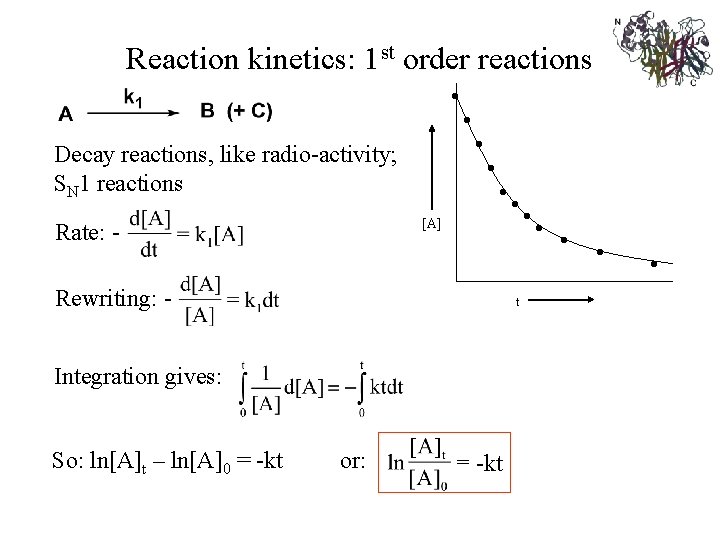
Reaction kinetics 1 st order reactions Decay reactions

![Enzyme Kinetics ß-fructofuranosidase: Sucrose + H 2 O glucose + fructose When [S] » Enzyme Kinetics ß-fructofuranosidase: Sucrose + H 2 O glucose + fructose When [S] »](https://slidetodoc.com/presentation_image/3fff3caf21af6e94bfd2771c992e2087/image-4.jpg)

![The Michaelis-Menten Equation Letting [E] = [E]T - [ES] and rearranging yields The Michaelis The Michaelis-Menten Equation Letting [E] = [E]T - [ES] and rearranging yields The Michaelis](https://slidetodoc.com/presentation_image/3fff3caf21af6e94bfd2771c992e2087/image-7.jpg)




![kcat/KM Is a Measure of Catalytic Efficiency When [S] « KM, very little ES kcat/KM Is a Measure of Catalytic Efficiency When [S] « KM, very little ES](https://slidetodoc.com/presentation_image/3fff3caf21af6e94bfd2771c992e2087/image-18.jpg)




- Slides: 36

Reaction kinetics: 1 st order reactions • Decay reactions, like radio-activity; SN 1 reactions [A] Rate: - • • • Rewriting: - t Integration gives: So: ln[A]t – ln[A]0 = -kt or: -kt • • •

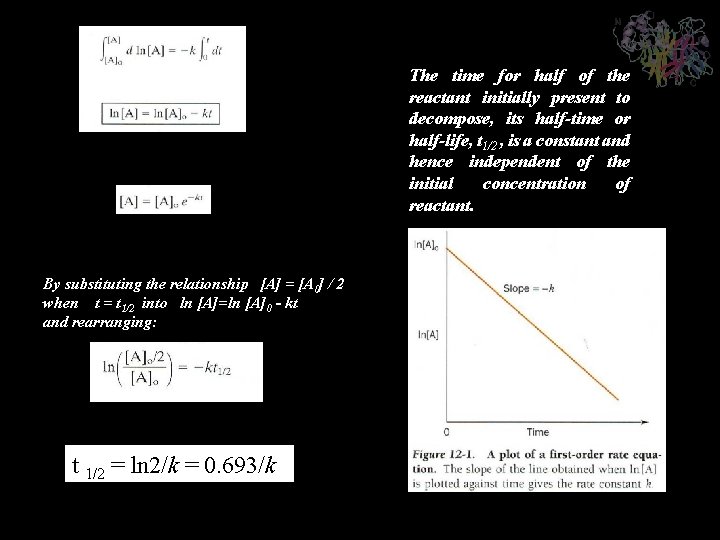
The time for half of the reactant initially present to decompose, its half-time or half-life, t 1/2 , is a constant and hence independent of the initial concentration of reactant. By substituting the relationship [A] = [A 0] / 2 when t = t 1/2 into ln [A]=ln [A]0 - kt and rearranging: t 1/2 = ln 2/k = 0. 693/k

Second-order reaction 2 A The half-time for a second order reaction is expressed P t 1/2 = 1/k [A]0 and therefore, in contrast to a first order reaction depends on the initial reactant concentration. A+B P Here, the reaction is said to be first order in A and first order in B. Unimolecular and bimolecular reactions are common. Termolecular reactions are unusual because the simultaneous collision of three molecules is a rare event. Fourth and higher order reactions are unknown.
![Enzyme Kinetics ßfructofuranosidase Sucrose H 2 O glucose fructose When S Enzyme Kinetics ß-fructofuranosidase: Sucrose + H 2 O glucose + fructose When [S] »](https://slidetodoc.com/presentation_image/3fff3caf21af6e94bfd2771c992e2087/image-4.jpg)
Enzyme Kinetics ß-fructofuranosidase: Sucrose + H 2 O glucose + fructose When [S] » [E] : the rate is zero order with respect to sucrose

The Michaelis-Menten Equation This equation cannot be explicitly integrated, however, without simplifying assumptions, two possibilities are 1. Assumption of equilibrium. Leonor Michaelis and Maud Menten, building on the work of Victor Henri, assumed that k-1 » k 2, so that the first step of the reaction reaches equilibrium. Ks is the dissociation constant of the first step in the enzymatic reaction

