RATES OF REACTION Boardworks Ltd 2003 Rates of

RATES OF REACTION © Boardworks Ltd 2003

Rates of reaction Reactions can be very fast, like fireworks or explosives, but they can also be very slow – such as an apple turning brown. © Boardworks Ltd 2003

Rates of reaction Chemical reactions occur when particles of reactant collide with enough energy to react. © Boardworks Ltd 2003
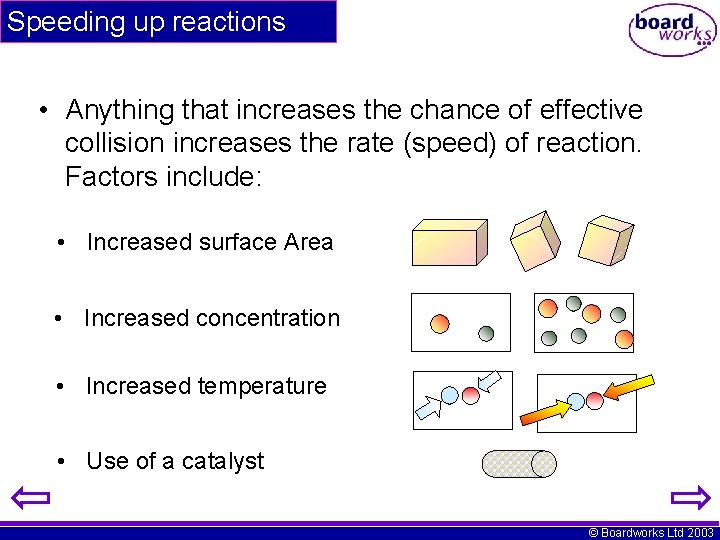
Speeding up reactions • Anything that increases the chance of effective collision increases the rate (speed) of reaction. Factors include: • Increased surface Area • Increased concentration • Increased temperature • Use of a catalyst © Boardworks Ltd 2003
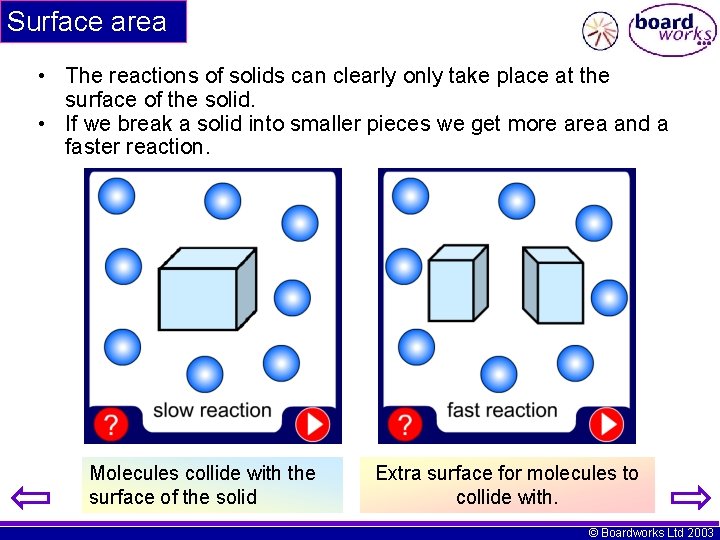
Surface area • The reactions of solids can clearly only take place at the surface of the solid. • If we break a solid into smaller pieces we get more area and a faster reaction. Molecules collide with the surface of the solid Extra surface for molecules to collide with. © Boardworks Ltd 2003
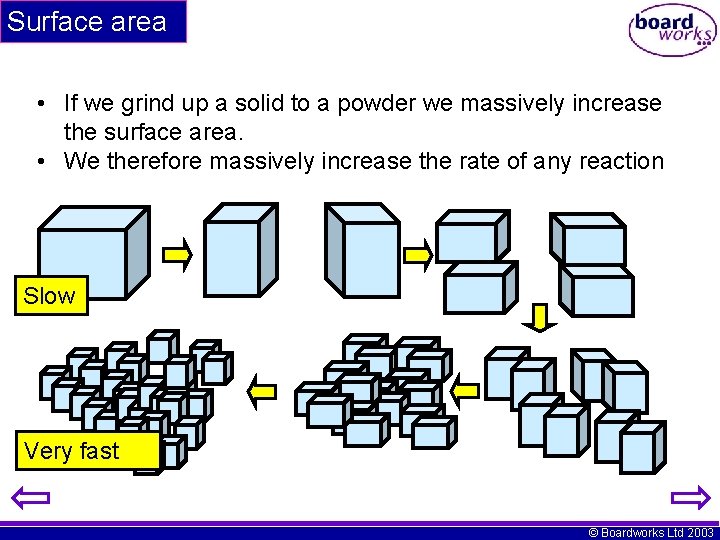
Surface area • If we grind up a solid to a powder we massively increase the surface area. • We therefore massively increase the rate of any reaction Slow Very fast © Boardworks Ltd 2003

