Reaction Pathways Syllabus Statement Deduce reaction pathways given

Reaction Pathways

Syllabus Statement • Deduce reaction pathways given the starting material and the product. • (This is the same for HL and SL – HL just have to know MORE pathways!)

• Sometimes a product can’t be made directly from the given starting material. • E. g. convert 1 -iodopropane into propanal • This can’t be done directly – we have no mechanism for converting a haloalkane into an aldehyde.

• It is usually easier to approach the problem backwards! • What substance CAN be made into an aldehyde? • We only know 1. • A primary alcohol

• So we know our pathway ends. . . • Alcohol aldehyde • Or more specifically. . . • Propan-1 -ol + [O] propanal + H 2 O • What conditions would we use? • Distil with acidified pot dichromate(VI)

• So can we make a primary alcohol from -iodopropane? • Yes! • Hydrolyse with aqueous OH- 1

• So the full reaction mechanism is. . . • 1 -iodopropane + OH- 1 -propanol +I • 1 -propanol +[O] propanal + H 2 O

Another example. . . • Give a pathway for producing butanone from but-2 -ene. • The only way of making a ketone is from a secondary alcohol. . . • So we must make butan-2 -ol

• We can make butan-2 -ol from but-2 -ene by reacting the alkene with water. (addition reaction) • So the full mechanism • But-2 -ene + H 2 O butan-2 -ol • (this should have H+ catalyst written above the arrow, but I can’t do that on Power. Point) • Butan-2 -ol + [O] butanone • (reflux with hot acidified dichromate(VI)

This can be summarised for SL
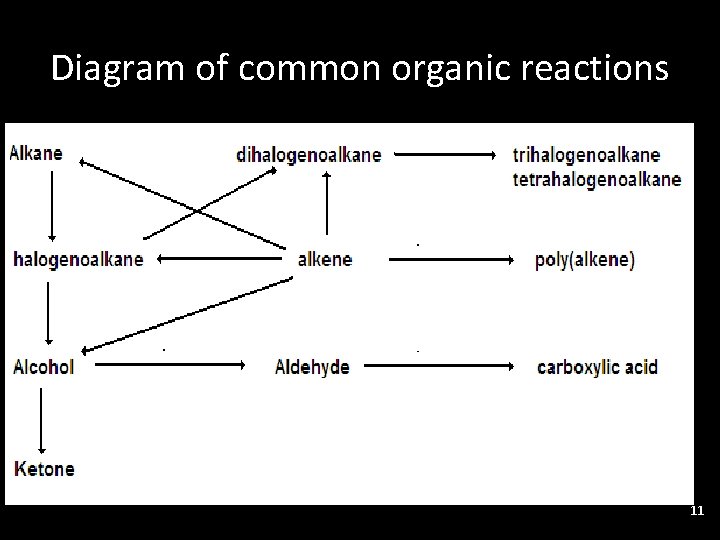
Diagram of common organic reactions • . 11

And the additional bits for HL. . .

Diagram of common organic reactions 13

• I find these summaries confusing. • And they don’t have the conditions needed for the reactions. • So I would like you to produce your own summary in the form of a table.
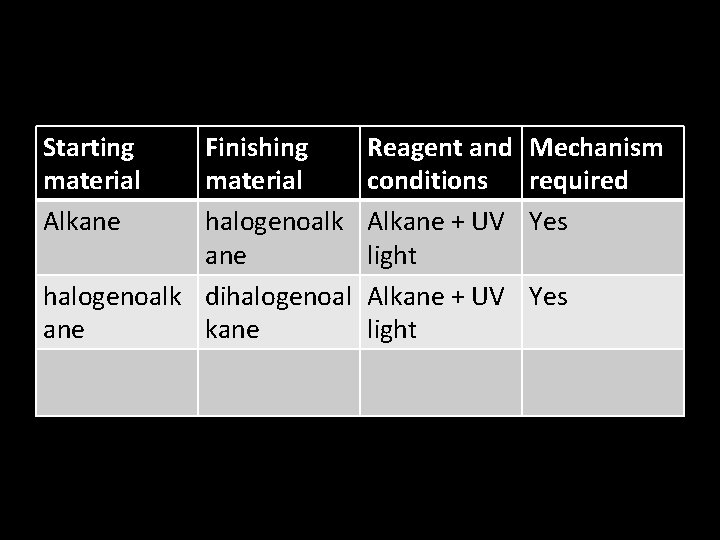
Starting material Alkane Finishing material halogenoalk ane halogenoalk dihalogenoal ane kane Reagent and conditions Alkane + UV light Mechanism required Yes
- Slides: 15