REACTIONS IN AQUEOUS SOLUTIONS Solution Properties Precipitate Reations

























- Slides: 28

REACTIONS IN AQUEOUS SOLUTIONS Solution Properties, Precipitate Reations,

CONCENTRATION OF SOLUTION For now we will talk about concentration in terms of only molarity. Eventually we will expand it to include molality and others.

MOLARITY Just one way to express concentration Do not confuse with mole fraction Do not confuse with molality

WHAT IS MOLARITY? Molarity is a statement about a concentration of a solution. ∞ Molarity is the number of moles per 1 Liter That looks like : ∞ # mol solute = 1 L solution

KEEP IN MIND! Molarity equivalent sentences looks like : ∞ # mol solute = 1 L solution The units for Molarity is a M Molarity will be found in a problem as most likely some # M. This will be read as some # molar.

EXAMPLES What will be found in the problem may be the concentration of 0. 785 M table salt (Na. Cl). How you would read it… “the concentration of. 785 Molar table salt (or Sodium Chloride)” What you would write in the equivalent sentence 3 ∞ 0. 785 mol Na. Cl = 1 L Na. Cl (aq) Or 3 ∞ 0. 785 mol Na. Cl = 1 L Na. Cl solution

IF YOU CAN REMEMBER THIS YOU WILL BE FINE… The unit for molarity is This is read as This is understood to be # mol solute per 1 L solution This is written as # mol solute = 1 L solution M molar

CONCENTRATION OF ELECTROLYTE When ionic compounds dissolve, the concentration of the ions is dependent upon the chemical formula. Example: Nickel (II) nitrate solution would have a concentration of nitrate ions that is twice the concentration of the nickel (II) nitrate solution because the formula is Ni(NO 3 ) 2 Look at the subscript 2!

CONCENTRATION OF ELECTROLYTE When ionic compounds dissolve, the concentration of the ions is dependent upon the chemical formula. Example: Nickel (II) nitrate solution would have a concentration of nitrate ions that is twice the concentration of the nickel (II) nitrate solution because the formula is Ni(NO 3 ) 2 Look at the subscript 2!


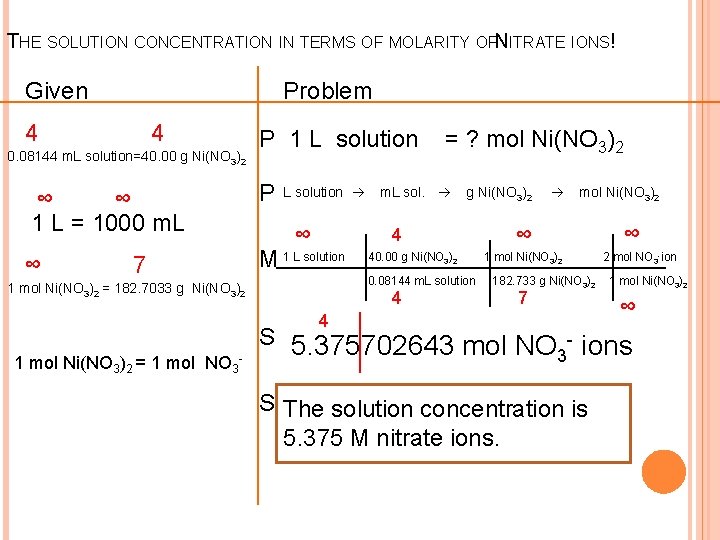
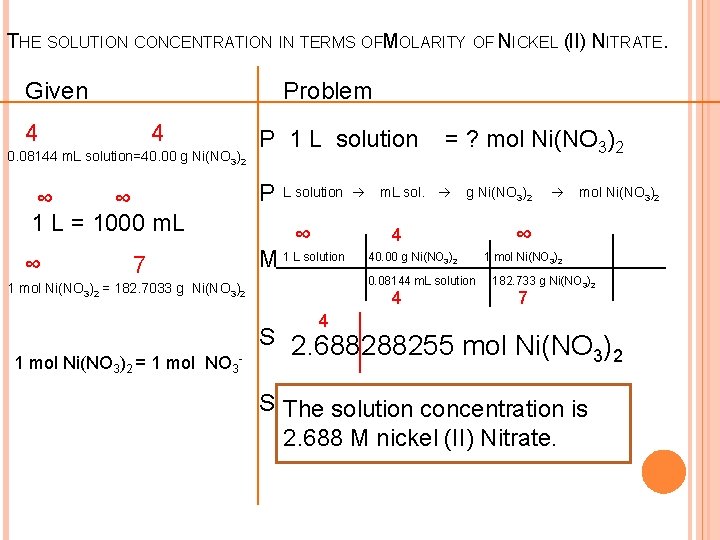
REVIEW THE PROBLEM A sample of a solution has a volume of 0. 08144 L and contains 40. 00 grams of nickel (II) nitrate, Ni(NO 3)2. What is the concentration of the nitrate ions in solution?

THE SOLUTION CONCENTRATION IN TERMS OF MOLARITY OFNITRATE IONS! Given Problem 4 4 0. 08144 m. L solution=40. 00 g Ni(NO 3)2 P 1 L solution ∞ ∞ 1 L = 1000 m. L P ∞ M 1 L solution 7 L solution ∞ = ? mol Ni(NO 3)2 m. L sol. g Ni(NO 3)2 4 40. 00 g Ni(NO 3)2 0. 08144 m. L solution 1 mol Ni(NO 3)2 = 182. 7033 g Ni(NO 3)2 4 mol Ni(NO 3)2 ∞ 1 mol Ni(NO 3)2 182. 733 g Ni(NO 3)2 7 4 1 mol Ni(NO 3)2 = 1 mol NO 3 - ∞ 2 mol NO 3 -ion 1 mol Ni(NO 3)2 ∞ S 5. 375702643 mol NO - ions 3 S The solution concentration is 5. 375 M nitrate ions.