An indigestion tablet fizzes in water – but fizzes much faster if it is crushed. Which glass has the crushed tablet? d oli S A Cru she d B © Boardworks Ltd 2003

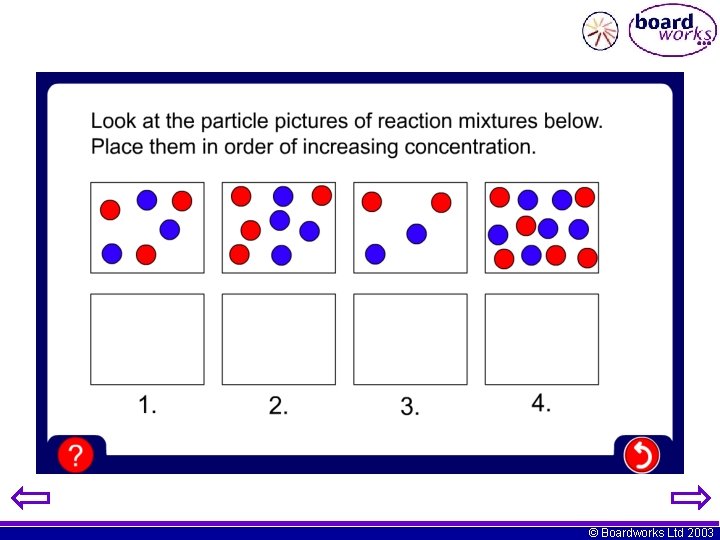
Concentration • Reactions in solution involve dissolved paticles that must collide before reaction is possible. • The more crowded (concentrated) the solution, the faster the reaction. Collisions infrequent Collisions frequent © Boardworks Ltd 2003
© Boardworks Ltd 2003

Pressure • Reactions involving gases are affected by the pressure of the gases present. • If we cover one end of a bicycle pump and push in the plunger we increase the pressure. Low pressure High pressure • What we are doing is squeezing the gas molecules closer together or making them more concentrated. • And so - pressure speeds up gas reactions © Boardworks Ltd 2003
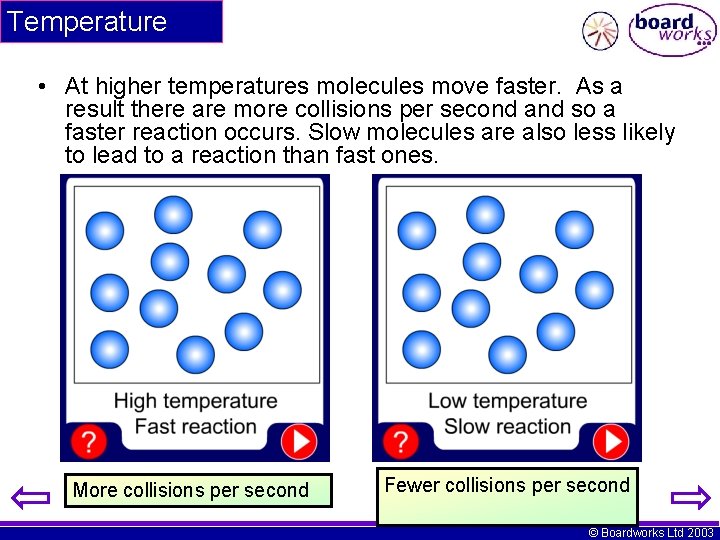
Temperature • At higher temperatures molecules move faster. As a result there are more collisions per second and so a faster reaction occurs. Slow molecules are also less likely to lead to a reaction than fast ones. More collisions per second Fewer collisions per second © Boardworks Ltd 2003

