Recap Hemoglobin has a sigmoidal not hyperbolic oxygen
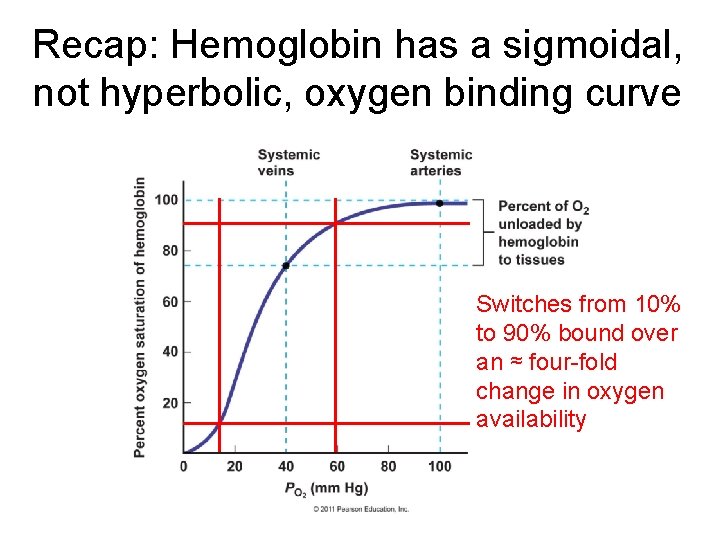
Recap: Hemoglobin has a sigmoidal, not hyperbolic, oxygen binding curve Switches from 10% to 90% bound over an ≈ four-fold change in oxygen availability

Hemoglobin has four oxygen binding sites: Is this responsible for its sigmoidal curve?

Suppose all four sites are identical and independent (i. e. , each site is unaffected by the binding of O 2 elsewhere) with binding reaction rate kon and dissociation reaction rate koff P: Protein (hemoglobin) X: Ligand (O 2)

Finding the binding curve when sites are independent What is the total concentration of binding sites? What is the total concentration of bound sites? What is the fraction of sites bound?

Finding the binding curve when sites are independent Assume that equilibrium has been reached:

Finding the binding curve when sites are independent Assume that equilibrium has been reached:

Finding the binding curve when sites are independent Find expressions for the concentration of each complex by this method: Then plug them into our expression for the fraction of sites bound:

Finding the binding curve when sites are independent Four independent binding sites cannot explain hemoglobin’s sigmoidal binding curve!

If the sites are not independent, then binding affinity at each site depends on the occupancy of the other sites. Perhaps the sites are also not identical. How could this be modeled?

Modeling with non-identical, non-independent binding sites A C B D How many distinct states of oxygen binding are there? 24 = 16 How many transitions are there out of any given state (that involve gaining or losing a single O 2 molecule)? 4 How many “on” and “off” reaction rates are there to fit? 16 x 4 = 64

“With four parameters I can make an elephant, and with five I can make him wiggle his trunk. ” -- John von Neumann

Added slide: actual elephants made with four parameters (fifth makes the trunk wiggle and eye blink) For more details, please see: http: //perfdynamics. blogspot. com/2011/06/winking-pink-elephant. html

Modeling with identical, non-independent binding sites How many distinct states of oxygen binding are there? 5 How many transitions are there out of a given state (that involve gaining or losing a single O 2 molecule)? 1 (P and PX 4) or 2 (PX, PX 2, and PX 3) How many “on” and “off” reaction rates are there to fit? 8
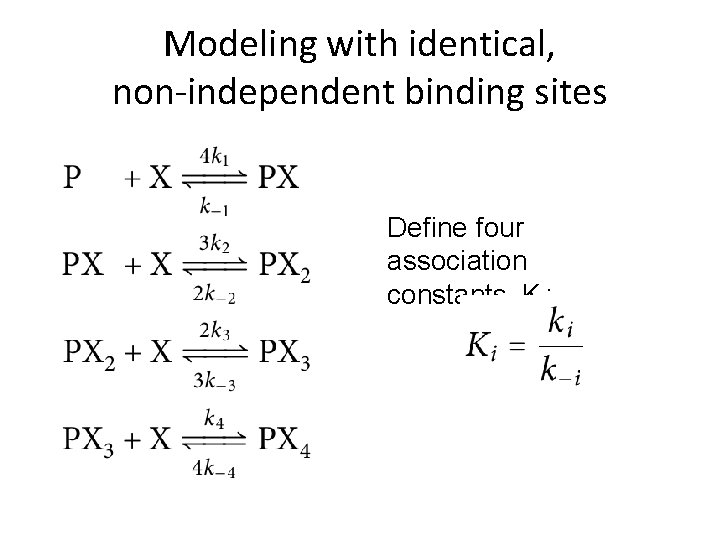
Modeling with identical, non-independent binding sites Define four association constants, Ki:

