Reaction Engineering Continuous culture Fin Fout 0 V







































- Slides: 51

Reaction Engineering

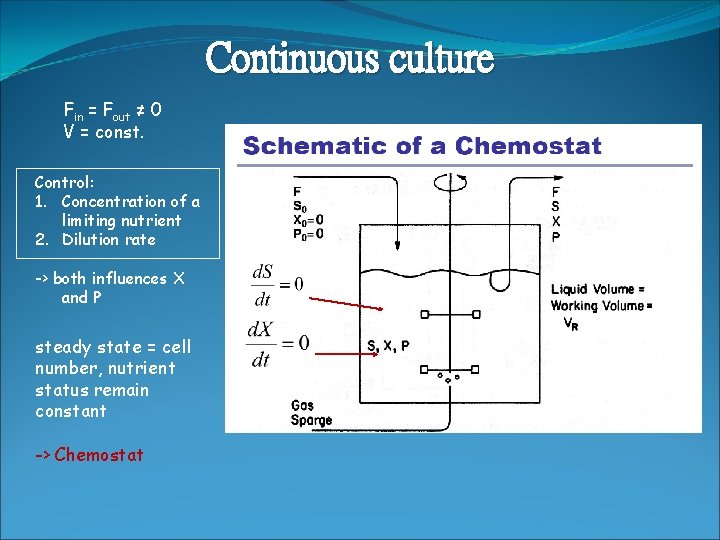
Continuous culture Fin = Fout ≠ 0 V = const. Control: 1. Concentration of a limiting nutrient 2. Dilution rate -> both influences X and P steady state = cell number, nutrient status remain constant -> Chemostat


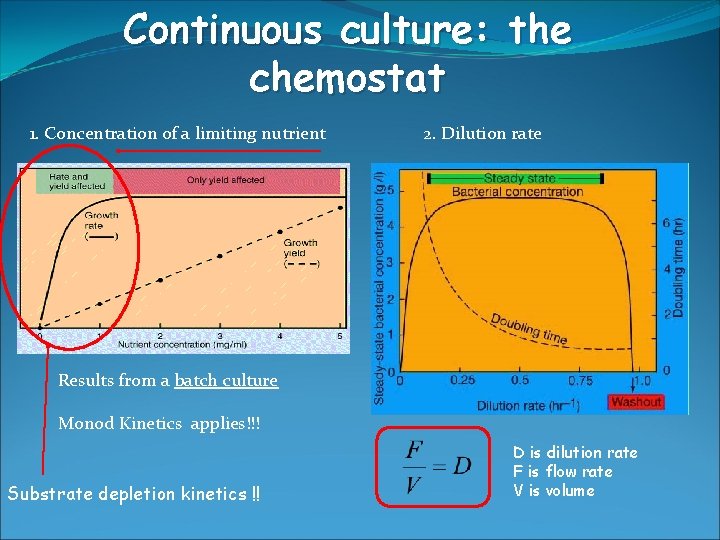
Continuous culture: the chemostat 1. Concentration of a limiting nutrient 2. Dilution rate Results from a batch culture Monod Kinetics applies!!! Substrate depletion kinetics !! D is dilution rate F is flow rate V is volume

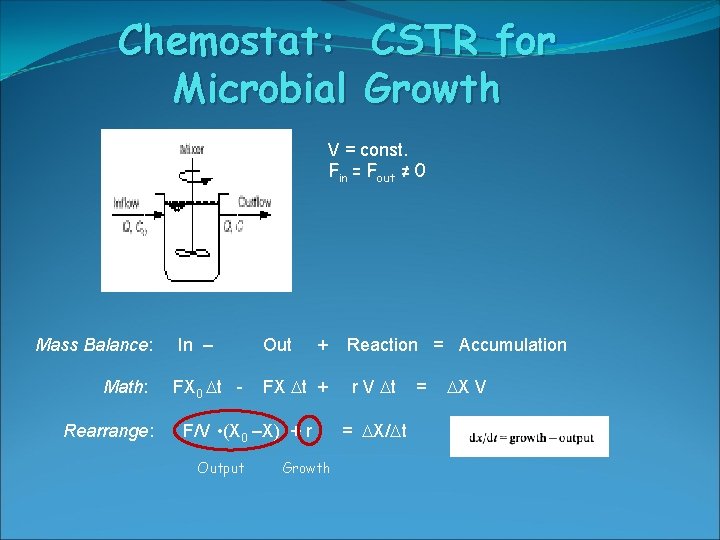
Chemostat: Microbial V = const. Fin = Fout ≠ 0 CV Mass Balance: Math: Rearrange: CSTR for Growth In – Out + FX 0 t - FX t + r V t F/V • (X 0 –X) + r Output Growth Reaction = Accumulation = X/ t = X V