• Food spoils because of chemical reactions that occur. • Why does food remain good for so much longer if it is kept in a freezer? The reactions that cause the food to go off will be slower because there will be fewer and “softer” collisions between molecules at a reduced temperature. © Boardworks Ltd 2003

• Before microwave ovens were common many people used pressure cookers. • It was a pan that stopped the water boiling until it reached about 115 o. C. • How would this help cooking? The molecules move faster and collide more often and with more energy. Cooking times were greatly reduced. © Boardworks Ltd 2003

Catalysts and rate of reaction For chemical reactions to occur: • Existing bonds have to begin breaking so that new ones can be formed. • The molecules have to collide in such a way that the reacting parts of the molecules are brought together. Catalysts can help with either or both of these processes. A catalyst is a substance that speeds up a reaction without getting used up in the process. © Boardworks Ltd 2003

Measuring reaction rates Rate implies we are measuring how things change over a period of time. To measure the rate of a reaction we have to track the manner in which the amount of product (or reactant) changes over time. Rate of gas formation can be measured using a syringe. For a reaction in which sulfur is precipitated we can time how long the solution takes to go cloudy. © Boardworks Ltd 2003
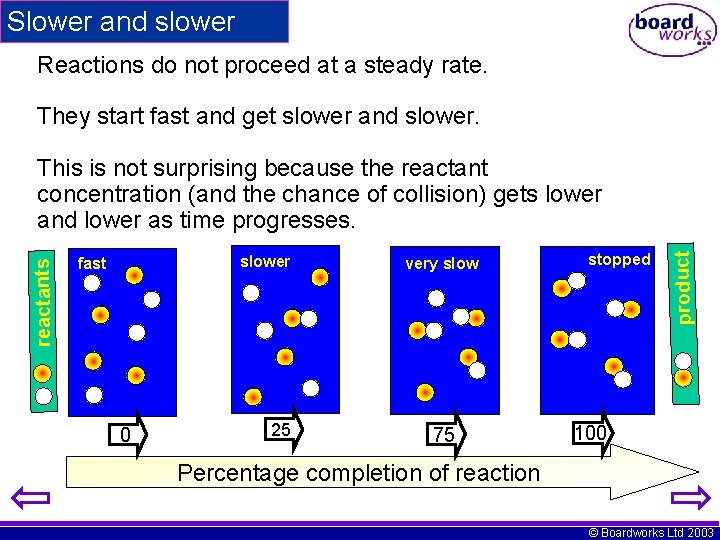
Slower and slower Reactions do not proceed at a steady rate. They start fast and get slower and slower fast 0 25 very slow 75 stopped product reactants This is not surprising because the reactant concentration (and the chance of collision) gets lower and lower as time progresses. 100 Percentage completion of reaction © Boardworks Ltd 2003
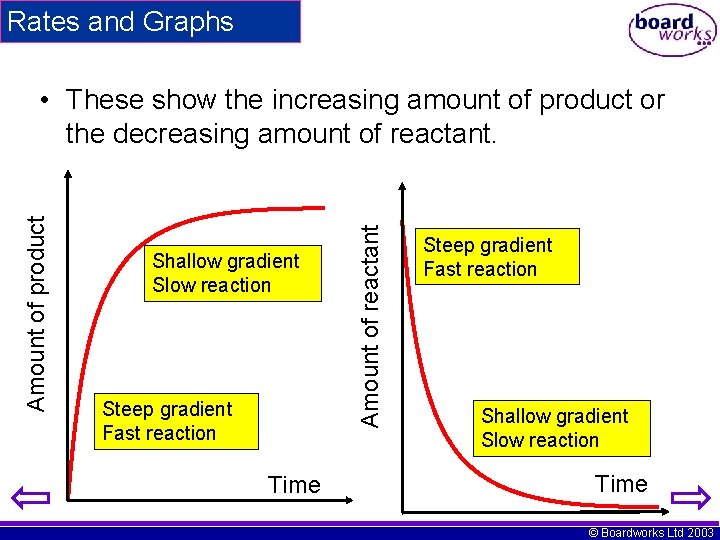
Rates and Graphs Shallow gradient Slow reaction Steep gradient Fast reaction Time Amount of reactant Amount of product • These show the increasing amount of product or the decreasing amount of reactant. Steep gradient Fast reaction Shallow gradient Slow reaction Time © Boardworks Ltd 2003