Finding an expression for the fraction of sites bound Find expressions for the concentration of each complex by this method Then plug them into our expression for the fraction of sites bound:

Finding an expression for the fraction of sites bound This general expression is called the Adair equation.

Finding an expression for the fraction of sites bound A special case: K 4 >> K 1, K 2, K 3 This is an example of cooperativity because the binding of earlier molecules “helps” the last one bind.

Cooperativity and Hill curves … is just a specific example of a Hill equation. The Hill equation: Hill coefficient (always ≤ # of binding sites)

The best-fit Hill coefficient is often less than the number of binding sites
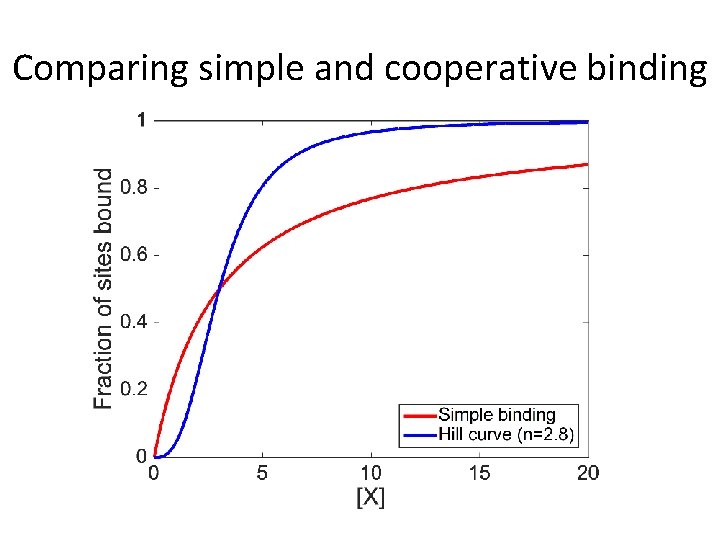
Comparing simple and cooperative binding

How hard is it to switch from “mostly not bound” to “mostly bound”? What is [X] when 10% of sites are bound? What is [X] when 90% of sites are bound? …need to increase [X] by a factor of 811/2. 8 ≈ 4. 8
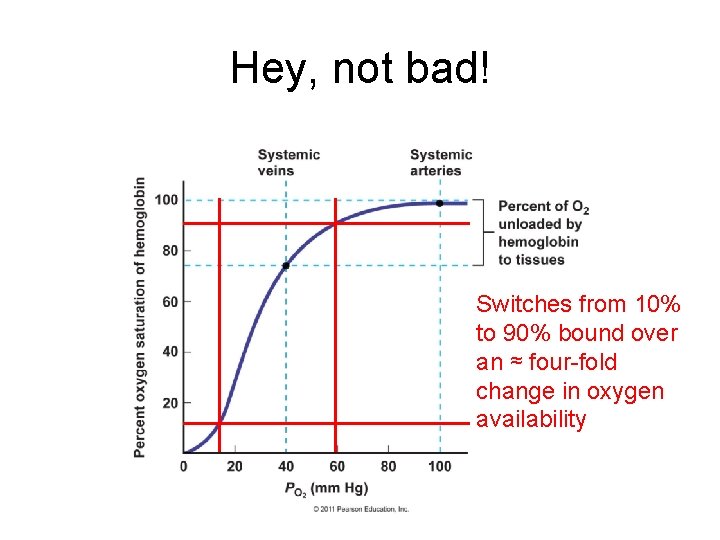
Hey, not bad! Switches from 10% to 90% bound over an ≈ four-fold change in oxygen availability