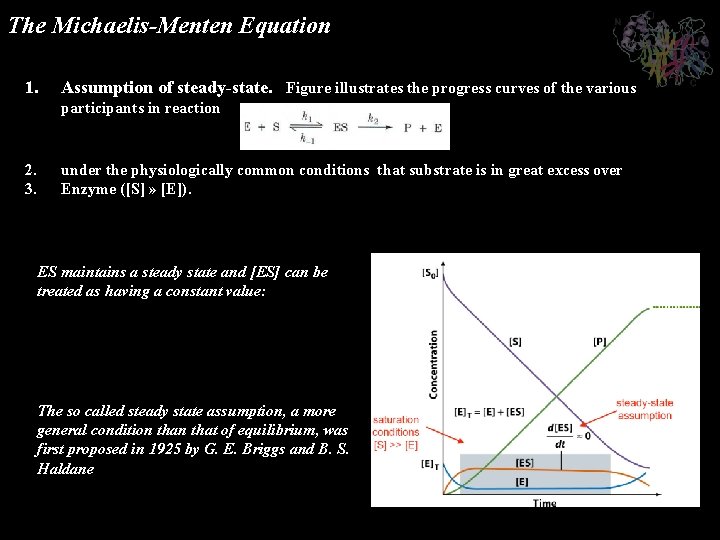
The Michaelis-Menten Equation 1. Assumption of steady-state. Figure illustrates the progress curves of the various participants in reaction 2. 3. under the physiologically common conditions that substrate is in great excess over Enzyme ([S] » [E]). ES maintains a steady state and [ES] can be treated as having a constant value: The so called steady state assumption, a more general condition that of equilibrium, was first proposed in 1925 by G. E. Briggs and B. S. Haldane
![The MichaelisMenten Equation Letting E ET ES and rearranging yields The Michaelis The Michaelis-Menten Equation Letting [E] = [E]T - [ES] and rearranging yields The Michaelis](https://slidetodoc.com/presentation_image/3fff3caf21af6e94bfd2771c992e2087/image-7.jpg)
The Michaelis-Menten Equation Letting [E] = [E]T - [ES] and rearranging yields The Michaelis constant, KM , is defined as Solving for [ES],

The Michaelis-Menten Equation The expression of the initial velocity (v 0) of the reaction, the velocity at t=0, thereby becomes The maximal velocity of a reaction, Vmax occurs at high substrate concentrations when the enzyme is saturated, that is, when it is entirely in the ES form Therefore, combining the last two equations, we obtain: This expression, the Michaelis-Menten equation, is the basic equation of enzyme kinetic.

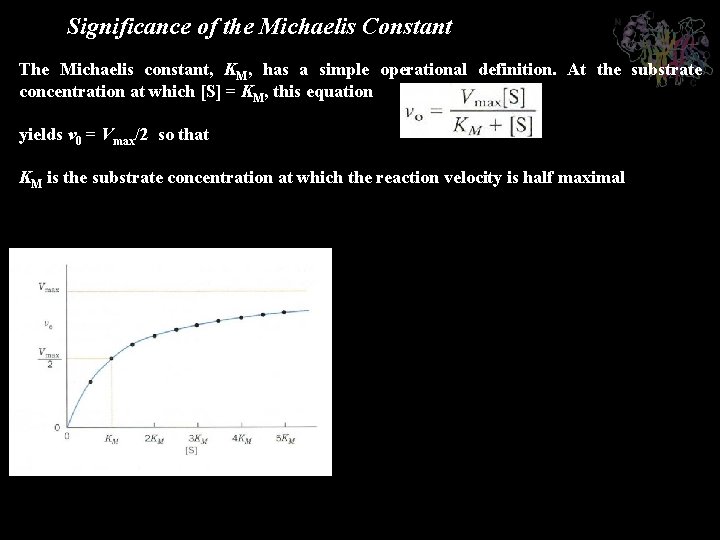
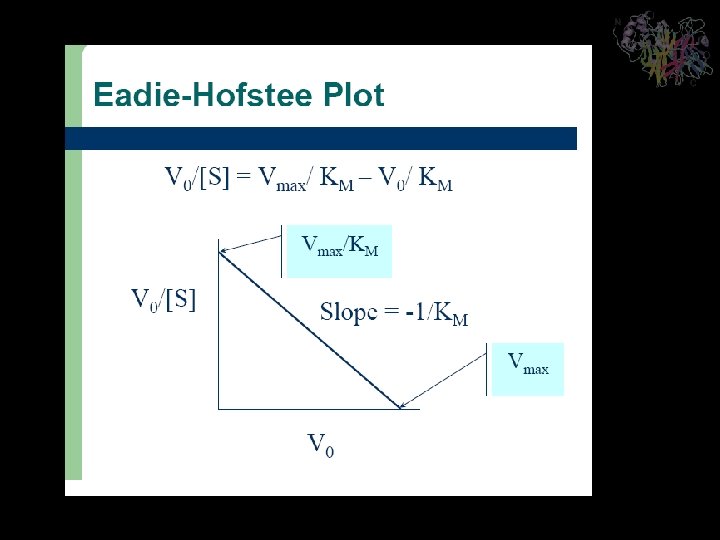
Significance of the Michaelis Constant The Michaelis constant, KM, has a simple operational definition. At the substrate concentration at which [S] = KM, this equation yields v 0 = Vmax/2 so that KM is the substrate concentration at which the reaction velocity is half maximal