Chemostat: CFSTR for Microbial Growth Take limits as X and t 0 F/V • (X 0 –X) + r = Substitute exponential growth equation for “r” Set X 0 = 0 (no influent cells) Make steady state (SS) assumption (no net accumulation or depletion): Let F/V = D = dilution rate Rearrange: D=m


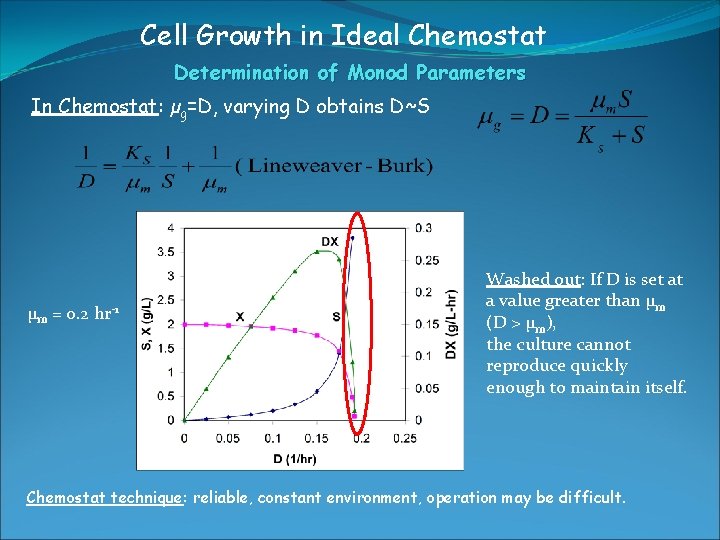
Cell Growth in Ideal Chemostat Determination of Monod Parameters In Chemostat: µg=D, varying D obtains D~S µm = 0. 2 hr-1 Washed out: If D is set at a value greater than µm (D > µm), the culture cannot reproduce quickly enough to maintain itself. Chemostat technique: reliable, constant environment, operation may be difficult.









Fed batch fermentation -> In batch reactor, S and X are high. No transport of S or X and no control on µ. -> In chemostat, S and X are low. Transport of S or X and control on µ. -> In fed batch reactor. Substrate transport in, not out. No biomass transport. Why fed batch? 1. Low S no toxicity / osmotic problem 2. High X high P easier downstream processing 3. Control of µ?

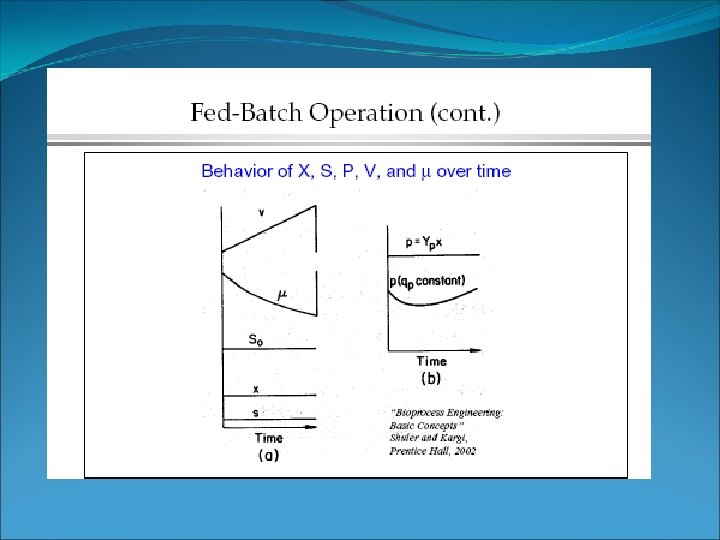
Fed batch fermentation Start feeding S 0 S Batch phase Feeding phase under substrate limited conditions S = 1 – 50 mg/l. S 0 5000 – 20000 mg/l time In substrate limited feeding phase, S is very low. Thus, one can use the pseudo steady state condition for substrate mass balance -> Useful for Antibiotic fermentation -> to overcome substrate inhibition!!