The Hill function is simple but gives no mechanistic insight X Can a biologically-inspired model do as well while keeping parameter number low?
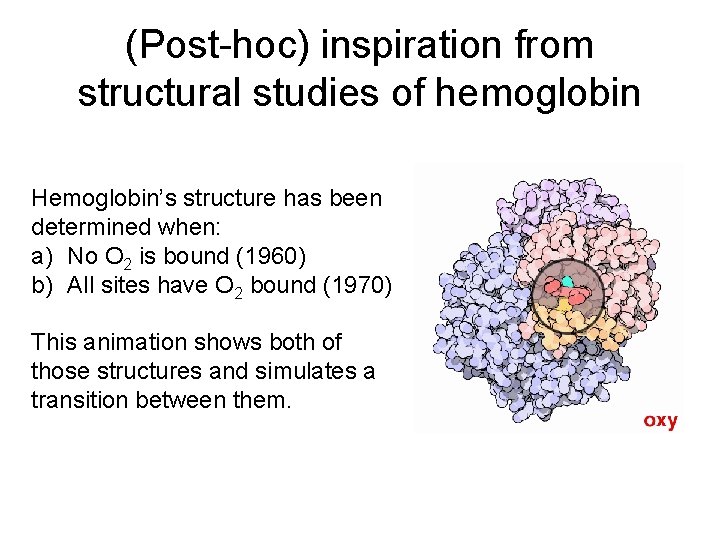
(Post-hoc) inspiration from structural studies of hemoglobin Hemoglobin’s structure has been determined when: a) No O 2 is bound (1960) b) All sites have O 2 bound (1970) This animation shows both of those structures and simulates a transition between them.

Monod-Wyman-Changeux (1965) • Hemoglobin exists in two folding states, tensed/taut (T) and relaxed (R) – Essentially the “oxy” and “deoxy” crystal structures just shown, but with variable numbers of O 2 molecules bound • All four binding sites behave identically • Binding sites in R-state hemoglobin have a higher affinity for oxygen
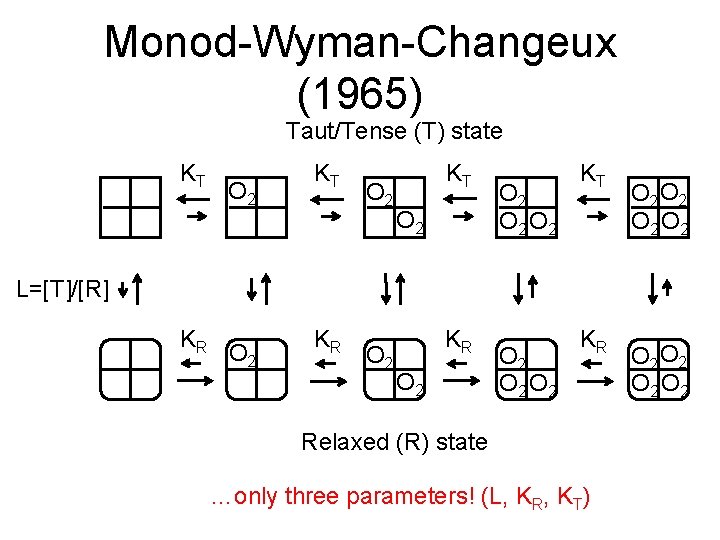
Monod-Wyman-Changeux (1965) Taut/Tense (T) state KT O 2 O 2 O 2 O 2 L=[T]/[R] KR O 2 O 2 KR Relaxed (R) state …only three parameters! (L, KR, KT) O 2 O 2

