Reading Solubility Curves What is Solubility Solubility the

Reading Solubility Curves

What is Solubility? Solubility: the maximum amount of solute that will dissolve in a certain amount of solvent at a given temperature n Solute = substance dissolved n Solvent = does the dissolving n Example: __ grams of salt (Na. Cl) in 100 g of water at __ ˚C. n

Temperature and Solubility n Temperature affects how much of the solute can be dissolved by the solvent. ¨ Note: Increasing the temperature does not always increase the solubility n Solubility curves: curves used to show the solubility of a substance changes with temperature.
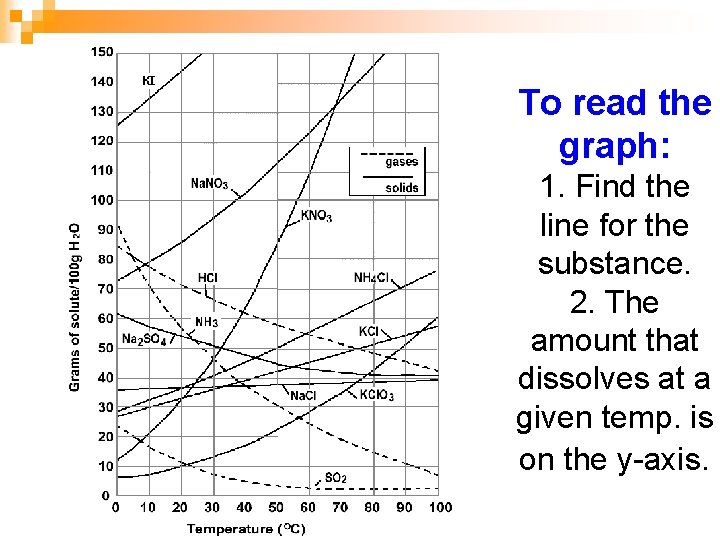
To read the graph: 1. Find the line for the substance. 2. The amount that dissolves at a given temp. is on the y-axis.
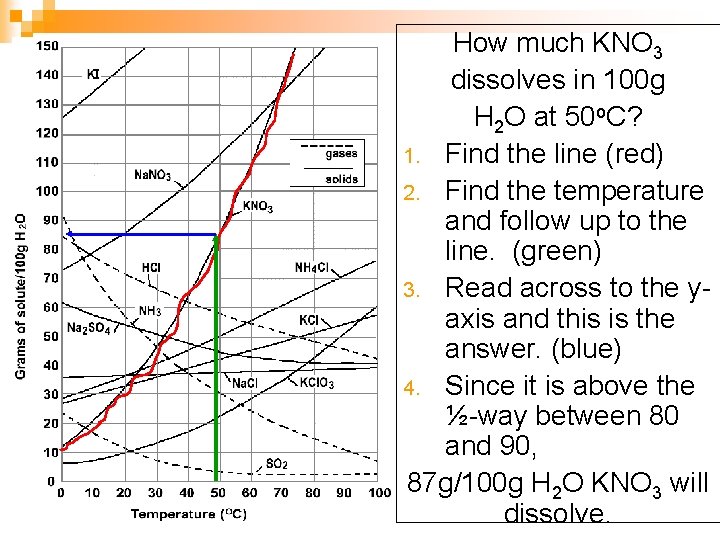
How much KNO 3 dissolves in 100 g H 2 O at 50 o. C? 1. Find the line (red) 2. Find the temperature and follow up to the line. (green) 3. Read across to the yaxis and this is the answer. (blue) 4. Since it is above the ½-way between 80 and 90, 87 g/100 g H 2 O KNO 3 will dissolve.