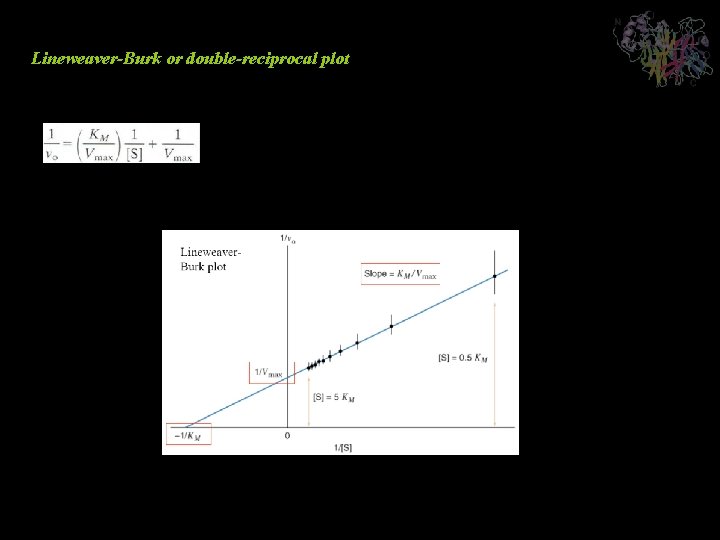
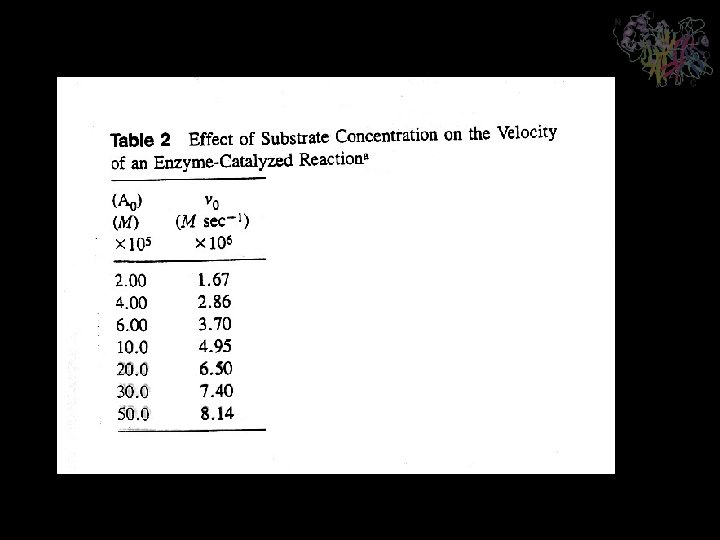
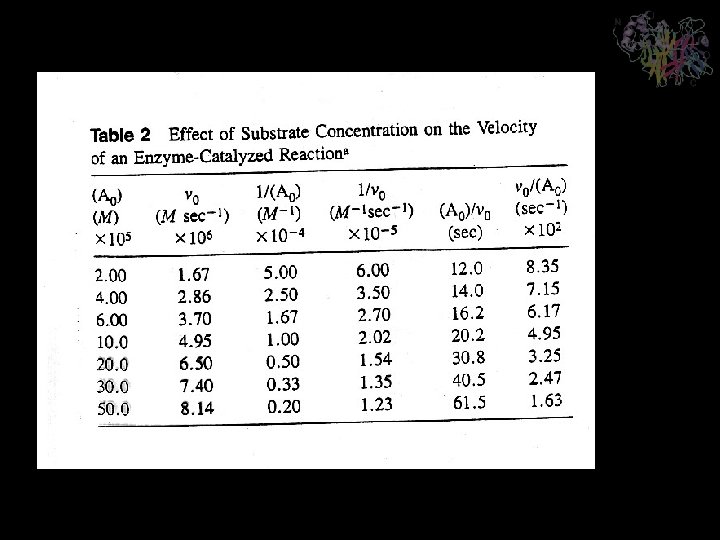
Lineweaver-Burk or double-reciprocal plot Analysis of Kinetic Data


S >> Km vi=Vmax= k 2 Et



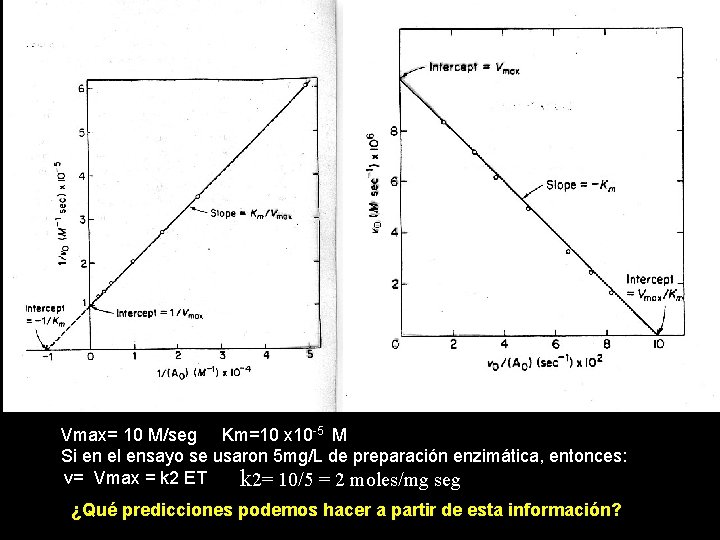
Vmax= 10 M/seg Km=10 x 10 -5 M Si en el ensayo se usaron 5 mg/L de preparación enzimática, entonces: v= Vmax = k 2 ET k 2= 10/5 = 2 moles/mg seg ¿Qué predicciones podemos hacer a partir de esta información?

