Reaction of alkane Sharifah Zati Hanani Syed Zuber

Reaction of alkane Sharifah Zati Hanani Syed Zuber sharifahzati@unimap. edu. my

Reaction of alkanes • Alkanes have only σ bonds- the electrons in the C-H and C-C are shared equally. • The electrons in C-H and C-C σ bonds- shared equally by the bonding atoms. Thus, none atoms in an alkane has significant charge. • Neither nucleophiles nor electrophiles- thus neither nucleophiles/electrophiles are attracted to them. • Alkanes = relatively unreactive compounds.

Chlorination and bromination • Alkanes react with Cl 2 and Br 2 to form alkyl chlorides/alkyl bromides. • Halogenation reactions- take place at high temp/ in the presence of light (hv). • The only reactions that alkanes involved (w/out metal catalyst) with exception of combustion (burning).

• When a bond breaks so that both of its electrons stay with 1 of the atoms- process called heterolytic bond cleavage/heterolysis • When a bond breaks so that each of the atoms retains one of the bonding electrons- homolytic bond cleavage/homolysis.

Mechanism for monochlorination of methane

Mechanism for the monobromination of ethane • Bromination of alkanes has the same mechanism as chlorination

Exercise • Show the initiation, propagation and termination steps of monochlorination of ethane

Exercise • Write the mechanism for the formation of carbon tetrachloride, CCl 4 from the reaction of methane with Cl 2 and hv. • Show the initiation, termination steps propagation and

Radical stability • Radical stability depends on the no of alkyl groups attached to the carbon with the unpaired electron • Radicals are stabilize by electron-donating alkylgroups • Relative stabilities follow the order of primary, secondary and tertiary alkyl radicals.

Distribution of products depends on probability and reactivity • 2 different alkyl halides are obtained from the monochloronation of butane. • Substitution of a H bonded to 1 of the terminal carbon produces 1 -chlorobutane • Substitution of a H bonded to 1 of the internal carbons forms 2 -chlorobutane.

• The more stable the radical, the more easier it is formed- the stability of radical is reflected in the stability of transition state leading to its formation.

• At room temp. it is 5. 0 times easier for Cl radical to remove H atom from tertiary C than from a primary C, and it is 3. 8 times easier to remove H atom from secondary C than from a primary C.

• To determine the relative amount of different products obtained from radical chlorination of an alkane, consider both probability and reactivity • Probability= the no of H that can be removed that will lead to product formation • Reactivity= relative rate at which a particular H is removed.

• When both factors are considered:

The Reactivity-selectivity principle • The relative rates of radical formation by a bromine radical are different from the relative rates of radical formation by a chlorine radical. • Radical bromination is more selective than radical chlorination:

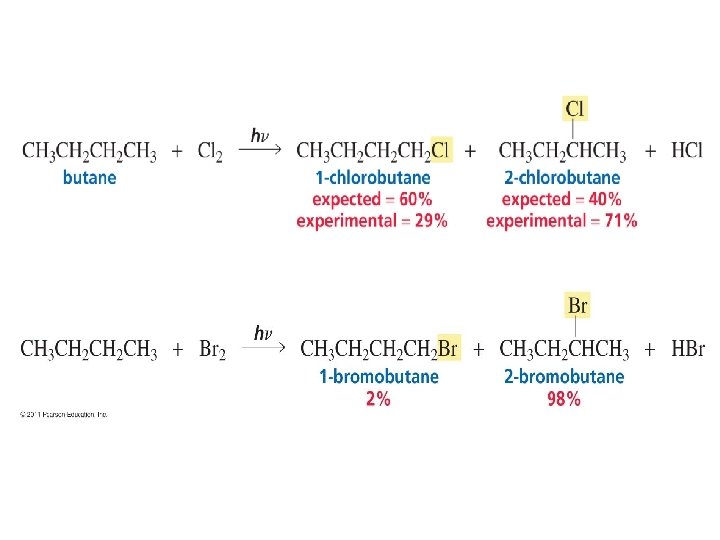

Why are the relative rates of radical formation is different? • Relative rates of radical formation is different when a Br radical rather than Cl radical is used as the H-removing reagent is due to: - Bromination is a much slower reaction than chlorination - The activation energy for removing a H atom by Br radical is 4. 5 times greater than that for removing a H atom by a Cl radical.
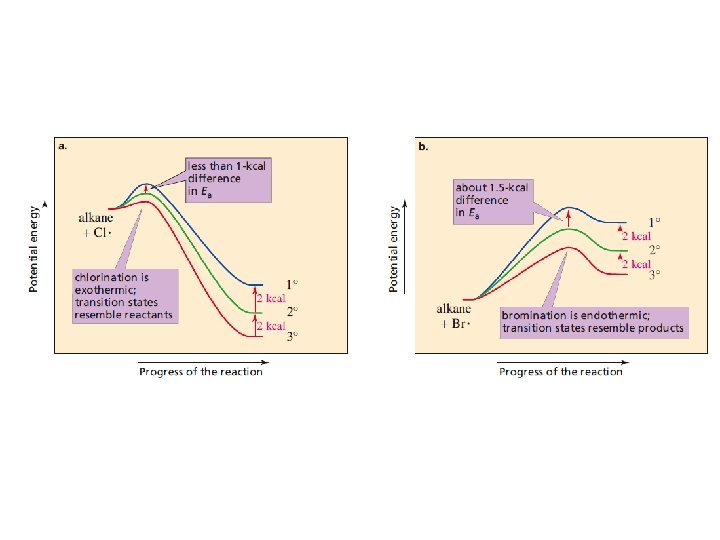

• Since Br radical is relatively unreactive- it is highly selective of which H atom it removes • The more reactive Cl radical is consider less selective • The reactivity-selectivity principle: the greater the reactivity of a species, the less selective it will be. • The fluorine radical is the most reactive halogen radical- reacts violently with alkanes • Iodine is the least reactive of the halogen radicals. It unable to remove H atom from an alkane.
- Slides: 20