Versus:

THE SOLUTION CONCENTRATION IN TERMS OFMOLARITY OF NICKEL (II) NITRATE. Given Problem 4 4 0. 08144 m. L solution=40. 00 g Ni(NO 3)2 P 1 L solution ∞ ∞ 1 L = 1000 m. L P ∞ M 1 L solution 7 L solution ∞ = ? mol Ni(NO 3)2 m. L sol. g Ni(NO 3)2 4 40. 00 g Ni(NO 3)2 0. 08144 m. L solution 1 mol Ni(NO 3)2 = 182. 7033 g Ni(NO 3)2 4 mol Ni(NO 3)2 ∞ 1 mol Ni(NO 3)2 182. 733 g Ni(NO 3)2 7 4 1 mol Ni(NO 3)2 = 1 mol NO 3 - S 2. 688288255 mol Ni(NO ) 3 2 S The solution concentration is 2. 688 M nickel (II) Nitrate.

Be careful of answering the question!

MOLARITY Allows us to convert between moles and liters of a solution.

DILUTION Process of taking a stock solution and adding water to lower the concentration of the solution. The concentration is becoming more dilute. Dilute solutions have fewer moles of the solute per one liter of solution. Dilute solutions have fewer moels of the solute per the same amount of volume of solution. Moles of solute before dilution = moles of solute after dilution

M CONC X V CONC = M DIL X V DIL Moles of solute before dilution = moles of solute after dilution Mconc x Vconc = Mdil x Vdil M c o n c is the molarity of the concentrated solution V c o n c is the volume of the concentrated solution M d i l is the moalrity of the dilute solution V d i l is the volume of the dilute solution This is just for dilution a solution. This is not for a stoichiometric problem where the solution is changing. The volumes must be the same units for this to work properly If volumes are different, just use unit conversions to change.

SAMPLE: M CONC X V CONC = M DIL X V DIL If you have 400. 0 m. L of a solution of HCl that has a concentration of 12. 00 M HCl and you want to dilute that solution to a concentration of 3. 48 M HCl, how much water would you need to add? Mconc x Vconc = Mdil x Vdil Algebraically rearrange the formula to find what you do not have. Mconc x Vconc = Vdil Mdil

SAMPLE: M CONC X V CONC = M DIL X V DIL If you have 400. 0 m. L of a solution of HCl that has a concentration of 12. 00 M HCl and you want to dilute that solution to a concentration of 3. 48 M HCl, how much water would you need to add? Mconc Mdil Vconc = Vdil Plug and Chug. 12. 00 M HCl 400. 0 m. L HCl = Vdil 3. 48 Mdil HCl

SAMPLE: M CONC X V CONC = M DIL X V DIL If you have 400. 0 m. L of a solution of HCl that has a concentration of 12. 00 M HCl and you want to dilute that solution to a concentration of 3. 48 M HCl, how much water would you need to add? Plug and Chug. 12. 00 M HCl 400. 0 m. L HCl = Vdil 3. 48 Mdil HCl 1379. 310345 m. L = Vdil 1380 m. L = Vdil

MAKE SURE YOU ANSWER THE QUESTION! If you have 400. 0 m. L of a solution of HCl that has a concentration of 12. 00 M HCl and you want to dilute that solution to a concentration of 3. 48 M HCl, how much water would you need to add? Subtract original concentrated volume from diluted volume to find how much to add. 1380 m. Ldil – 400. 0 m. Lconc = amount to add 980 m. L = amount to add

STOICHIOMETRY AND SOLUTIONS Remember to use the Molpellar if needed!

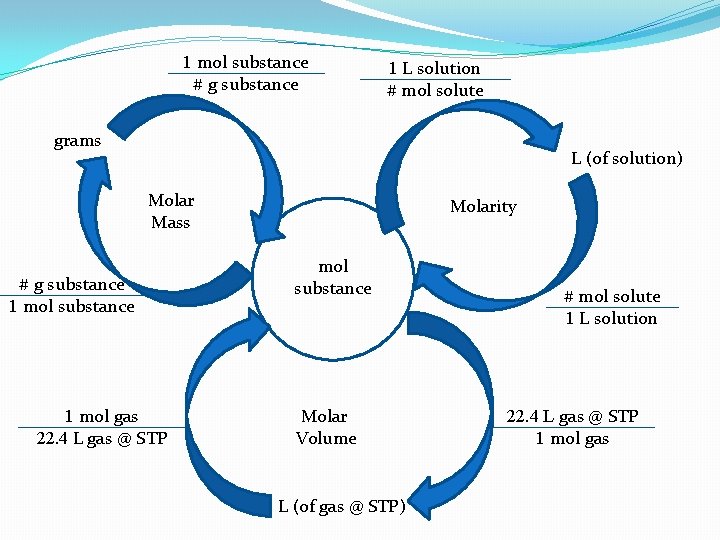
1 mol substance # g substance 1 L solution # mol solute grams L (of solution) Molar Mass # g substance 1 mol gas 22. 4 L gas @ STP Molarity mol substance Molar Volume L (of gas @ STP) # mol solute 1 L solution 22. 4 L gas @ STP 1 mol gas

Stoichiometry and solutions �We will be adding to the molepellar in the future, but the core is always the mole!

TITRATIONS Combining a solution of known solute concentration, standard solution, with a solution of unknown solute concentration.

TITRATIONS Equivalence Point: stoichiometrically equal quantities are reacted. Reactants and products all react. No limiting reactant.

I am so excited that I can titrate better than puny Iron Man! I wish I could titrate.