Fed batch §Substrate balance – no outflow (Fcout = 0), sterile feed §St = SV and Xt = XV (mass of substrate or cells in reactor at a given time) §S 0 = substrate in feed stream substrate in Substrate balance Cell balance substrate consumed no substrate out (Flow out = 0)

Fed-batch Cell balance – sterile feed This can be a steady state reactor if substrate is consumed as fast as it enters (quasi-steady-state). Then d. X/dt = 0 and μ = D, like in a chemostat. Recall, D = F / V


Fed batch • What this means • the total amount of cells in the reactor increases with time -> with increasing V • dilution rate and μ decrease with time in fed batch culture • Since μ = D, the growth rate is controlled by the dilution rate.


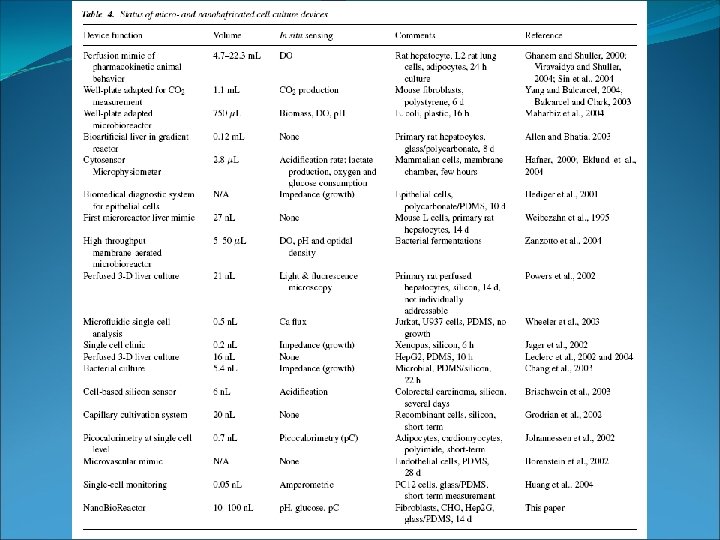
Minibioreactors -> Volumes below 100 ml Characterized by: -> area of application -> mass transfer -> mixing characteristics

Minibioreactors Why do we want to scale down ? - Parallelization (optimization, screening) - automatization - cost reduction What can you optimize? - Biocatalyst (organism) design - medium (growth conditions) design - process design

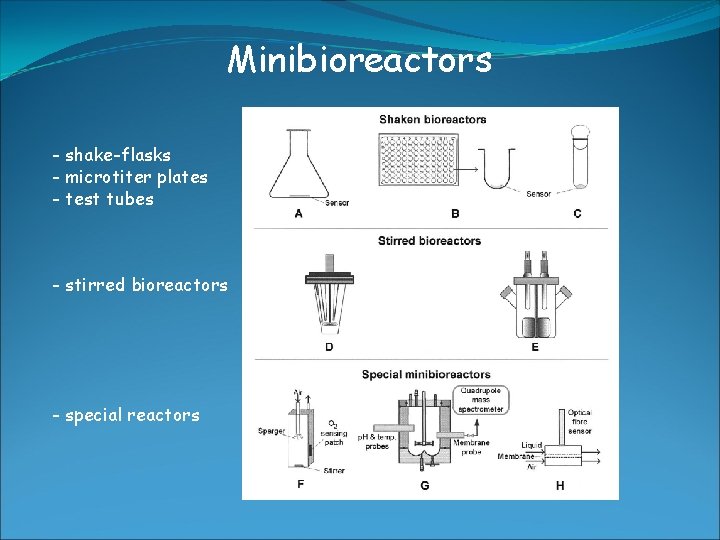
Minibioreactors - shake-flasks - microtiter plates - test tubes - stirred bioreactors - special reactors

