Rate Constant for the Reactions Between OH and

Rate Constant for the Reactions Between OH and Perfluorinated Alkenes Vladimir L. Orkin, Gregory A. Poskrebyshev, Michael J. Kurylo, J. Phys. Chem. A 115 (2011) 6568 -6574. Jeff Joens CHM 4930 May 26, 2015
Chlorofluorocarbons (CFCs) - Halogenated alkanes - Can be liquified by compression - Chemically inert - Relatively inexpensive http: //www. dynamicscience. com. au
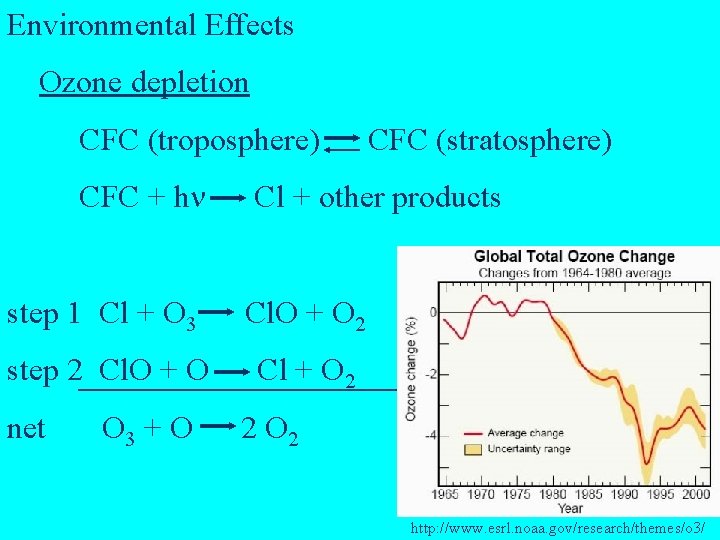
Environmental Effects Ozone depletion CFC (troposphere) CFC + h Cl + other products step 1 Cl + O 3 Cl. O + O 2 step 2 Cl. O + O Cl + O 2 net O 3 + O CFC (stratosphere) 2 O 2 http: //www. esrl. noaa. gov/research/themes/o 3/
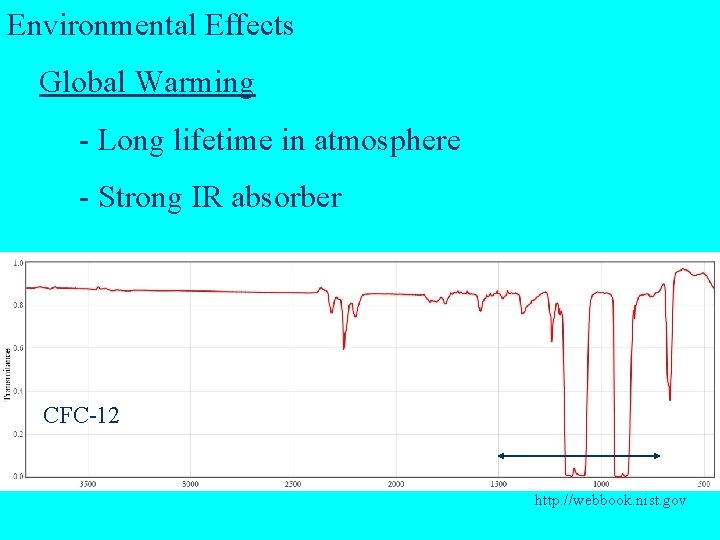
Environmental Effects Global Warming - Long lifetime in atmosphere - Strong IR absorber CFC-12 http: //webbook. nist. gov

Fluorinated Alkenes (FA) - Similar properties to CFCs - Expected to be more reactive in troposphere - Reaction in troposphere FA + OH “products” FA = CF 2=CF 2=CFCF 3 cis- , trans- CF 3 CF=CFCF 3 (CF 3)2 C=CFC 2 H 5