Rate graphs and reactant concentrations Amount of product All product Mix of reactant And product Reactant Concentration falls reactants Rate of Reaction falls product Gradient of graph decreases All reactant Time © Boardworks Ltd 2003

© Boardworks Ltd 2003
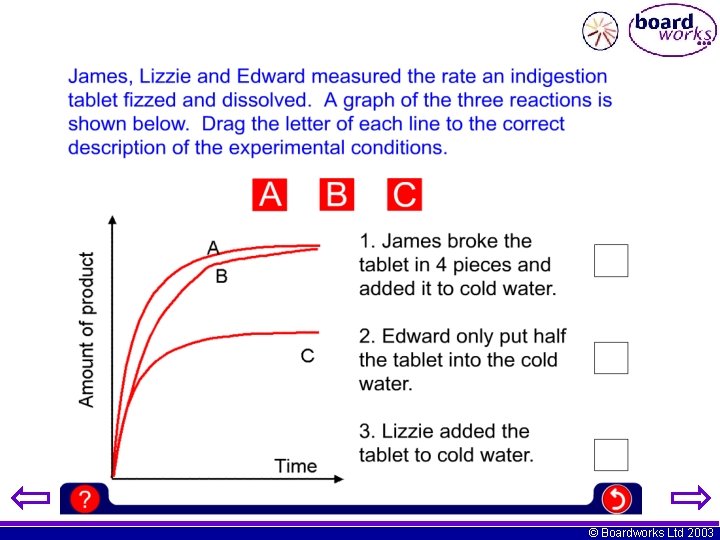
© Boardworks Ltd 2003

Some Reaction Rates Experiments The following slides describe the four chemical reactions that are commonly used as examples. © Boardworks Ltd 2003
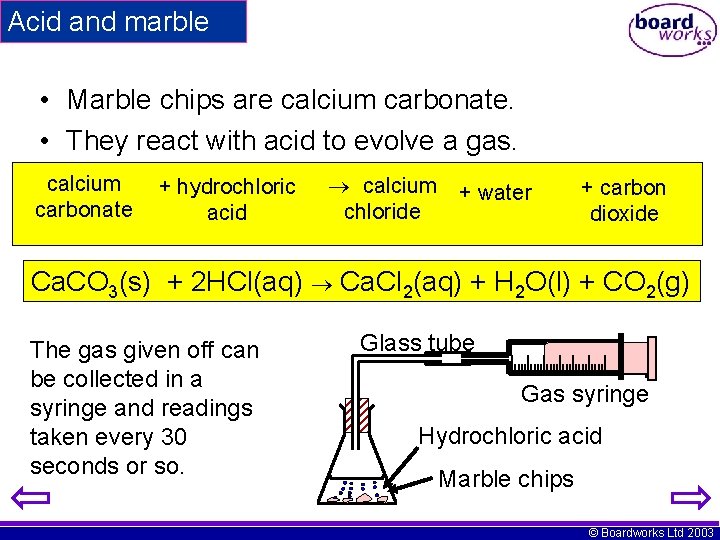
Acid and marble • Marble chips are calcium carbonate. • They react with acid to evolve a gas. calcium carbonate + hydrochloric acid calcium + water chloride + carbon dioxide Ca. CO 3(s) + 2 HCl(aq) Ca. Cl 2(aq) + H 2 O(l) + CO 2(g) The gas given off can be collected in a syringe and readings taken every 30 seconds or so. Glass tube Gas syringe Hydrochloric acid Marble chips © Boardworks Ltd 2003