Significance of the Michaelis Constant The magnitude of KM varies widely with the identity of the enzyme and the nature of the substrate. It is also a function of temperature and p. H. The Michaelis constant can be expressed as Since Ks is the dissociation constant of the Michaelis complex, as Ks decreases, the enzyme’s affinity for substrate increases. KM in therefore also a measure of the affinity of the enzyme for its substrate, provided k 2/k 1 is small compared to Ks, that is k 2 ‹ k-1 so that the ES P reaction proceeds more slowly than ES reverts to E + S kcat/KM Is a Measure of Catalytic Efficiency We can define the catalytic constant, kcat, of an enzyme as This quantity is also known as the turnover number of an enzyme because it is the number of reaction processes (turnovers) that each active site catalyzes per unit time.

Turn Over Numbers of Enzymes Substrate Catalase H 2 O 2 Carbonic anhydrase HCO 3 - 400, 000 Acetylcholinesterase Acetylcholine 140, 000 b-Lactamase Benzylpenicillin Fumarase Fumarate Rec. A protein (ATPase) ATP kcat (s-1) 40, 000 2, 000 800 0. 4 The number of product transformed from substrate by one enzyme molecule in one second Adapted from Nelson & Cox (2000) Lehninger Principles of Biochemistry (3 e) p. 263
![kcatKM Is a Measure of Catalytic Efficiency When S KM very little ES kcat/KM Is a Measure of Catalytic Efficiency When [S] « KM, very little ES](https://slidetodoc.com/presentation_image/3fff3caf21af6e94bfd2771c992e2087/image-18.jpg)
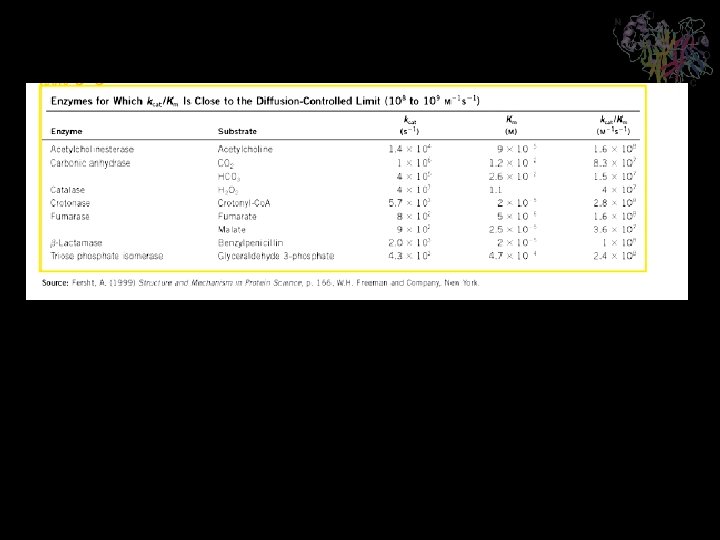
kcat/KM Is a Measure of Catalytic Efficiency When [S] « KM, very little ES is formed. Consequently, [E] ≈ [E]T, so reduces to a second-order rate equation: The quantity kcat/KM is a measure of an enzyme’s catalytic efficiency. There is an upper limit to the value of kcat/KM : It can be not greater than k 1; that is, the decomposition of ES to E + P can occur no more frequently than E and S come together to form ES. The most efficient enzymes have kcat/KM values near to the diffusion-controlled limit of 108 to 109 M-1. s-1


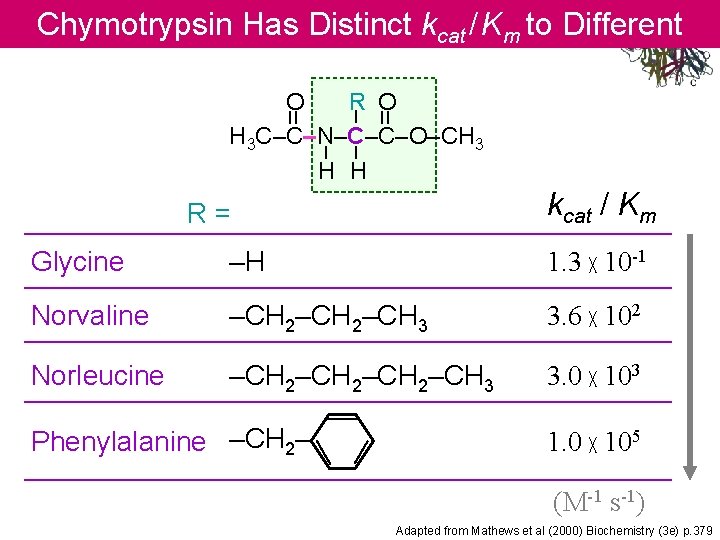
Chymotrypsin Has Distinct kcat / Km to Different Substrates = – – – = O R O H 3 C–C–N–C–C–O–CH 3 H H kcat / Km R= Glycine –H 1. 3 ╳ 10 -1 Norvaline –CH 2–CH 3 3. 6 ╳ 102 Norleucine –CH 2–CH 3 3. 0 ╳ 103 Phenylalanine –CH 2– 1. 0 ╳ 105 (M-1 s-1) Adapted from Mathews et al (2000) Biochemistry (3 e) p. 379

