sea salt), Fe and Ca - the method is directly applicable to manual gravimetric samples • Weaknesses include - inability to apportion the primary carbonaceous component - limited information on the sources of coarse “mineral” dusts

URBAN AIR QUALITY: PARTICULATE MATTER Which sources are important? Source Local traffic exhaust Chemical tracer (bulk samples) elemental carbon and organic compounds Long range transport - SO 2 oxidation - NOx oxidation - ammonia emissions sulphates nitrates ammonium Soil and road dusts Construction and demolition Sea salt iron calcium sodium chloride
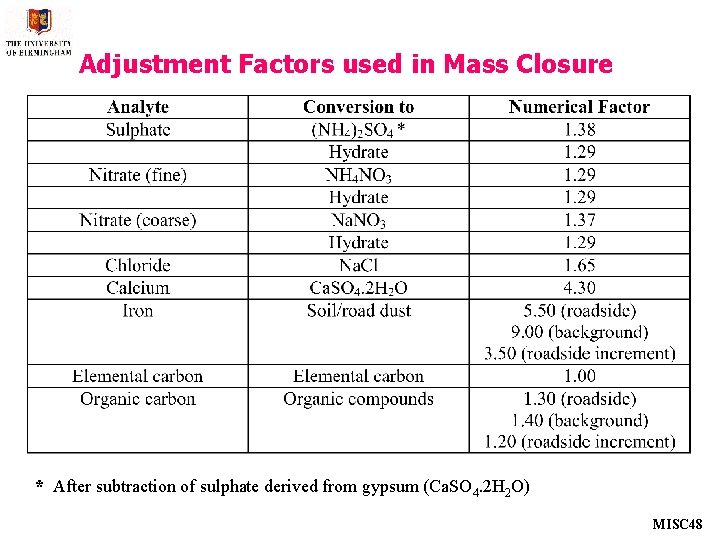
Adjustment Factors used in Mass Closure * After subtraction of sulphate derived from gypsum (Ca. SO 4. 2 H 2 O) MISC 48
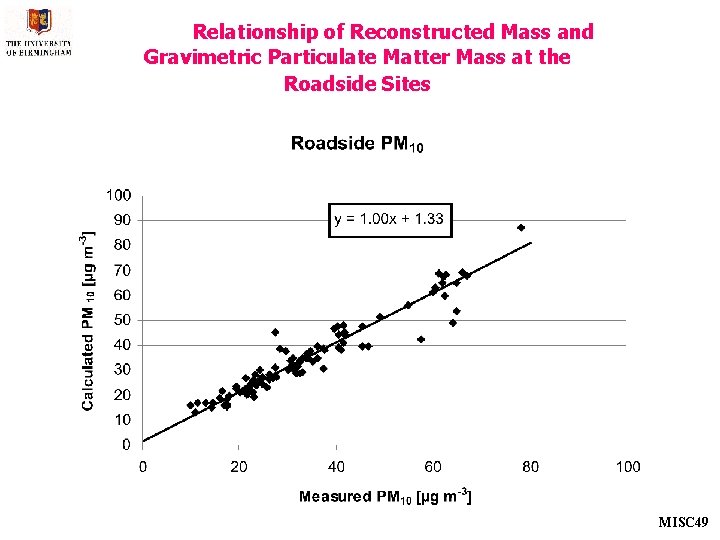
Relationship of Reconstructed Mass and Gravimetric Particulate Matter Mass at the Roadside Sites MISC 49
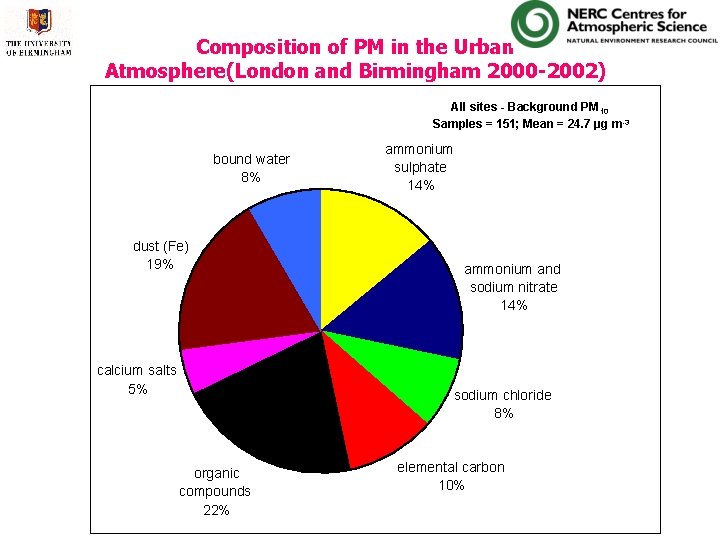
Composition of PM in the Urban Atmosphere(London and Birmingham 2000 -2002) All sites - Background PM 10 Samples = 151; Mean = 24. 7 µg m-3 bound water 8% dust (Fe) 19% calcium salts 5% ammonium sulphate 14% ammonium and sodium nitrate 14% sodium chloride 8% organic compounds 22% elemental carbon 10%
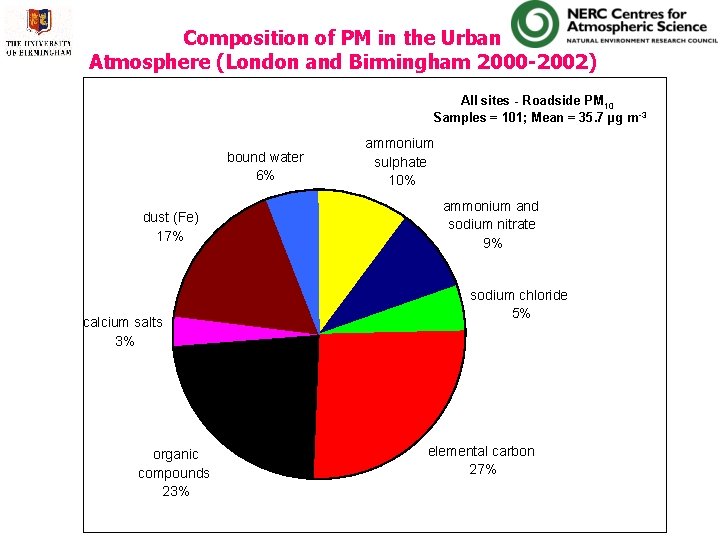