Acid and marble 1. Measure the agreed mass of marble chips 2. Set up the syringe, flask and connector 3. Measure the acid / water. 4. Add the marble chips and quickly insert the bung and start stop clock. 5. Take syringe readings at 30 second intervals. Time 0 s Reading 0 cm 3 © Boardworks Ltd 2003
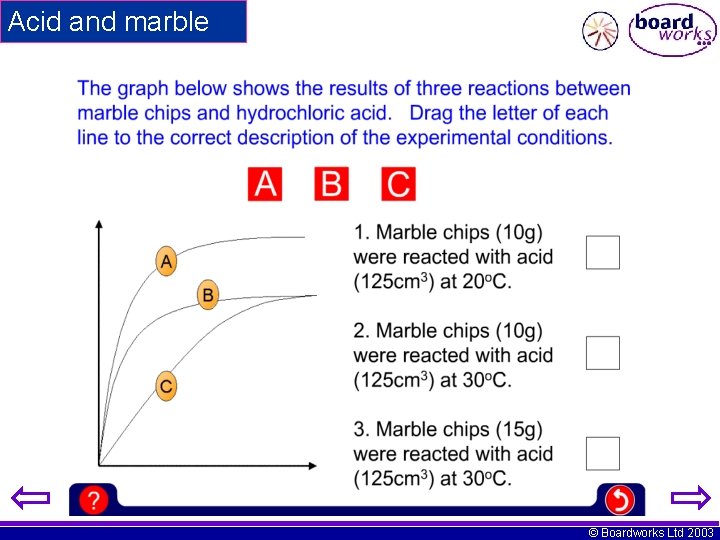
Acid and marble © Boardworks Ltd 2003
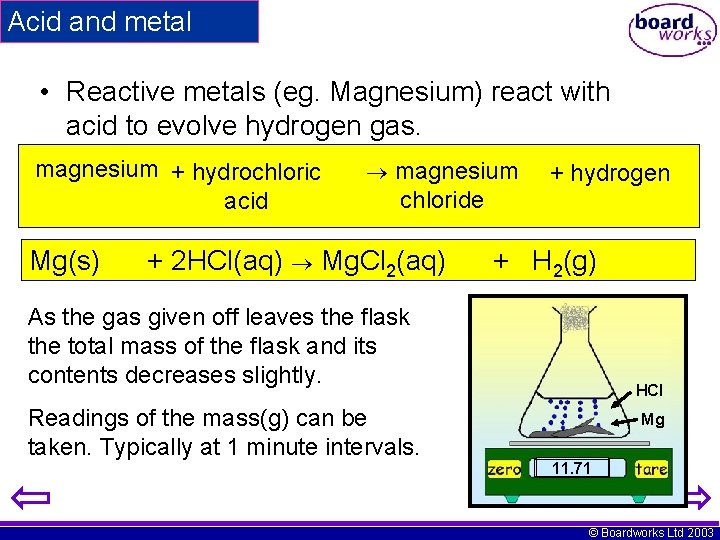
Acid and metal • Reactive metals (eg. Magnesium) react with acid to evolve hydrogen gas. magnesium + hydrochloric acid Mg(s) magnesium chloride + 2 HCl(aq) Mg. Cl 2(aq) + hydrogen + H 2(g) As the gas given off leaves the flask the total mass of the flask and its contents decreases slightly. Readings of the mass(g) can be taken. Typically at 1 minute intervals. HCl Mg 11. 73 11. 71 11. 80 11. 72 11. 74 11. 77 © Boardworks Ltd 2003

Acid and metal 1. Measure the agreed volume of acid / water into the conical flask. 2. Have a loose plug of cotton wool to prevent “spitting” of droplets of liquid. 3. Have a piece of magnesium of known mass ready. 4. Add the magnesium, place the cotton wool in the neck and start taking mass readings immediately. Cotton wool HCl Mg 11. 73 11. 71 11. 80 11. 72 11. 74 11. 77 Time 0 s Reading 0 cm 3 60 120 © Boardworks Ltd 2003

Acid and metal © Boardworks Ltd 2003

Decomposition of Hydrogen Peroxide • Hydrogen peroxide decomposes into water and oxygen. Hydrogen peroxide 2 H 2 O 2(aq) water 2 H 2 O(l) + oxygen + O 2(g) Oxygen gas is given off and can be measured using a gas syringe or a balance. The reaction is catalysed by a wide range of solids. Remember the catalyst NEVER produces more product - just quicker © Boardworks Ltd 2003