Minibioreactors Shaking flasks: -> easy to handle -> low price -> volumne 25 ml – 5 L (filled with medium 20% of volumne) -> available with integrated sensors (O 2, p. H) -> limitation: O 2 limitation (aeration) -> during growth improved by 1. baffled flasks 2. membranes instead of cotton -> during sampling

Minibioreactors Microtiter plates: -> large number of parallel + miniature reactors -> automation using robots -> 6, 12, 24, 48, 96, 384, 1536 well plates -> volumne from 25 μl – 5 ml -> integrated O 2 sensor available Increased throughput rates allow applications: - screening for metabolites, drugs, new biocatalysts (enzymes) - cultivation of clone libraries - expression studies of recombinant clones - media optimization and strain development

Minibioreactors Microtiter plates: -> Problems: - O 2 limitation (aeration) -> faster shaking -> contamination - cross-contamination - evaporation -> close with membranes - sampling (small volumne -> only micro analytical methods + stop shaking disturbs the respiration)

Minibioreactors Test tubes: -> useful for developing inoculums -> screening -> volumne 2 -25 ml (20% filled with medium) -> simple and low costs -> O 2 transfer rate low -> usually no online monitoring (p. H and O 2) -> interruption of shaking during sampling

Minibioreactors Stirred Systems: -> homogeneous environment -> sampling, online monitoring, control possible without disturbance of culture -> increased mixing (stirring) + mass transfer (gassing rate)

Minibioreactors Stirred Systems – Stirred Minibioreactor -> T, p. H, dissolved O 2 can be controlled -> Volumne from 50 ml – 300 ml -> small medium requirenments -> low costs (isotope labeling) -> good for research -> good for continous cultivation -> Limitation: - system expensive due to minimization (control elements) - not good for high-throughput applications

Minibioreactors Stirred Systems – Spinner flask -> designed to grow animal cells -> high price instrument -> shaft containing a magnet for stirring -> shearing forces can be too big -> side arms for inoculation, sampling, medium inlet, outlet, ph probe, air (O 2) inlet, air outlet -> continous reading of p. H and O 2 possible

Minibioreactors Special Devices – Cuvette based microreactor -> optical sensors (measuring online: p. H, OD, O 2) -> disposable -> volumne 4 ml -> air inlet/outlet -> magnet bead -> stirring -> similar performance as a 1 L batch reactor

Minibioreactors Special Devices – Miniature bioreactor with integrated membrane for MS measurement: -> custom made -> expensive -> a few ml -> online analysis of H 2, CH 4, O 2, N 2, CO 2, and many other products, substrate, . . . -> used to follow respiratory dynamics of culture (isotope labeling) -> stirred vessel with control of T, O 2, p. H -> MS measurements within a few seconds to minutes -> continous detection -> fast kinetic measurements, metabolic studies

Minibioreactors Special Devices – Microbioreactor: -> Vessel 5 mm diameter round chamber -> Really small working volumne -> 5 μl -> integrated optical sensors for OD, O 2, p. H -> made out of polydimethylsiloxane (PDMS) -> transparent (optical measurements), permeable for gases (aeration) -> E. coli sucessfully grown -> batch and continous cultures possible -> similar profile as 500 ml batch reactors -> limitation: sampling (small volumne -> analytical methods !!!)