Flash Photolysis-Resonance Fluorescence
![Experimental Conditions Pseudo-first order ( [FA] >> [OH] ) Inlet gas mixture (argon + Experimental Conditions Pseudo-first order ( [FA] >> [OH] ) Inlet gas mixture (argon +](http://slidetodoc.com/presentation_image_h2/79b54e6fea59cc81ddbb41434ac23b1a/image-7.jpg)
Experimental Conditions Pseudo-first order ( [FA] >> [OH] ) Inlet gas mixture (argon + H 2 O) + (argon + FA) OH production H 2 O + Xe flash OH + H OH detection resonance fluorescence
![Pseudo-first Order Kinetics FA + OH “products” Rate = kbi [FA] [OH] k 1 Pseudo-first Order Kinetics FA + OH “products” Rate = kbi [FA] [OH] k 1](http://slidetodoc.com/presentation_image_h2/79b54e6fea59cc81ddbb41434ac23b1a/image-8.jpg)
Pseudo-first Order Kinetics FA + OH “products” Rate = kbi [FA] [OH] k 1 [OH] k 1 = kbi [FA]0 >> [OH] = [OH]0 exp(-k 1 t)
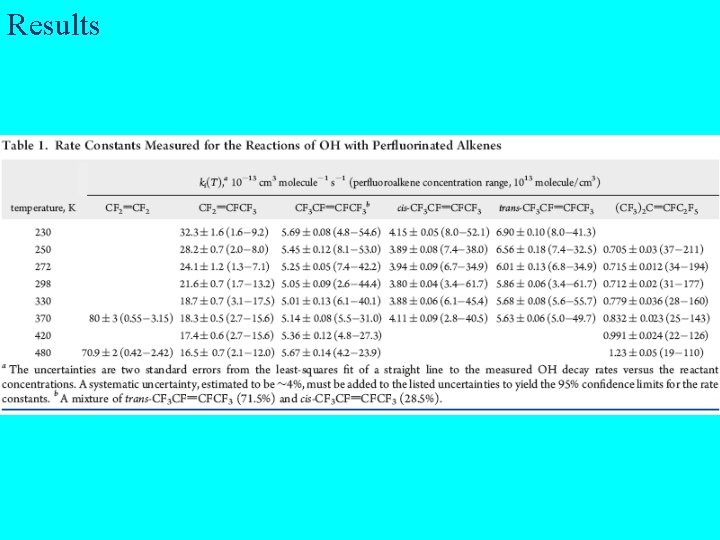
Results
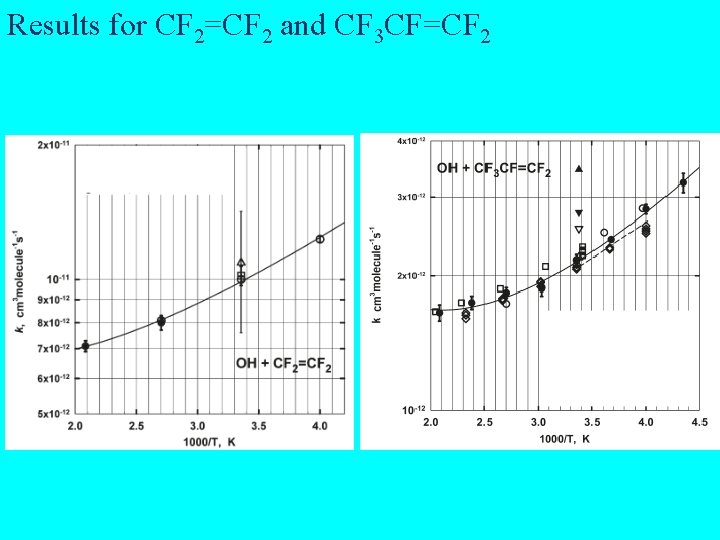
Results for CF 2=CF 2 and CF 3 CF=CF 2
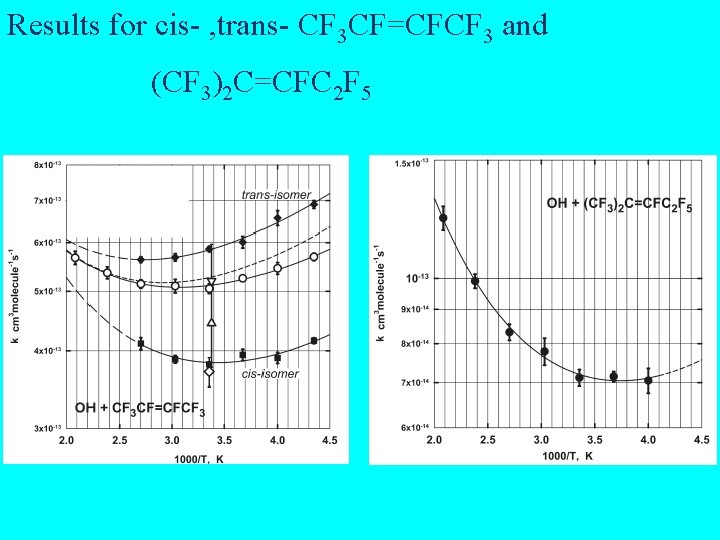
Results for cis- , trans- CF 3 CF=CFCF 3 and (CF 3)2 C=CFC 2 F 5