Al iniciar: t = 0, S = So A cualquier tiempo: T=t S=S X = (So-S)/So - d. S/dt = vi = So d. X/dt


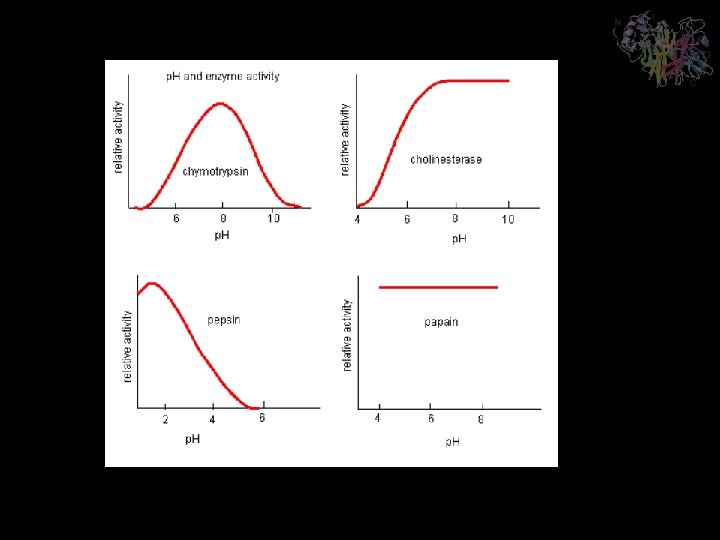
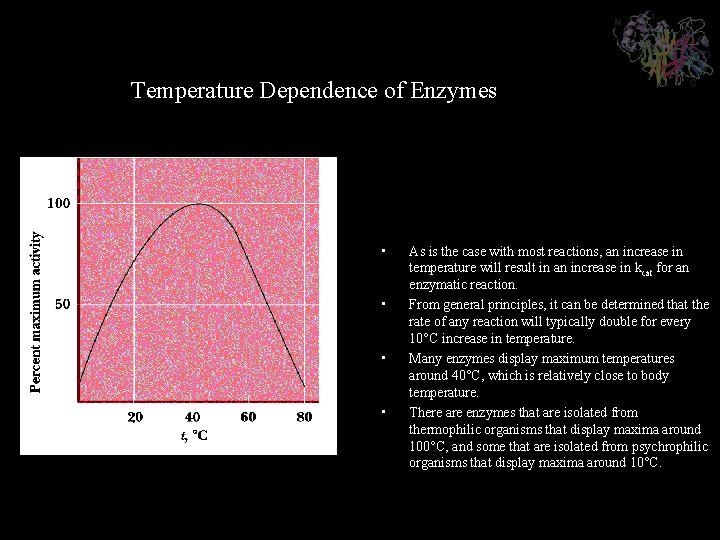
Temperature Dependence of Enzymes • • As is the case with most reactions, an increase in temperature will result in an increase in kcat for an enzymatic reaction. From general principles, it can be determined that the rate of any reaction will typically double for every 10°C increase in temperature. Many enzymes display maximum temperatures around 40°C, which is relatively close to body temperature. There are enzymes that are isolated from thermophilic organisms that display maxima around 100°C, and some that are isolated from psychrophilic organisms that display maxima around 10°C.

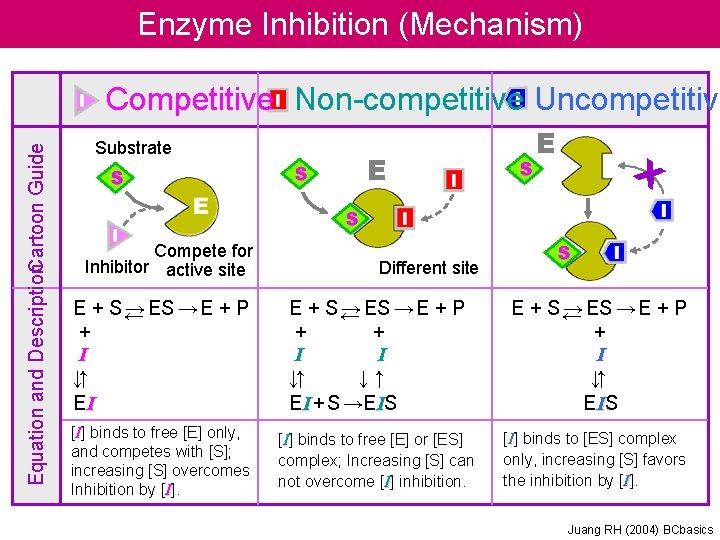
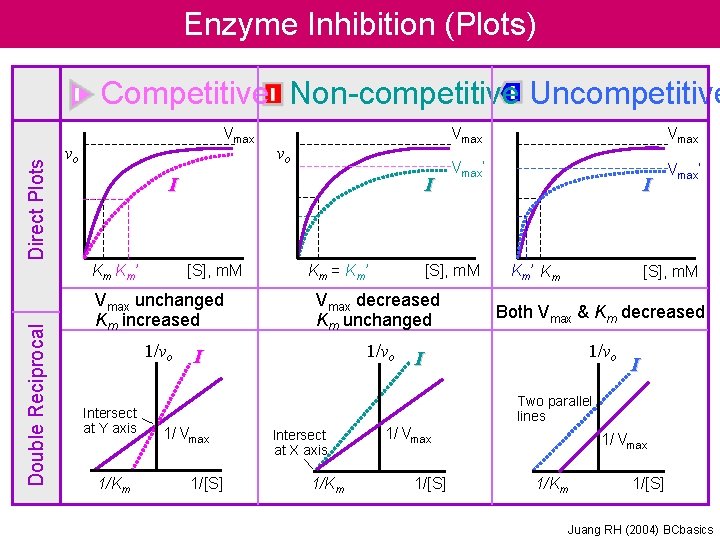
Enzyme Inhibition (Mechanism) Equation and Description. Cartoon Guide Competitive Non-competitive Uncompetitive Substrate Compete for Inhibitor active site E E X Different site E + S← → ES → E + P + I ↓↑ EI E + S← → ES → E + P + + I I ↓↑ ↓↑ EI + S →EIS E + S← → ES → E + P + I ↓↑ E IS [I] binds to free [E] only, and competes with [S]; increasing [S] overcomes Inhibition by [I] binds to free [E] or [ES] complex; Increasing [S] can not overcome [I] inhibition. [I] binds to [ES] complex only, increasing [S] favors the inhibition by [I]. Juang RH (2004) BCbasics