© Boardworks Ltd 2003

Acid and Sodium Thiosulphate • In this reaction sulphur is precipitated which makes the solution turn cloudy. + hydrochloric sodium + water + sulphur Sodium acid thiosulphate chloride dioxide Na 2 S 2 O 3(aq) + 2 HCl(aq) 2 Na. Cl(aq) + H 2 O(l) + SO 2(g) + S(s) The effect of changing conditions such as temperature or concentrations can be studied by measuring how long it takes to produce enough sulphur to make the solution opaque (non see-through). © Boardworks Ltd 2003

1. 2. 3. 4. 5. Measure the agreed volume of thiosulphate / water into the conical flask. Prepare a piece of paper with a cross drawn on it. Measure the required volume of acid in a measuring cylinder. Add the acid to the flask, start the clock, swirl the flask. Look down through the flask until the cross disappears. Note the time. Look down here © Boardworks Ltd 2003
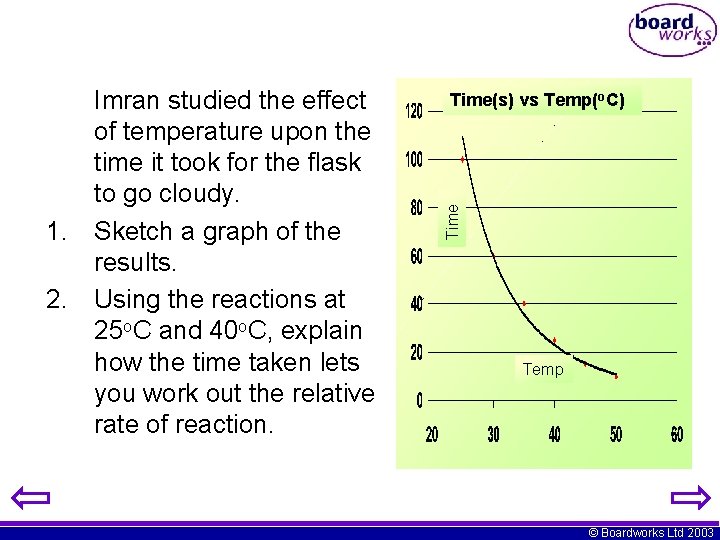
Time(s) vs Temp(o. C) Temp (o. C) Time taken (s) 25 100 30 60 35 40 40 25 Time Imran studied the effect of temperature upon the time it took for the flask to go cloudy. 1. Sketch a graph of the results. 2. Using the reactions at 25 o. C and 40 o. C, explain how the time taken lets you work out the relative rate of reaction. 45 50 Temp 15 10 © Boardworks Ltd 2003

Some Rates Questions © Boardworks Ltd 2003
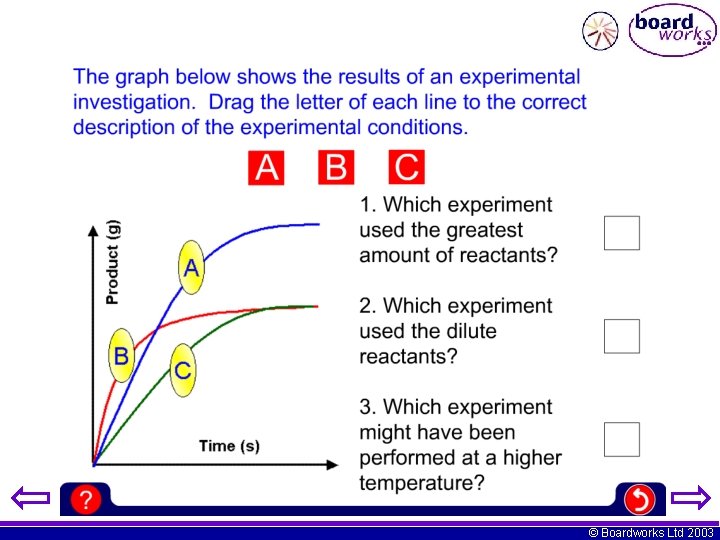
© Boardworks Ltd 2003

