RATES REACTION Temperature Surface Area Stirring Reaction Rate









- Slides: 13

RATES REACTION

Temperature Surface Area Stirring Reaction Rate Factors Concentration Pressure Catalysts

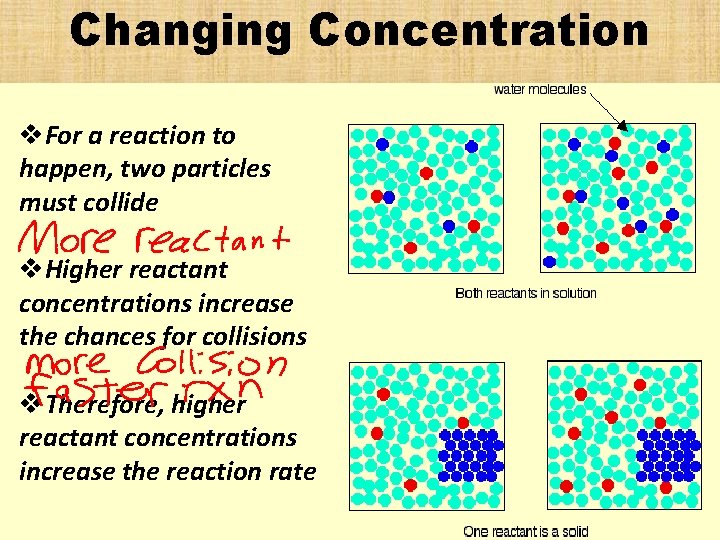
Changing Concentration v. For a reaction to happen, two particles must collide v. Higher reactant concentrations increase the chances for collisions v. Therefore, higher reactant concentrations increase the reaction rate

Demonstrate: Will the following reaction happen faster in 2. 0 M Hydrochloric acid (strong), or 0. 5 M Hydrochloric acid (weak)? Zn + 2 HCl Zn. Cl 2 +H 2

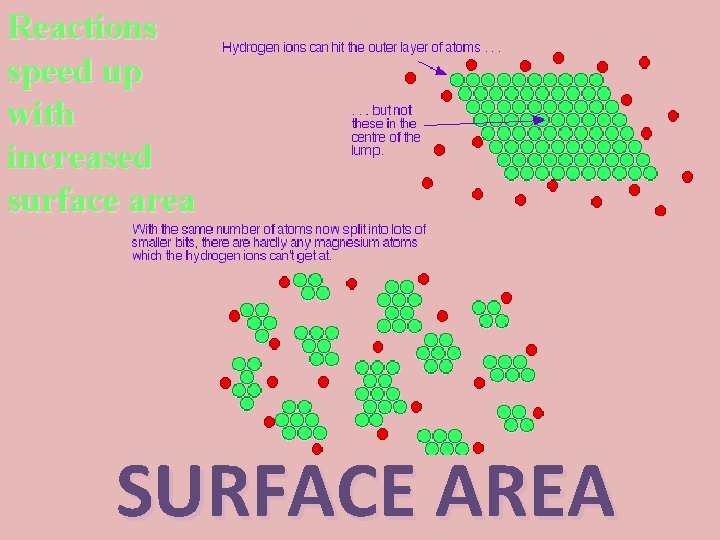
Reactions speed up with increased surface area SURFACE AREA

Demonstrate: Will sugar dissolve faster in water as a powder or a cube?

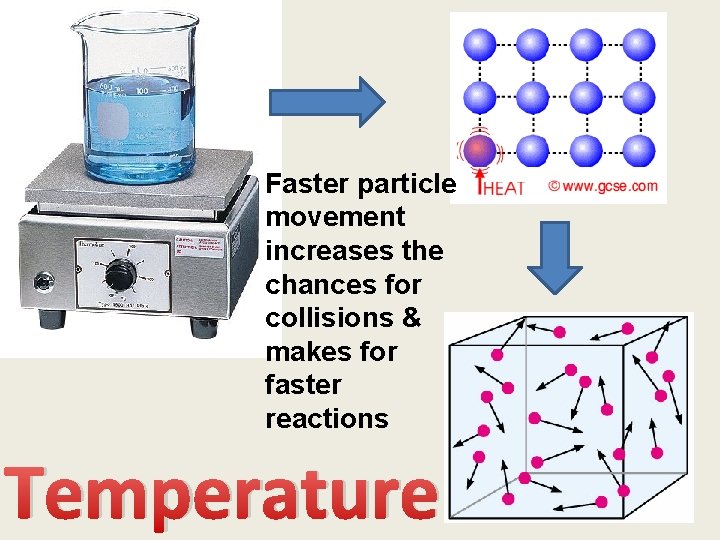
Faster particle movement increases the chances for collisions & makes for faster reactions Temperature

The energy needed to make a reaction happen is called the activation energy. A catalyst lowers the amount of energy needed so that a reaction can happen easier. A catalyst is about energy; it doesn't have to be another molecule.

Demonstrate: Decomposition of hydrogen peroxide using manganese (IV) oxide as a catalyst §Place 20 m. L of hydrogen peroxide in a large test tube §Place 20 m. L of hydrogen peroxide in a 2 nd test tube §Add a small amount of manganese (IV) oxide to the second test tube §Gently heat both test tubes in a water bath on a hot plate

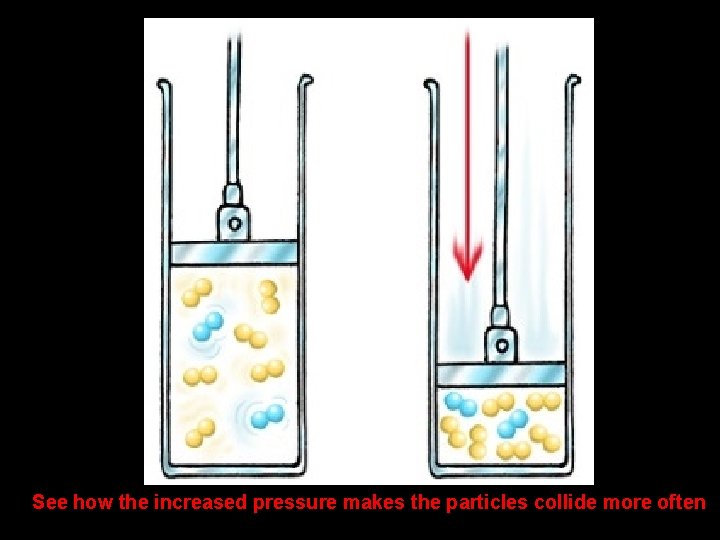
PRESSURE Pressure = faster reaction • Increasing pressure gives the particles less room to move • Less room to move increases the chances that particles will collide speeding up the reaction Pressure = slower reaction • Decreasing pressure gives the particles more room to move • More room to move decreases the chances that particles will collide slowing down the reaction

See how the increased pressure makes the particles collide more often

v. Helps particles move faster v. Increases the rate of particle collisions v. Makes the reaction happen faster STIRRING

Good thing we have learned a bit more about chemistry.