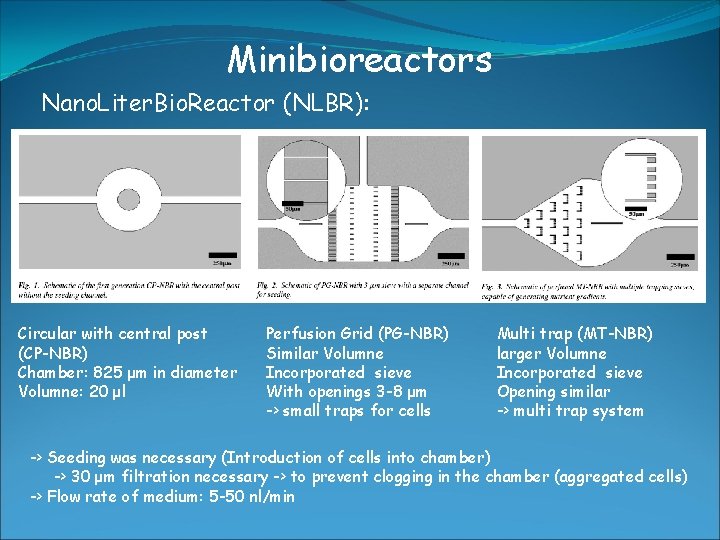
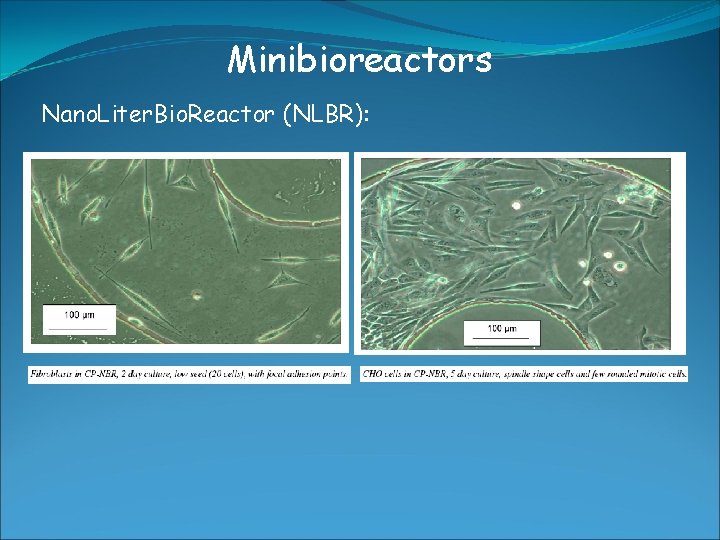
Minibioreactors Nano. Liter. Bio. Reactor (NLBR): -> used for growing up to several 100 mammalien cells -> culture volumne around 20 μl -> online control of O 2, p. H, T -> culture chamber with inlet/outlet ports (microfluidic systems) -> manufactured by soft-lithography techniques -> made out of polydimethylsiloxane (PDMS) -> transparent (optical measurements), permeable for gases (aeration) -> direct monitoring of culture condition -> PDMS is transparent -> flourescence microscope -> limitation: batch culture very difficult-> too small volumne -> suffers from nutrient limitation -> But in principle system allows -> batch, fed-batch, continous

Minibioreactors Nano. Liter. Bio. Reactor (NLBR): Circular with central post (CP-NBR) Chamber: 825 μm in diameter Volumne: 20 μl Perfusion Grid (PG-NBR) Similar Volumne Incorporated sieve With openings 3 -8 μm -> small traps for cells Multi trap (MT-NBR) larger Volumne Incorporated sieve Opening similar -> multi trap system -> Seeding was necessary (Introduction of cells into chamber) -> 30 μm filtration necessary -> to prevent clogging in the chamber (aggregated cells) -> Flow rate of medium: 5 -50 nl/min

Minibioreactors Nano. Liter. Bio. Reactor (NLBR):

Minibioreactors Nano. Liter. Bio. Reactor (NLBR):


Minibioreactors Why do we want micro-and nano reactors? Applications in: - Molecular biology - Biochemistry - Cell biology - Medical devices - Biosensors -> with the aim to look at single cells !!!