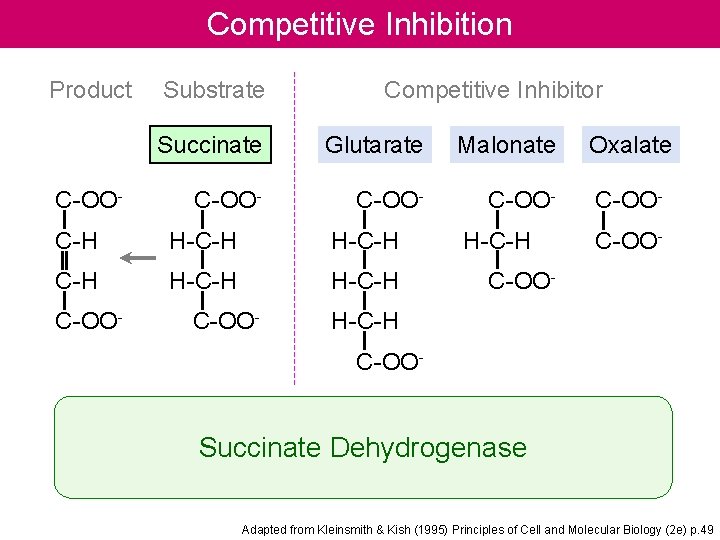
Competitive Inhibition Product C-OO- Substrate Competitive Inhibitor Succinate Glutarate Malonate C-OO- C-H H-C-H C-OO- H-C-H Oxalate C-OO- H-C-H C-OO- Succinate Dehydrogenase Adapted from Kleinsmith & Kish (1995) Principles of Cell and Molecular Biology (2 e) p. 49

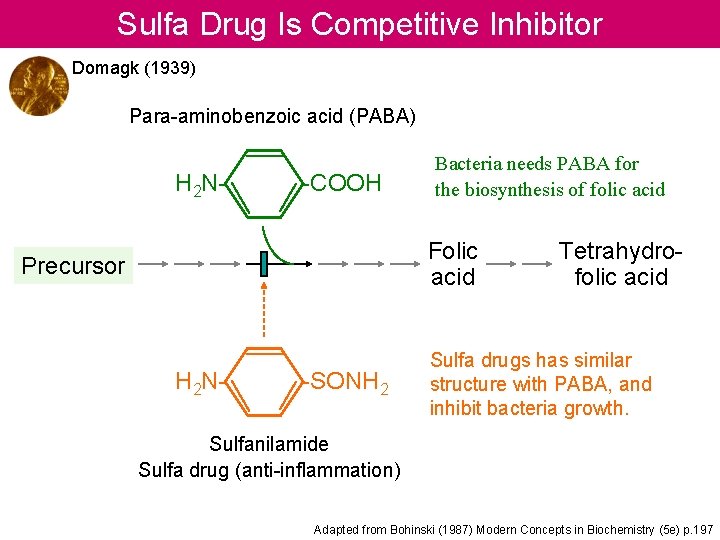
Sulfa Drug Is Competitive Inhibitor Domagk (1939) Para-aminobenzoic acid (PABA) H 2 N- -COOH Bacteria needs PABA for the biosynthesis of folic acid Folic acid Precursor H 2 N- -SONH 2 Tetrahydrofolic acid Sulfa drugs has similar structure with PABA, and inhibit bacteria growth. Sulfanilamide Sulfa drug (anti-inflammation) Adapted from Bohinski (1987) Modern Concepts in Biochemistry (5 e) p. 197

