Rate law 1 rate a Br 2 rate

Rate law 1
![rate a [Br 2] rate = k [Br 2] rate = rate constant k= rate a [Br 2] rate = k [Br 2] rate = rate constant k=](http://slidetodoc.com/presentation_image_h2/67eb6985379e581b827bb91fea6b1695/image-2.jpg)
rate a [Br 2] rate = k [Br 2] rate = rate constant k= [Br 2] = 3. 50 x 10 -3 s-1 2

The rate law expresses the relationship of the rate of a reaction to the rate constant and the concentrations of the reactants raised to some powers. a. A + b. B c. C + d. D Rate = k [A]x[B]y Reaction is xth order in A Reaction is yth order in B Reaction is (x +y)th order overall 3

Rate Laws • Rate laws are always determined experimentally. • Reaction order is always defined in terms of reactant (not product) concentrations. • The order of a reactant is not related to the stoichiometric coefficient of the reactant in the balanced chemical equation. F 2 (g) + 2 Cl. O 2 (g) 2 FCl. O 2 (g) rate = k [F 2][Cl. O 2] 1 4

F 2 (g) + 2 Cl. O 2 (g) 2 FCl. O 2 (g) rate = k [F 2]x[Cl. O 2]y Double [F 2] with [Cl. O 2] constant Rate doubles x=1 Quadruple [Cl. O 2] with [F 2] constant rate = k [F 2][Cl. O 2] Rate quadruples y=1 5

Example The reaction of nitric oxide with hydrogen at 1280°C is From the following data collected at this temperature, determine (a) the rate law (b) the rate constant (c) the rate of the reaction when [NO] = 12. 0 × 10− 3 M and = 6. 0 × 10 − 3 M [H 2]
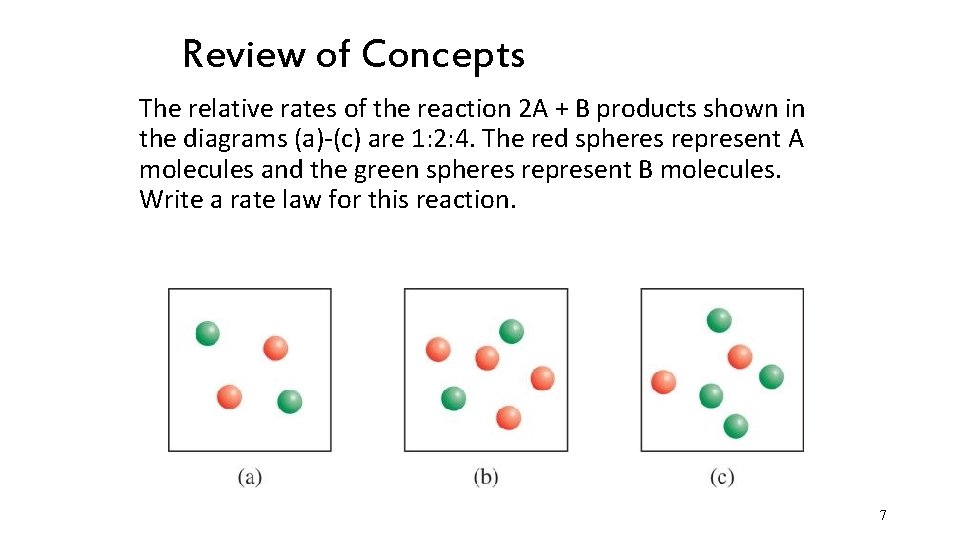
Review of Concepts The relative rates of the reaction 2 A + B products shown in the diagrams (a)-(c) are 1: 2: 4. The red spheres represent A molecules and the green spheres represent B molecules. Write a rate law for this reaction. 7

Integrated rate law 8
![First-Order Reactions A k= product D[A] rate = Dt rate M/s = = 1/s First-Order Reactions A k= product D[A] rate = Dt rate M/s = = 1/s](http://slidetodoc.com/presentation_image_h2/67eb6985379e581b827bb91fea6b1695/image-9.jpg)
First-Order Reactions A k= product D[A] rate = Dt rate M/s = = 1/s or s-1 M [A] = [A]0 e−kt rate = k [A] D[A] = k [A] Dt [A] is the concentration of A at any time t [A]0 is the concentration of A at time t=0 ln[A] = -kt + ln[A]0 9
![Second-Order Reactions A product D[A] rate = Dt rate M/s = = 1/M • Second-Order Reactions A product D[A] rate = Dt rate M/s = = 1/M •](http://slidetodoc.com/presentation_image_h2/67eb6985379e581b827bb91fea6b1695/image-10.jpg)
Second-Order Reactions A product D[A] rate = Dt rate M/s = = 1/M • s k= 2 2 M [A] 1 1 = + kt [A]0 rate = k [A]2 D[A] = k [A]2 Dt [A] is the concentration of A at any time t [A]0 is the concentration of A at time t=0 t½ = t when [A] = [A]0/2 1 t½ = k[A]0 10
![Zero-Order Reactions A product D[A] rate = Dt D[A] =k Dt rate = M/s Zero-Order Reactions A product D[A] rate = Dt D[A] =k Dt rate = M/s](http://slidetodoc.com/presentation_image_h2/67eb6985379e581b827bb91fea6b1695/image-11.jpg)
Zero-Order Reactions A product D[A] rate = Dt D[A] =k Dt rate = M/s k= 0 [A] = [A]0 - kt rate = k [A]0 = k [A] is the concentration of A at any time t [A]0 is the concentration of A at time t = 0 t½ = t when [A] = [A]0/2 [A]0 t½ = 2 k 11

Summary of the Kinetics of Zero-Order, First-Order and Second-Order Reactions Order 0 Rate Law rate = k 1 rate = k [A] 2 [A]2 rate = k Concentration-Time Equation [A] = [A]0 - kt Half-Life t½ = [A]0 2 k ln[A] = ln[A]0 - kt t½ = ln 2 k 1 1 = + kt [A]0 1 t½ = k[A]0 12

Rate Laws Summary 1. Assume rxn studied under conditions where only forward rxn is important 2. Two types of rate laws a. Differential rate law b. Integrated rate law 3. Use method of initial rates to obtain rate law 13
- Slides: 13