A pupil performed an investigation into the rate of reaction between a metal and an acid. The results below where obtained. i) Plot a graph of gas volume (y-axis) against time (x-axis) ii) When was the rate of reaction fastest? iii) Use the graph to find the volume of gas produced after 35 seconds. iv) Use the graph to tell after how long the reaction stopped. v) On the graph sketch a line showing the experiment repeated at a higher temperature. © Boardworks Ltd 2003
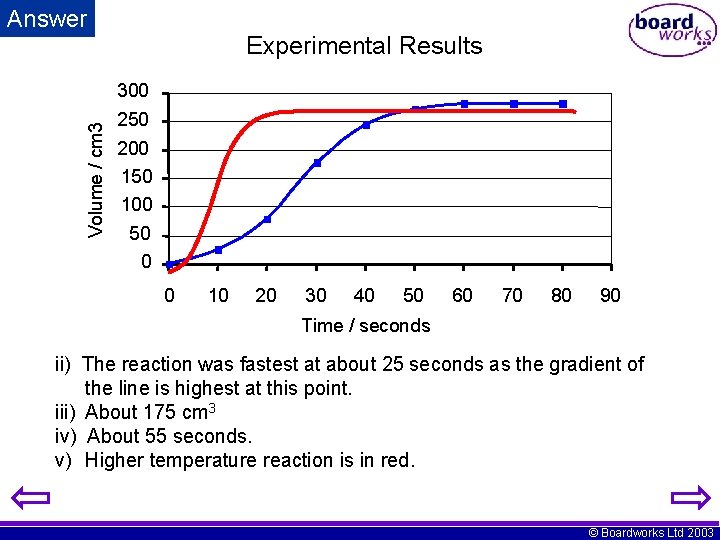
Volume / cm 3 Answer Experimental Results 300 250 200 150 100 50 0 0 10 20 30 40 50 60 70 80 90 Time / seconds ii) The reaction was fastest at about 25 seconds as the gradient of the line is highest at this point. iii) About 175 cm 3 iv) About 55 seconds. v) Higher temperature reaction is in red. © Boardworks Ltd 2003