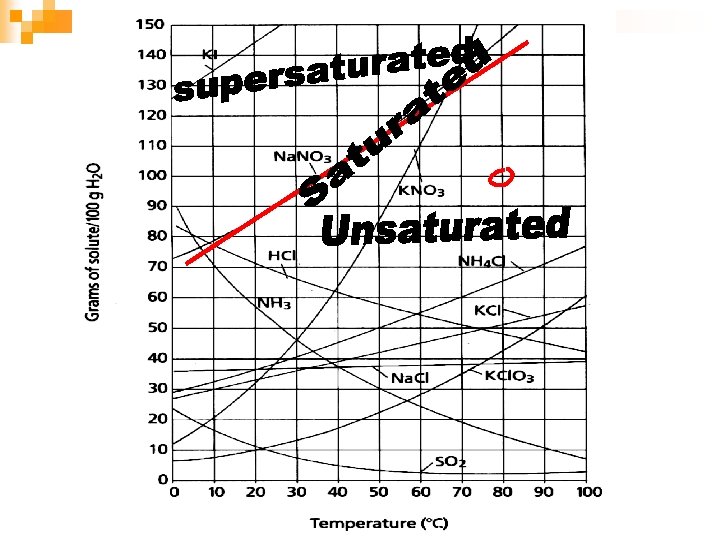
Types of Solutions: n Saturated solution: point on the line ¨ Contains maximum amount of solute at given temp ¨ Contains what it should hold n Supersaturated: above the line ¨ Contains more solute than a saturated solution ¨ Contains more than it should hold n Unsaturated: below the line is ¨ Contains less solute than saturated solution ¨ Contains less solute than it could hold
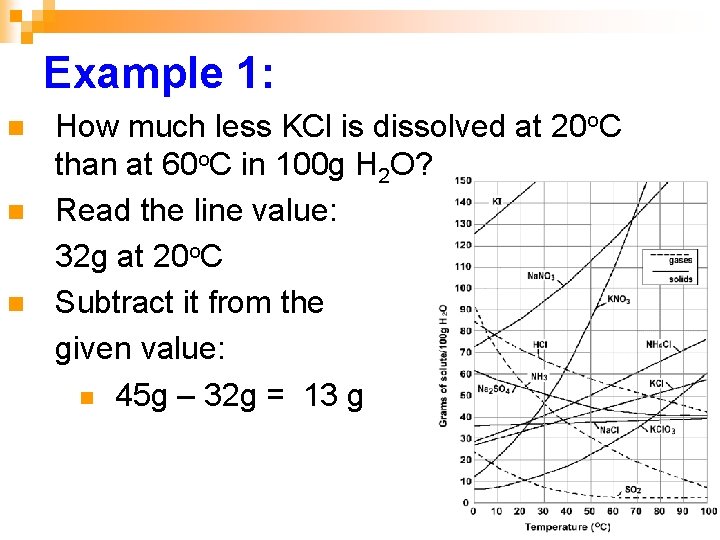
Example 1: n n n How much less KCl is dissolved at 20 o. C than at 60 o. C in 100 g H 2 O? Read the line value: 32 g at 20 o. C Subtract it from the given value: n 45 g – 32 g = 13 g
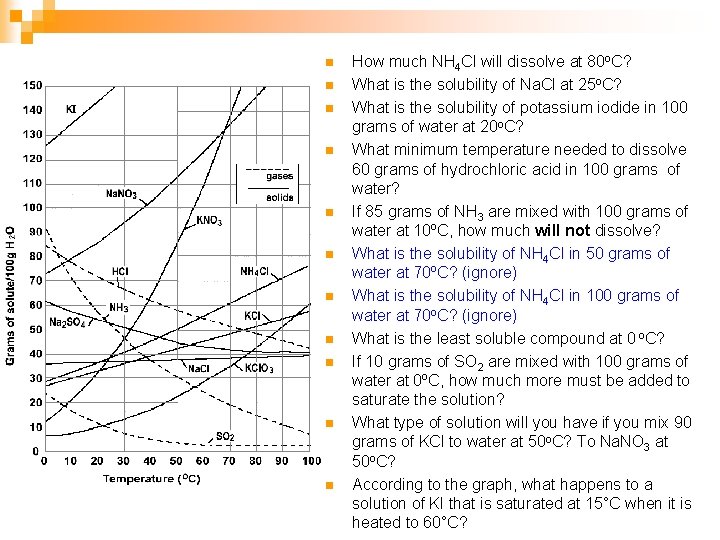
n n n How much NH 4 Cl will dissolve at 80 o. C? What is the solubility of Na. Cl at 25 o. C? What is the solubility of potassium iodide in 100 grams of water at 20 o. C? What minimum temperature needed to dissolve 60 grams of hydrochloric acid in 100 grams of water? If 85 grams of NH 3 are mixed with 100 grams of water at 10ºC, how much will not dissolve? What is the solubility of NH 4 Cl in 50 grams of water at 70ºC? (ignore) What is the solubility of NH 4 Cl in 100 grams of water at 70 o. C? (ignore) What is the least soluble compound at 0 o. C? If 10 grams of SO 2 are mixed with 100 grams of water at 0ºC, how much more must be added to saturate the solution? What type of solution will you have if you mix 90 grams of KCl to water at 50 o. C? To Na. NO 3 at 50 o. C? According to the graph, what happens to a solution of KI that is saturated at 15°C when it is heated to 60°C?