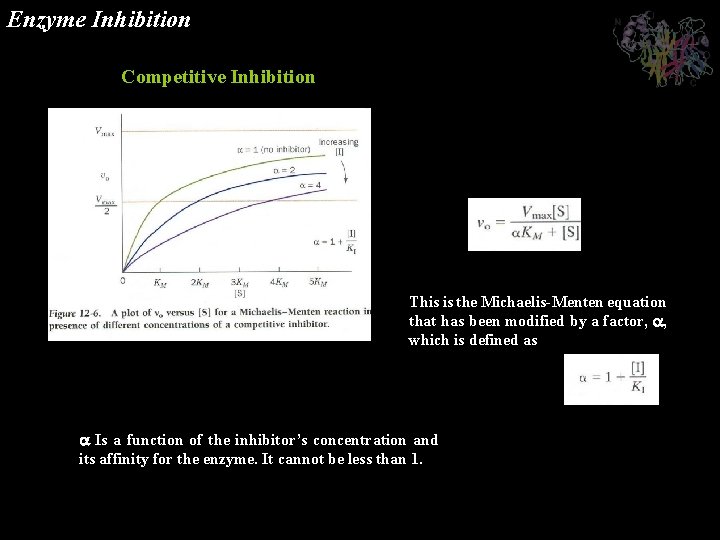
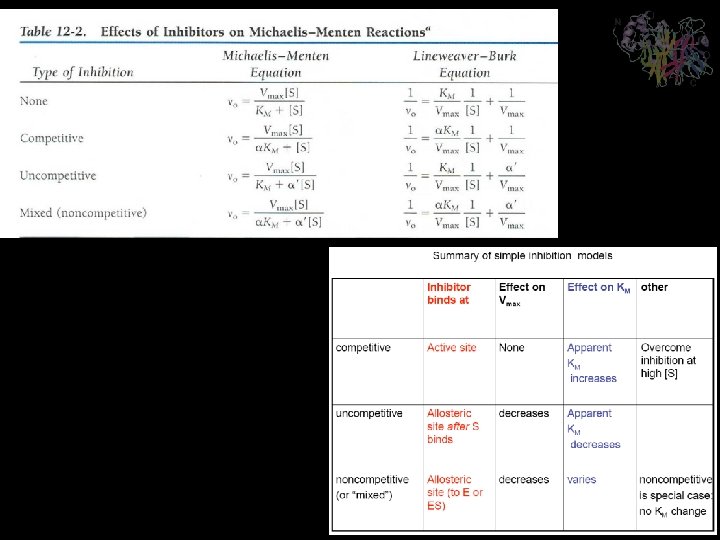
Enzyme Inhibition Many substances alter the activity of an enzyme by reversibly combining with it in a way what influence the binding of substrate and/or its turnover number. Substances that reduce an enzyme’s activity in this way are known as inhibitors Competitive Inhibition A substance that competes directly with a normal substrate for an enzyme’s substratebinding site is known as a competitive inhibitor. Here it is assumed that I, the inhibitor, bind reversibly to the enzyme and is in a rapid equilibrium with it so that And EI, the enzyme-inhibitor complex, is catalytically inactive. A competitive inhibitor therefore reduces the concentration of free enzyme available for substrate binding.

Enzyme Inhibition Competitive Inhibition This is the Michaelis-Menten equation that has been modified by a factor, , which is defined as Is a function of the inhibitor’s concentration and its affinity for the enzyme. It cannot be less than 1.

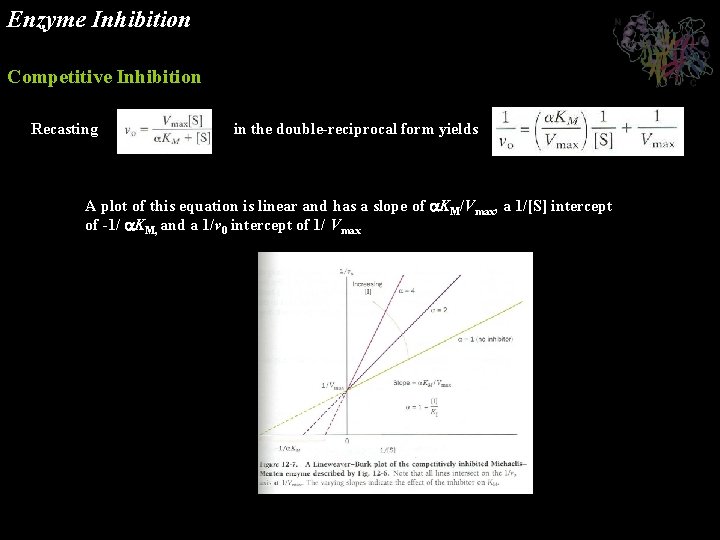
Enzyme Inhibition Competitive Inhibition Recasting in the double-reciprocal form yields A plot of this equation is linear and has a slope of KM/Vmax, a 1/[S] intercept of -1/ KM, and a 1/v 0 intercept of 1/ Vmax