Composition of PM in the Urban Atmosphere (London and Birmingham 2000 -2002) All sites - Roadside PM 10 Samples = 101; Mean = 35. 7 µg m -3 bound water 6% dust (Fe) 17% calcium salts 3% organic compounds 23% ammonium sulphate 10% ammonium and sodium nitrate 9% sodium chloride 5% elemental carbon 27%
![40 Days PM 10 > 50 µg m-3 [n = 10] 30 All days 40 Days PM 10 > 50 µg m-3 [n = 10] 30 All days](http://slidetodoc.com/presentation_image_h2/e57d70ad56769d43d131f681954ecfb8/image-15.jpg)
40 Days PM 10 > 50 µg m-3 [n = 10] 30 All days [n = 151] Bound water Iron rich dust Calcium sulphate Sodium chloride Ammonium sulphate Sodium nitrate Ammonium nitrate Organic compounds Elemental carbon 70 Mean mass Mean Concentration [µg m -3] Comparison of PM 10 Composition for Days with -3 PM 10 > 50 µg m with all Days: Urban background 60 50 20 10 0
![-3 Mean Concentration [µg m ] 0 Bound water Iron rich dust Calcium sulphate -3 Mean Concentration [µg m ] 0 Bound water Iron rich dust Calcium sulphate](http://slidetodoc.com/presentation_image_h2/e57d70ad56769d43d131f681954ecfb8/image-16.jpg)
-3 Mean Concentration [µg m ] 0 Bound water Iron rich dust Calcium sulphate Sodium chloride Ammonium sulphate 12 Sodium nitrate Ammonium nitrate Organic compounds Elemental carbon Comparison of PM 10 Composition for Days with PM 10 > 50 µg m with all Days: Urban background -3 14 Days PM 10 > 50 µg m-3 [n = 10] All days [n = 151] 10 8 6 4 2