The Nature of Solids Not all solids melt- wood and cane sugar tend to decompose when heated n Most solid substances are crystalline in structure n ¨ Organized pattern
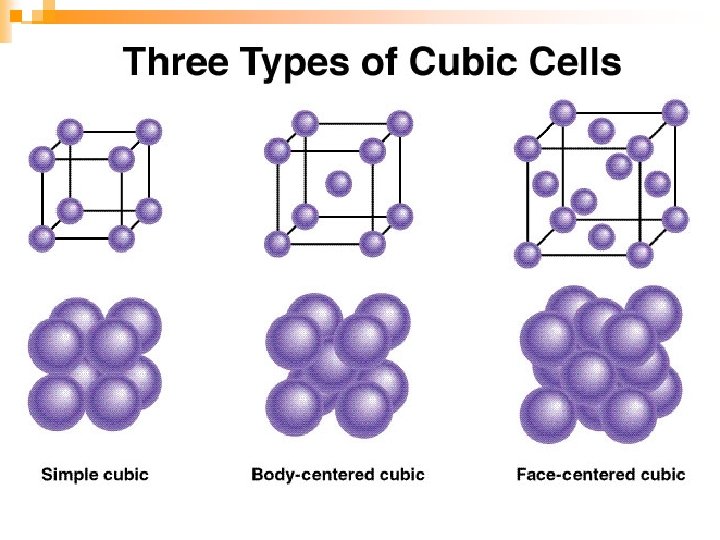
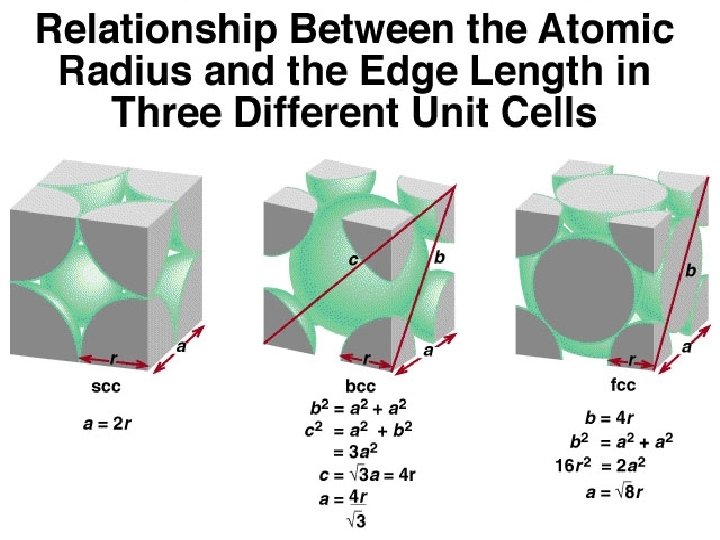

The Nature of Solids n Some solid substances can exist in more than one form called allotropes ¨Elemental carbon is an example - Diamond, formed by great pressure - Graphite, which is in your pencil - Buckminsterfullerene (“buckyballs”)
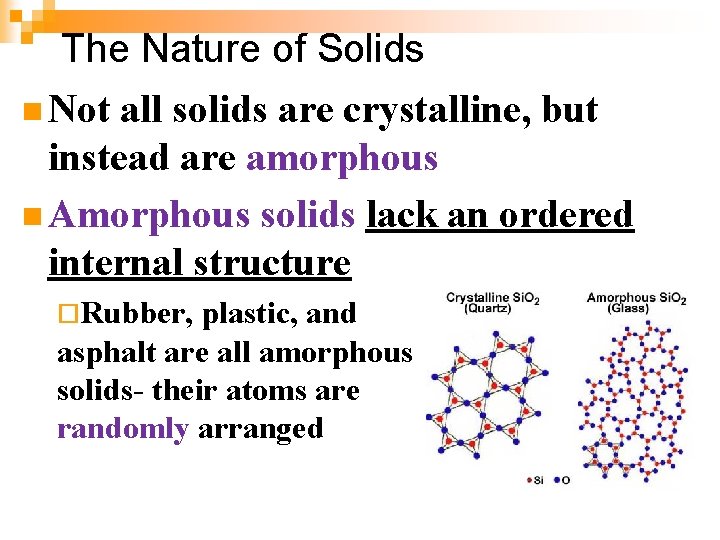
The Nature of Solids n Not all solids are crystalline, but instead are amorphous n Amorphous solids lack an ordered internal structure ¨Rubber, plastic, and asphalt are all amorphous solids- their atoms are randomly arranged

Study for Quiz Tomorrow!
- Slides: 16