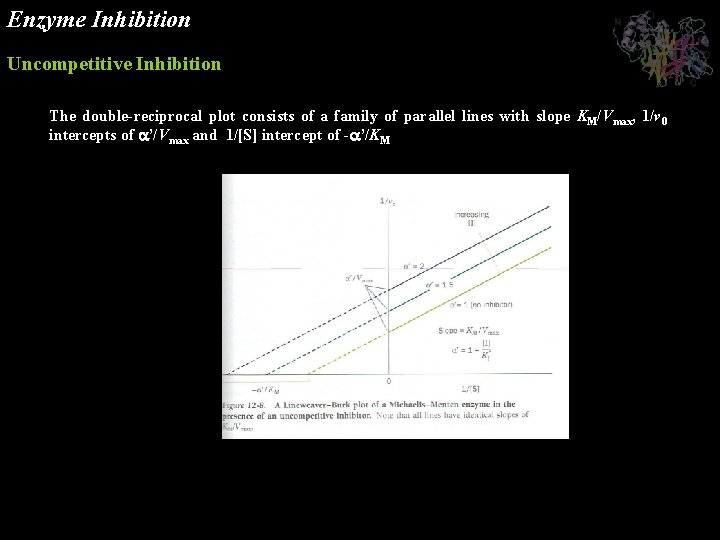
Enzyme Inhibition Uncompetitive Inhibition In uncompetitive inhibition, the inhibitor binds directly to the enzyme-substrate complex but not to the free enzyme In this case, the inhibitor binding step has the dissociation constant The uncompetitive inhibitor, which need not resemble the substrate, presumably distorts the active site, thereby rendering the enzyme catalytically inactive.

Enzyme Inhibition Uncompetitive Inhibition The double-reciprocal plot consists of a family of parallel lines with slope KM/Vmax, 1/v 0 intercepts of ’/Vmax and 1/[S] intercept of - ’/KM

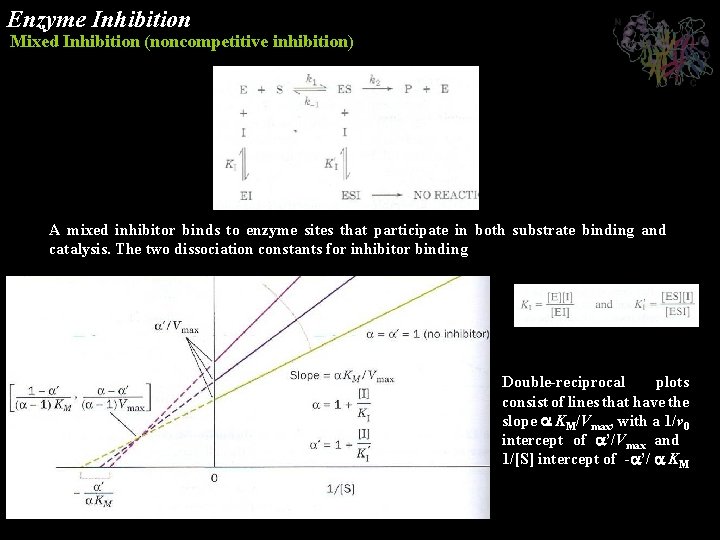
Enzyme Inhibition Mixed Inhibition (noncompetitive inhibition) A mixed inhibitor binds to enzyme sites that participate in both substrate binding and catalysis. The two dissociation constants for inhibitor binding Double-reciprocal plots consist of lines that have the slope KM/Vmax, with a 1/v 0 intercept of ’/Vmax and 1/[S] intercept of - ’/ KM

Enzyme Inhibition (Plots) Direct Plots Competitive Non-competitive Uncompetitive Vmax vo I Km Km’ Double Reciprocal vo I [S], m. M Km = Km’ Vmax’ [S], m. M I Km’ Km [S], m. M Vmax unchanged Km increased Vmax decreased Km unchanged Both Vmax & Km decreased 1/vo Intersect at Y axis 1/Km I I I Two parallel lines 1/ Vmax 1/[S] Intersect at X axis 1/Km 1/ Vmax 1/[S] 1/ Vmax 1/Km 1/[S] Juang RH (2004) BCbasics


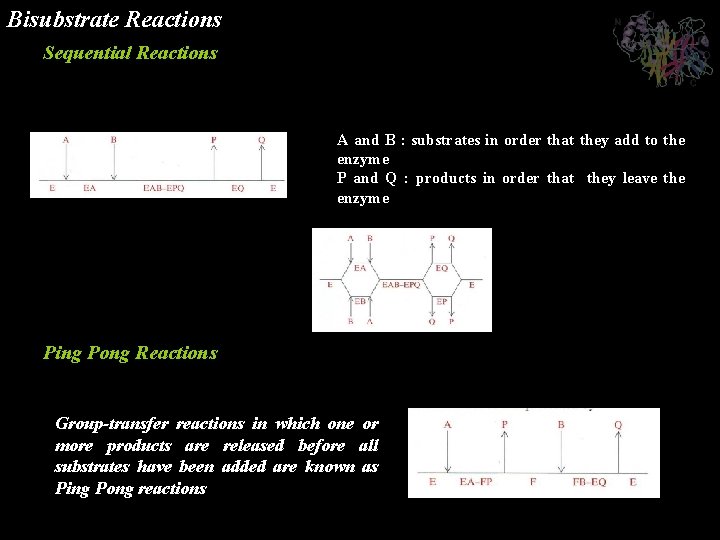
Bisubstrate Reactions Almost all of these so called bisubstrate reactions are either transferase reactions in which enzyme catalyzed the transfer of a specific functional group, X, from one of the substrates to the other: or oxidation-reduction reactions in which reducing equivalents are transferred between two substrates. Sequential Reactions in which all substrates must combine with the enzyme before a reaction can occur and products be released are known as Sequential reactions

Bisubstrate Reactions Sequential Reactions Ordered bisubstrate reaction A and B : substrates in order that they add to the enzyme P and Q : products in order that they leave the enzyme Random bisubstrate reaction Ping Pong Reactions Group-transfer reactions in which one or more products are released before all substrates have been added are known as Ping Pong reactions