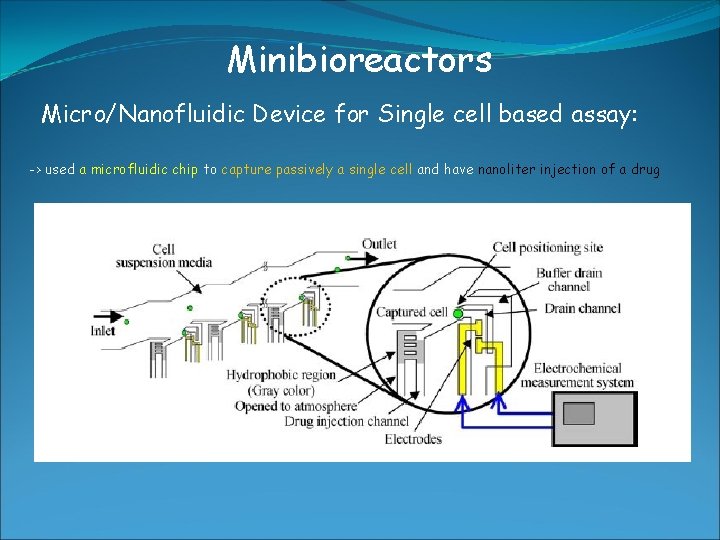
Minibioreactors Micro/Nanofluidic Device for Single cell based assay: -> used a microfluidic chip to capture passively a single cell and have nanoliter injection of a drug

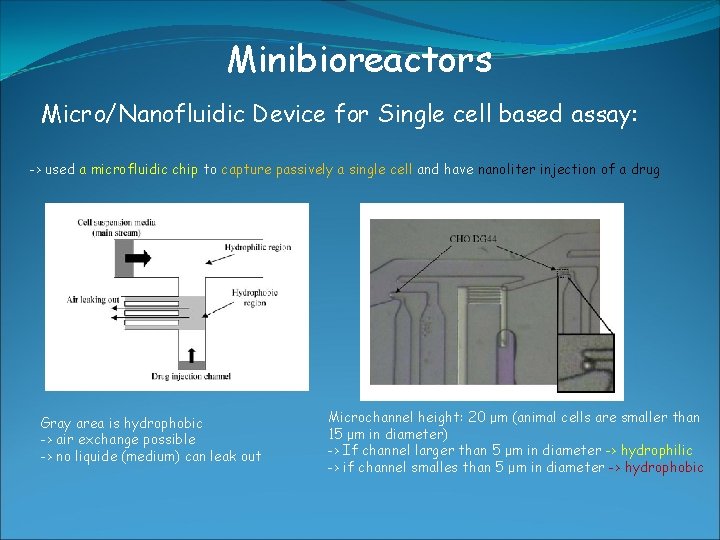
Minibioreactors Micro/Nanofluidic Device for Single cell based assay: -> used a microfluidic chip to capture passively a single cell and have nanoliter injection of a drug Gray area is hydrophobic -> air exchange possible -> no liquide (medium) can leak out Microchannel height: 20 μm (animal cells are smaller than 15 μm in diameter) -> If channel larger than 5 μm in diameter -> hydrophilic -> if channel smalles than 5 μm in diameter -> hydrophobic


Class Exercise Problem 6. 17 E. coli is cultivated in continuous culture under aerobic conditions with glucose limitation. When the system is operated at D= 0. 2 hr-1, determine the effluent glucose and biomass concentrations assuming Monod kinetics (S 0 = 5 g/l, mm= 0. 25 hr-1 , KS = 100 mg/L, Y x/s = 0. 4 g/g)



Class Exercise – 9. 4 Penicillin is produced in a fed-batch culture with the intermittent addition of glucose solution to the culture medium. The initial culture volume at quasisteady state is V 0= 500 L, and the glucose containing nutrient solution is added with a flow rate of F = 50 L/h. X 0 = 20 g/L, S 0 = 300 g/L, mm = 0. 2 h-1, Ks = 0. 5 g/L and Y x/s= 0. 3 g/g Determine culture volume at t = 10 h Determine concentration of glucose at t = 10 h Determine the concentration and total amount of cells at t = 10 h If qp = 0. 05 g product. g cells h and P 0 = 0. 1 g/L, determine the product concentration at t = 10 h