A flask was connected to a gas syringe by a glass delivery tube. 30 cm 3 of water and 0. 5 g of manganese dioxide were added to the flask. Then 5 cm 3 of hydrogen peroxide was added and the stopper quickly fitted. Readings of the volume of gas produced were taken every 10 seconds. i) Plot a graph of volume of gas (y-axis) against time (x-axis). Label this curve A. ii) Without emptying the flask another 10 cm 3 of water and a further 5 cm 3 of hydrogen peroxide were added. Sketch the shape of the second experiment and label it B. © Boardworks Ltd 2003
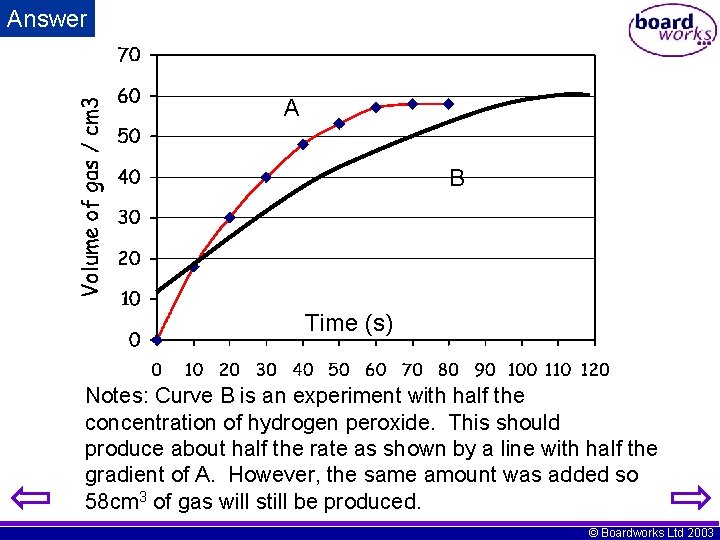
Answer A B Time (s) Notes: Curve B is an experiment with half the concentration of hydrogen peroxide. This should produce about half the rate as shown by a line with half the gradient of A. However, the same amount was added so 58 cm 3 of gas will still be produced. © Boardworks Ltd 2003
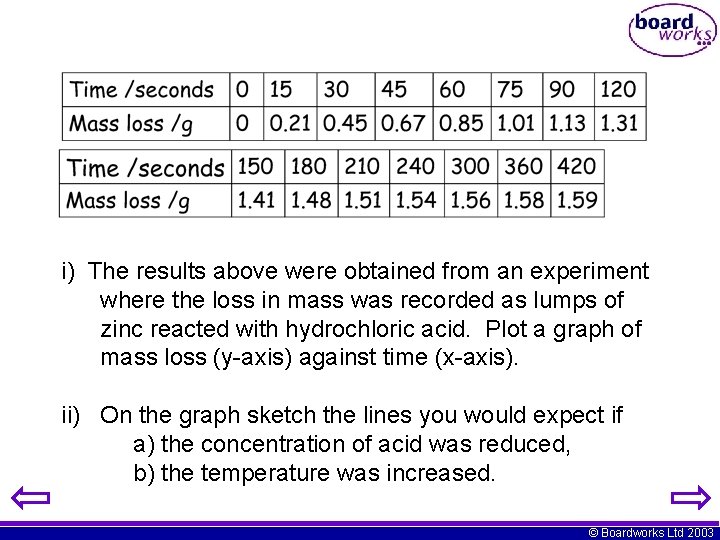
i) The results above were obtained from an experiment where the loss in mass was recorded as lumps of zinc reacted with hydrochloric acid. Plot a graph of mass loss (y-axis) against time (x-axis). ii) On the graph sketch the lines you would expect if a) the concentration of acid was reduced, b) the temperature was increased. © Boardworks Ltd 2003
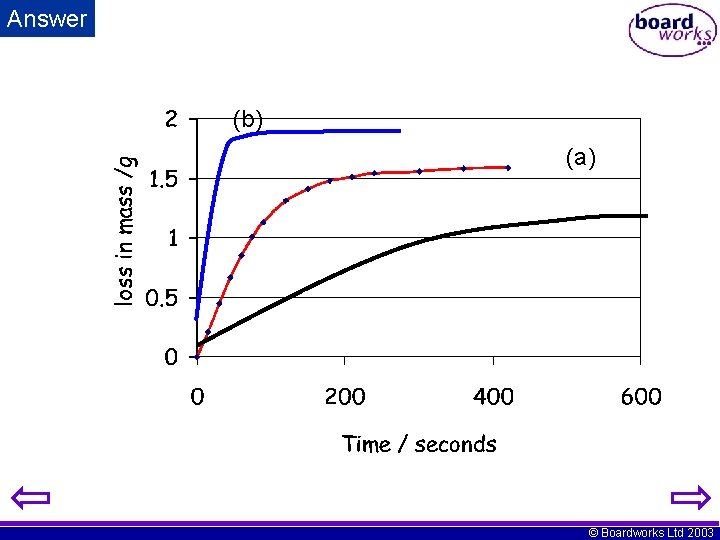
Answer (b) (a) © Boardworks Ltd 2003

Which of these would speed up the rate at which magnesium dissolves in acid? A. B. C. D. Cool the acid. Cut up the magnesium. Add water. Coat the magnesium in oil. © Boardworks Ltd 2003

Why does breaking up solids increase the rate of reaction? A. B. C. D. Makes more solid. Creates more energy. Increases surface area. Increases the concentration. © Boardworks Ltd 2003

Why does temperature increase the rate of reaction? A. B. C. D. Acts as a catalyst. Increases the concentration. Increases number of molecules. Makes collisions more frequent and harder. © Boardworks Ltd 2003

Why does a catalyst increase the rate of reaction? A. Provides a route with a lower activation energy. B. Helps provide energy for the reaction. C. Increases the speed of reactant molecules. D. Reduces the number of molecular collisions. © Boardworks Ltd 2003

Why do most reactions start fast and get slower and slower? A. They run out of energy. B. They run out of catalyst. C. The concentration of reactant molecules gets less and less. D. The surface area increases. © Boardworks Ltd 2003
- Slides: 45