Reaction Bottle Use a graduated cylinder to measure

Reaction Bottle: Use a graduated cylinder to measure 100 ml of water and 200 ml of ethanol. What is the ratio of water to ethanol? Why do you think water and ethanol are used in the solvent? Use an auto pippette to spike the solvent with 1. 60 ml of Te. CB.

Use an autopipette to take 2 -2 ml samples of the spiked solvent.

These samples will be spiked with pentene and capped tightly. Why do you think pentene is used? These samples will be placed on a shaker table for 1 hour and then will sit for 1 hour.

Use an analytic balance to mass 0. 20 g of Palladium (Pd) and 0. 90 g of Rhodium (Rh). Add the catalyst to the reaction bottle. Place the reaction bottle on the hydrogenation apparatus for 8 hours with Nitrogen gas. Why is Nitrogen gas used instead of Hydrogen gas?

Remove reaction bottle from hydrogenation apparatus. Using a syringe filter, take 15 ml of solvent from the reaction bottle. Remove the filter and expel the solvent into a beaker.

Use an autopippete to take 2 -2 ml samples of the filtered solvent. The samples will be spiked with pentene and capped tightly. These samples will be placed on a shaker table for 1 hour and then will sit for 1 hour.

The initial and final concentrations of Te. CB can be determined by using a gas chromatograph with an electron capture detector. Research why a gas chromatograph is used to determine the concentration of Te. CB.
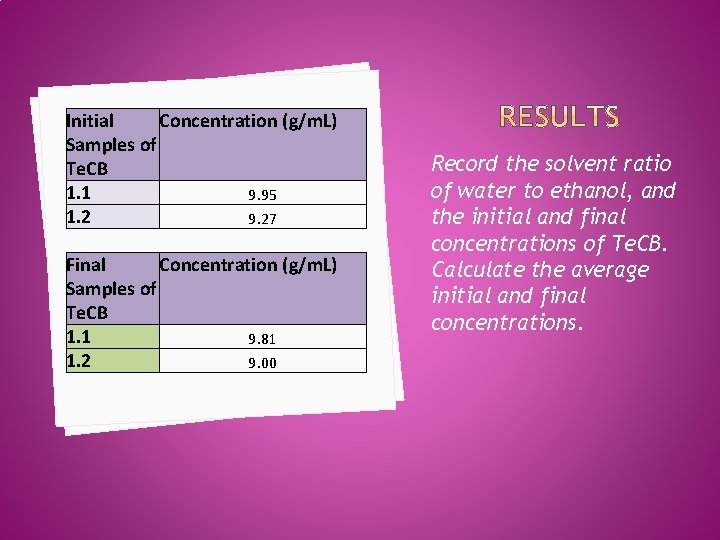
Initial Concentration (g/m. L) Samples of Te. CB 1. 1 9. 95 1. 2 9. 27 Final Concentration (g/m. L) Samples of Te. CB 1. 1 9. 81 1. 2 9. 00 Record the solvent ratio of water to ethanol, and the initial and final concentrations of Te. CB. Calculate the average initial and final concentrations.

Initial Concentration (g/m. L) Samples of Te. CB 2. 1 7. 55 2. 2 8. 55 Final Concentration (g/m. L) Samples of Te. CB 2. 1 4. 39 2. 2 6. 19 You repeat the experiment using 200 ml of water and 100 ml of ethanol. Calculate the new solvent ratio of water to ethanol. Record the solvent ratio of water to ethanol, and the initial and final concentrations of Te. CB. Calculate the average initial and final concentrations.

Complete the Lab Notes. They must be turned in with your scientific report.
- Slides: 10