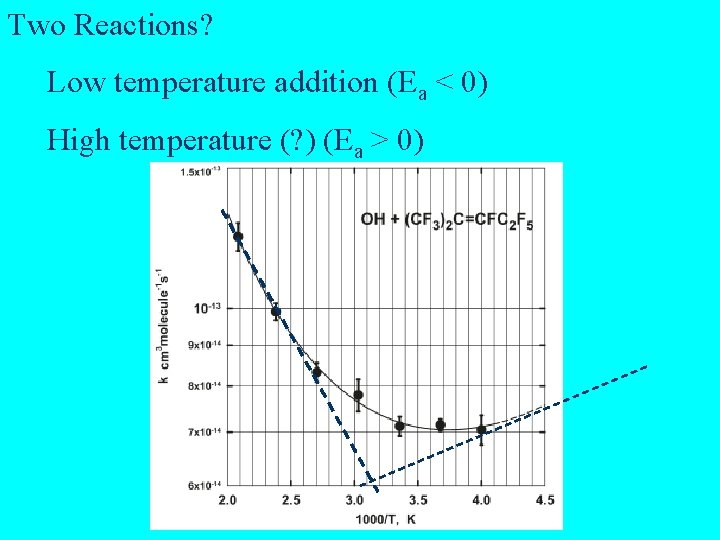
Two Reactions? Low temperature addition (Ea < 0) High temperature (? ) (Ea > 0)

Lifetimes in Troposphere Compound Lifetime CF 2=CF 2 1. 2 days CF 3 CF=CF 2 5. 3 days cis-CF 3 CF=CFCF 3 34. days trans-CF 3 CF=CFCF 3 21. days (CF 3)2 C=CFCF 2 182. days

Future Work - Extend the range of temperatures - Product analysis

Conclusions - Low molecular mass FAs are relatively harmless to the environment - Kinetic questions remain - Products of OH reaction are not known

References Finlayson-Pitts, B. J. , J. N. Pitts, Jr. Chemistry of the Upper and Lower Atmosphere. Academic Press: New York, 2000. Orkin, V, L. , G. A. Poskrebyshev, M. J. Kurylo “Rate Constants for the Reactions Between OH and Perfluorinated Alkenes” J. Phys. Chem. A 115 (2011) 6568 -6574. Velders, G. J. M. , A. R. Ravishankara, M. K. Miller, M. J. Molina, J. Alcamo, J. S. Daniel, D. W. Fahey, S. A. Montzka, S. Reimann “Preserving Montreal Protocol Climate Benefits by Limiting HFCs” Science 335 (2012) 922 -923.
- Slides: 16