Reaction Mechanisms KINETICS including steadystate approximation Reaction Mechanisms






![Another example: overall reaction: 2 NOCl 2 NO + Cl 2 rate = k[NOCl] Another example: overall reaction: 2 NOCl 2 NO + Cl 2 rate = k[NOCl]](https://slidetodoc.com/presentation_image_h2/7c830f6cfc00ce8db808019ca4c1676a/image-10.jpg)




- Slides: 16

Reaction Mechanisms KINETICS including steady-state approximation

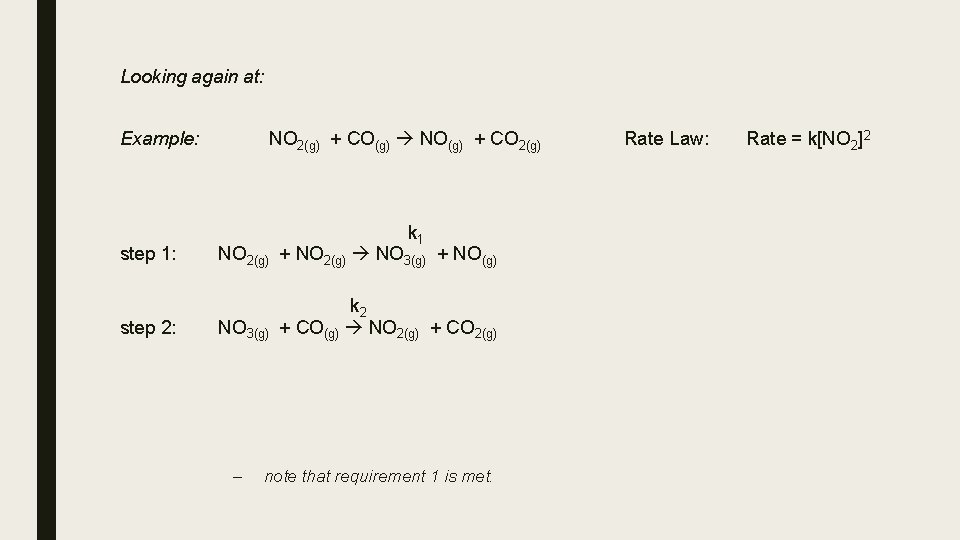
Reaction Mechanisms Most chemical reactions occur by a series of steps called the reaction mechanism. Example: NO 2(g) + CO(g) NO(g) + CO 2(g) Rate Law: Rate = k[NO 2]2 This reaction is actually more complicated than it appears from the balanced equation. The balanced equation only gives information about stoichiometry, reactants and products. There is no information about the reaction mechanism. The above reaction has been determined to proceed in two steps: step 1: k 1 NO 2(g) + NO 2(g) NO 3(g) + NO(g) step 2: k 2 + CO(g) NO 2(g) + CO 2(g) NO 3(g) SLOW FAST k 1 and k 2 are rate constants for the reaction in each step NO 3 is an intermediate – it is formed and consumed during the reaction sequence Each of these two reactions is called an elementary step – a reaction whose rate law can be written from its

Molecularity – number of species that must collide to produce the reaction indicated by that step Ø Unimolecular step – reaction involving one molecule Ø Bimolecular step – reaction involving 2 molecules Ø Termolecular step – reaction involving 3 molecules (quite rare since the probability of three molecules simultaneously colliding is v. small) elementary reaction or step rate law for that step A products Rate = k[A] A + B products Rate = k[A][B] 2 A products Rate = k[A]2 2 A + B products Rate = k[A]2[B]

■ AP practice question

Definition for reaction mechanism: A series of elementary steps that must satisfy two requirements: 1) The sum of the elementary steps must give the overall balanced equation for the reaction. 2) The mechanism must agree with the experimentally determined rate law.

Looking again at: Example: NO 2(g) + CO(g) NO(g) + CO 2(g) k 1 step 1: NO 2(g) + NO 2(g) NO 3(g) + NO(g) step 2: k 2 + CO(g) NO 2(g) + CO 2(g) NO 3(g) – note that requirement 1 is met. Rate Law: Rate = k[NO 2]2

To see about requirement 2, we must introduce a new concept: The rate determining step. It is the step in a multi-step reaction that is the slowest and thus determines the speed of the overall reaction. The reaction cannot be faster than the rate determining step.