Monod-Wyman-Changeux With some algebraic effort, one can show that the fraction of bound sites under this model is:
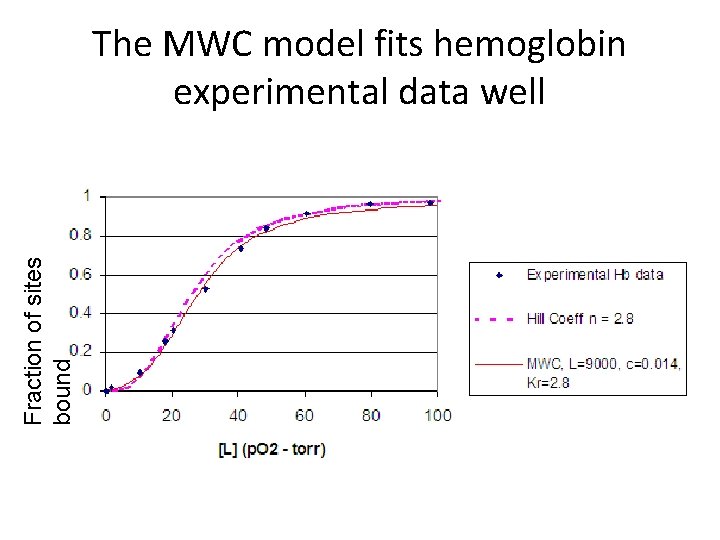
Fraction of sites bound The MWC model fits hemoglobin experimental data well
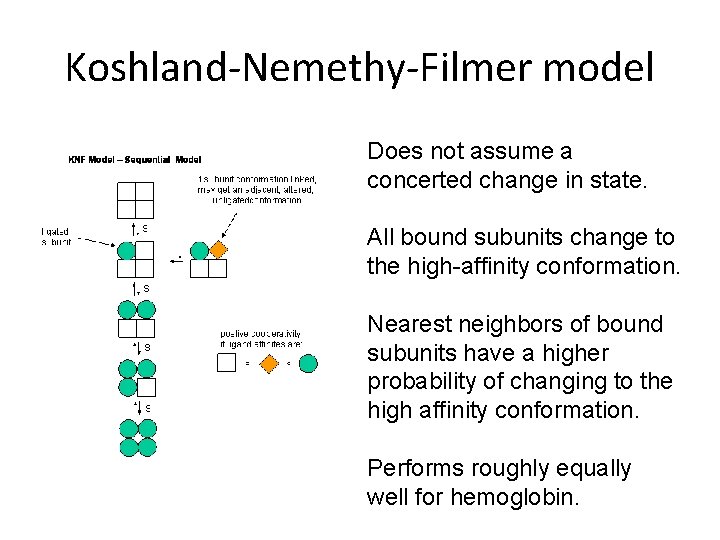
Koshland-Nemethy-Filmer model Does not assume a concerted change in state. All bound subunits change to the high-affinity conformation. Nearest neighbors of bound subunits have a higher probability of changing to the high affinity conformation. Performs roughly equally well for hemoglobin.
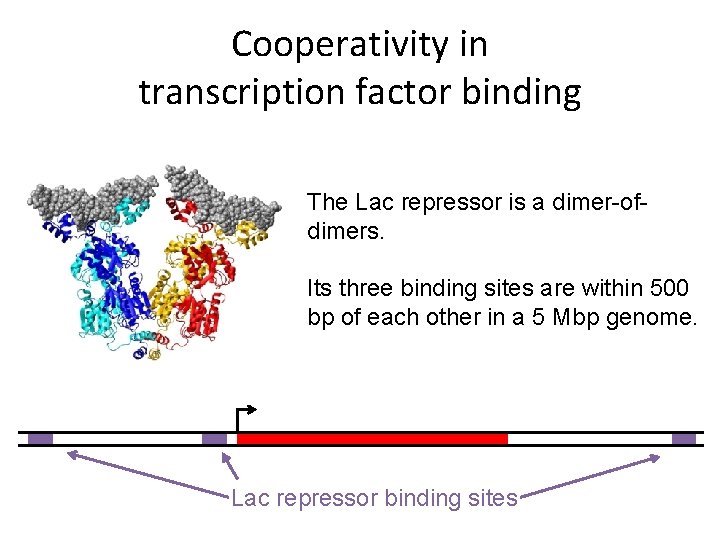
Cooperativity in transcription factor binding The Lac repressor is a dimer-ofdimers. Its three binding sites are within 500 bp of each other in a 5 Mbp genome. Lac repressor binding sites

Cooperativity in transcription factor binding Finding the first site is slow. Once bound, the other end of the protein more quickly finds a second, nearby, binding site.
Cooperativity in transcription factor binding Finding the first site is slow. Once bound, the other end of the protein more quickly finds a second, nearby, binding site. If the repressor falls off of one site, it can quickly reattach.

A Hill curve can describe the rate of target gene transcription For a gene regulated by a transcriptional activator (with maximum expression rate c): For a gene regulated by a transcriptional repressor:

Key concepts from biochemical approach to cooperativity • Cooperativity gives sigmoidal binding curves • The Hill equation models strong cooperative binding with reasonable accuracy • Plausible biological mechanisms for cooperativity include: – concerted changes – nearest-neighbor effects – tethering
- Slides: 34