RECEPTOR MODELLING IN THE UNITED KINGDOM Clearly the eight-component model is not in itself sufficient to allow prediction of future PM concentrations. Additional refinements should include: • Estimation of secondary organic carbon and modelling of source-receptor relationships for SOC • Estimation of contributions to primary EC and OC – Using dispersion modelling • Understanding more fully the sources of the “ironrich” and “calcium –rich” dust components. • Modelling source-receptor relationships for sulphate and nitrate, including possible effects of changing ammonia emissions.

RECEPTOR MODELLING OF AIRBORNE PARTICULATE MATTER • Use of the TSI AEROSOL TIME-OF-FLIGHT MASS SPECTROMETER • Advantages – Comprehensive information on single particle composition • Disadvantages – May not ionise the entire particle – Illustrates the immense complexity of urban particles – Post-emission particles processing makes source recognition difficult – Some sources show immense diversity of composition when analysed as single particles

The ATOFMS (Aerosol Time-of-Flight Mass Spectrometer) • Laser based system. • Measures the size and composition of individual particles. • Transportable. • Runs off 240 VAC
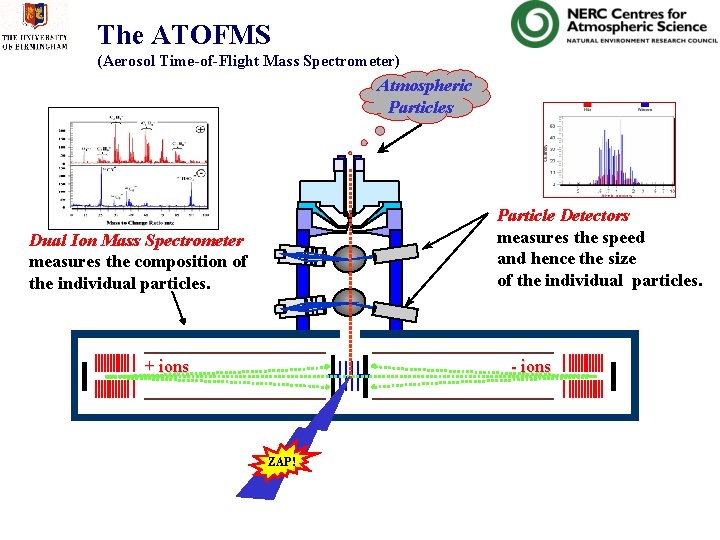
The ATOFMS (Aerosol Time-of-Flight Mass Spectrometer) Atmospheric Particles 0. 68 m Particle Detectors measures the speed and hence the size of the individual particles. Dual Ion Mass Spectrometer measures the composition of the individual particles. + ions - ions ZAP!

Example of ATOFMS Particle Characterisation Results (1)
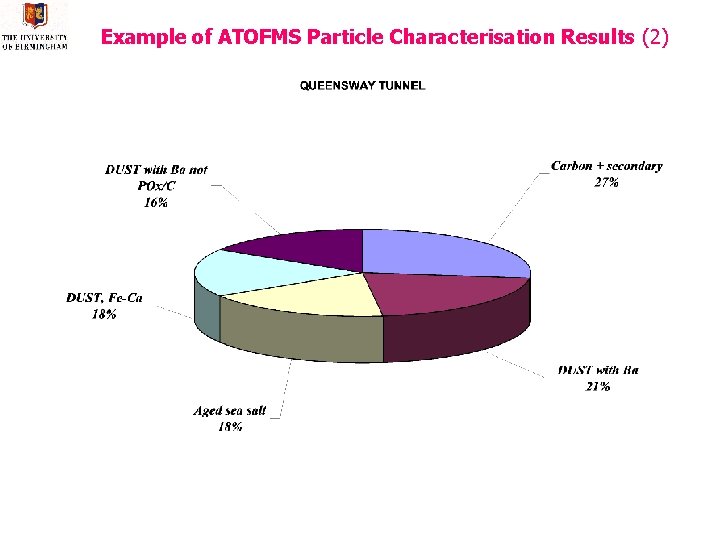
Example of ATOFMS Particle Characterisation Results (2)
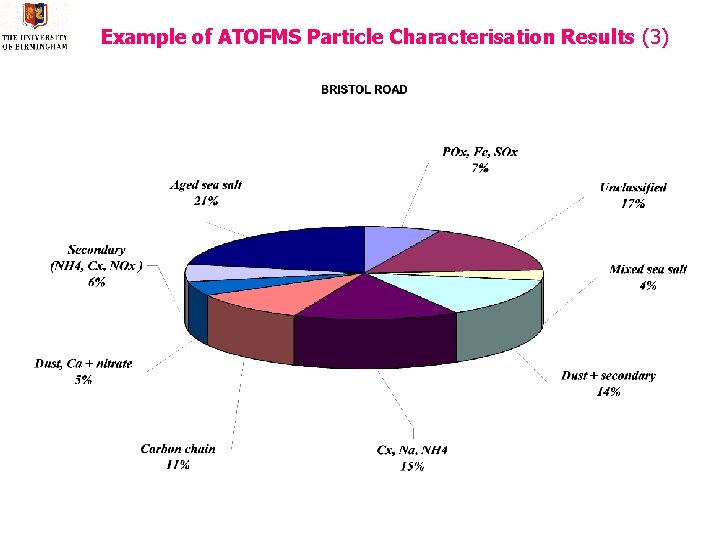
Example of ATOFMS Particle Characterisation Results (3)

SUMMARY AND CONCLUSIONS • Limited progress in source apportionment in the UK. • Receptor models based on major components. • Three component model now being superceded by eight component model. • Combination of receptor model and dispersion model more powerful. • Single particle mass spectrometry (ATOFMS) is powerful but poses series interpretational problems.
- Slides: 24