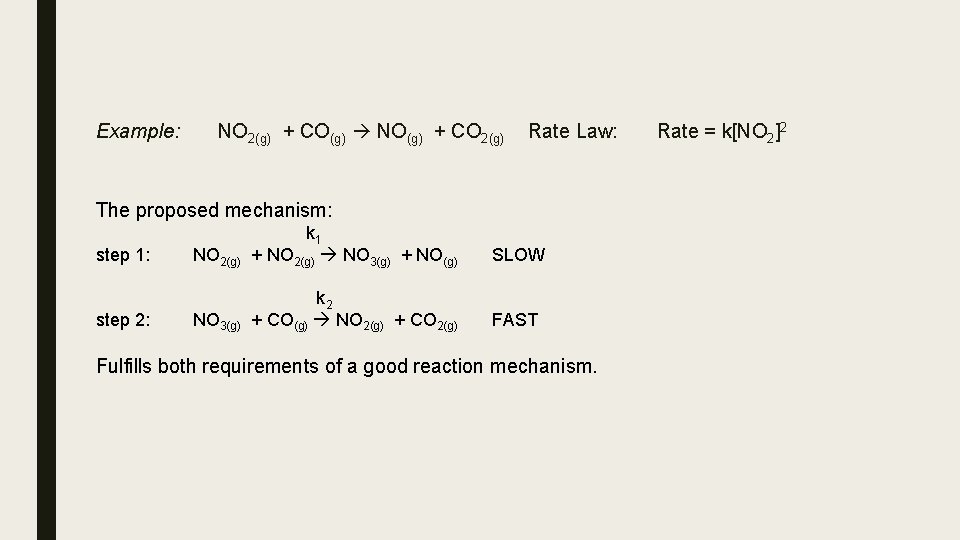
step 1: step 2: NO 2(g) k 1 + NO 2(g) NO 3(g) + NO(g) SLOW rate = k 1[NO 2]2 NO 3(g) k 2 + CO(g) NO 2(g) + CO 2(g) FAST rate = k 2[NO 3][CO] The first step is slow so it is the rate determining step so: rate = k 1[NO 2]2 Ø since NO 2 is involved in this step and CO is not. This step is bimolecular since 2 NO 2 molecules collide in this step; so the exponent is 2. So overall: rate = k 1[NO 2]2 rate law which is consistent with the experimentally determined

Example: NO 2(g) + CO(g) NO(g) + CO 2(g) Rate Law: The proposed mechanism: step 1: step 2: NO 2(g) k 1 + NO 2(g) NO 3(g) + NO(g) SLOW NO 3(g) k 2 + CO(g) NO 2(g) + CO 2(g) FAST Fulfills both requirements of a good reaction mechanism. Rate = k[NO 2]2
![Another example overall reaction 2 NOCl 2 NO Cl 2 rate kNOCl Another example: overall reaction: 2 NOCl 2 NO + Cl 2 rate = k[NOCl]](https://slidetodoc.com/presentation_image_h2/7c830f6cfc00ce8db808019ca4c1676a/image-10.jpg)
Another example: overall reaction: 2 NOCl 2 NO + Cl 2 rate = k[NOCl] (from expt) elementary steps: (proposed) slow: NOCl fast: Cl + NOCl NO + Cl 2 rate = k 1[NOCl] rate=k 2[Cl][NOCl]

Example question: A chemist proposes the following reaction mechanism for a certain reaction. Step 1: A + B C (slow) Step 2: C + A E + F (fast) Write the reaction equation and rate law that is consistent with this mechanism.

Example question: The following reaction mechanism for a certain reaction is proposed Step 1: A + B C (slow) Step 2: C + A E + B (fast) Write the reaction equation and rate law that is consistent with this mechanism. What is the role of C? What is the role of B?

■ AP practice questions

Steady-State Approximation When a reaction mechanism has several steps and the first one is not ratedetermining, a method called the steady-state approximation is used to develop the rate law. The steady-state approximation implies that you select an intermediate in the reaction mechanism, and calculate its concentration by assuming that it is consumed as quickly as it is generated.


■ AP practice questions