Randomized Phase II Trial of Polatuzumab Vedotin Addition

Randomized Phase II Trial of Polatuzumab Vedotin Addition to Bendamustine + Rituximab in Patients With R/R DLBCL Integrating New Hematology Findings Into Practice: Independent Conference Coverage of ASH 2017, * December 9 -12, Atlanta, Georgia *CCO is an independent medical education company that provides state-of-the-art medical information to healthcare professionals through conference coverage and other educational programs. This activity is supported by educational grants from Abb. Vie; Astra. Zeneca; Celgene Corporation; Genentech; Janssen Biotech, Inc administered by Janssen Scientific Affairs, LLC; Jazz Pharmaceuticals; Novartis Pharmaceuticals Corporation; Pharmacyclics Inc; Seattle Genetics; and Takeda Oncology.

Polatuzumab Vedotin + BR in R/R DLBCL: Background § ~ 50% of pts with R/R DLBCL are ineligible for transplant due to advanced age, comorbidities, and other factors[1] – Transplantation-ineligible R/R DLBCL associated with a poor prognosis and minimal likelihood of prolonged disease control § Polatuzumab vedotin: humanized anti-CD 79 b m. Ab conjugated to MMAE[2] – CD 79 b: B-cell–specific surface antigen expressed in NHL[3] § Durable responses and acceptable safety profile reported in phase Ib/II study of PV addition to BR or BG in transplantation-ineligible pts with R/R DLBCL and FL[4] – Among pts with R/R DLBCL, ORR of 50% with PV + BR and ORR of 57% to 83% with PV + BG § Current analysis evaluated efficacy and safety of PV addition to BR vs BR alone in phase II cohorts of transplantation-ineligible R/R DLBCL pts[5] 1. Friedberg JW. Hematology Am Soc Hematol Educ Program. 2011; 2011: 498 -505. 2. Dornan D, et al. Blood. 2009; 114: 2721 -2729. 3. Polson AG, et al. Blood. 2007; 110: 616 -623. 4. Matasar M, et al. EHA 2017. Abstract S 468. 5. Sehn LH, et al. ASH 2017. Abstract 2821. Slide credit: clinicaloptions. com

Polatuzumab Vedotin + BR in R/R DLBCL: Study Design § Analysis of phase II randomized R/R DLBCL cohorts from multicenter, open-label phase Ib/II trial Six 21 -day cycles Stratified by Do. R to last tx (≤ 12 vs > 12 mos) Pts with histologically confirmed R/R DLBCL, received ≥ 1 prior tx, ECOG PS 0 -2, ineligible for SCT, no grade IIIb FL, no transformed DLBCL, no CNS lymphoma, no prior allo HSCT, no auto HSCT within 100 days (N = 80) Sehn LH, et al. ASH 2017. Abstract 2821. Polatuzumab Vedotin 1. 8 mg/kg Day 1 (each cycle) + Bendamustine 90 mg/m 2 on Days 2, 3 (cycle 1), then Days 1, 2 (each cycle) + Rituximab 375 mg/m 2 Day 1 (each cycle) (n = 40) § Primary endpoint: IRCassessed CR* by PET at PRA† § Secondary endpoints: ORR by PET/CT at PRA, best ORR § Exploratory endpoints: Do. R, PFS, EFS, OS *CR defined as PET score 1 -3 with confirmatory negative BM biopsy. †PRA at 6 -8 wks after either cycle 6, Day 1 or last dose of tx. Slide credit: clinicaloptions. com
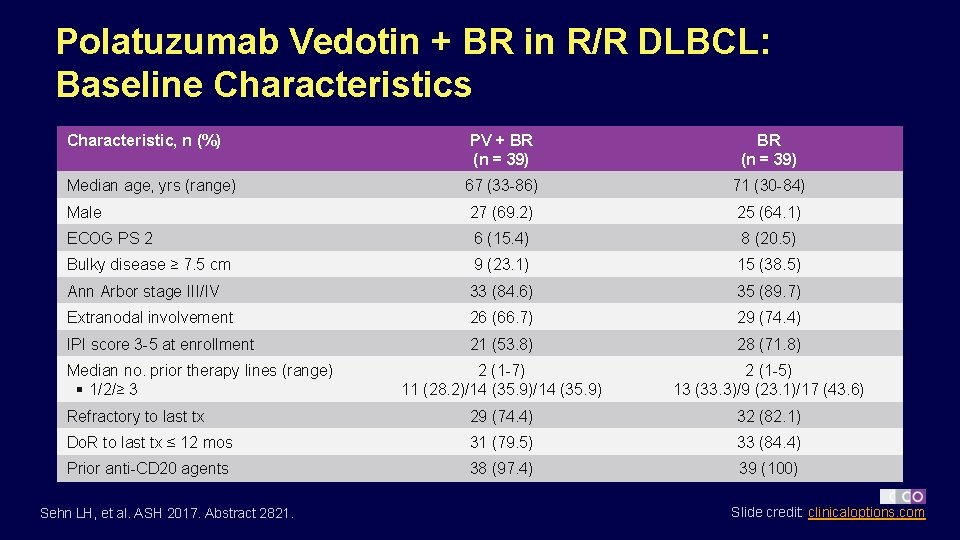
Polatuzumab Vedotin + BR in R/R DLBCL: Baseline Characteristics Characteristic, n (%) PV + BR (n = 39) 67 (33 -86) 71 (30 -84) Male 27 (69. 2) 25 (64. 1) ECOG PS 2 6 (15. 4) 8 (20. 5) Bulky disease ≥ 7. 5 cm 9 (23. 1) 15 (38. 5) Ann Arbor stage III/IV 33 (84. 6) 35 (89. 7) Extranodal involvement 26 (66. 7) 29 (74. 4) IPI score 3 -5 at enrollment 21 (53. 8) 28 (71. 8) 2 (1 -7) 11 (28. 2)/14 (35. 9) 2 (1 -5) 13 (33. 3)/9 (23. 1)/17 (43. 6) Refractory to last tx 29 (74. 4) 32 (82. 1) Do. R to last tx ≤ 12 mos 31 (79. 5) 33 (84. 4) Prior anti-CD 20 agents 38 (97. 4) 39 (100) Median age, yrs (range) Median no. prior therapy lines (range) § 1/2/≥ 3 Sehn LH, et al. ASH 2017. Abstract 2821. Slide credit: clinicaloptions. com
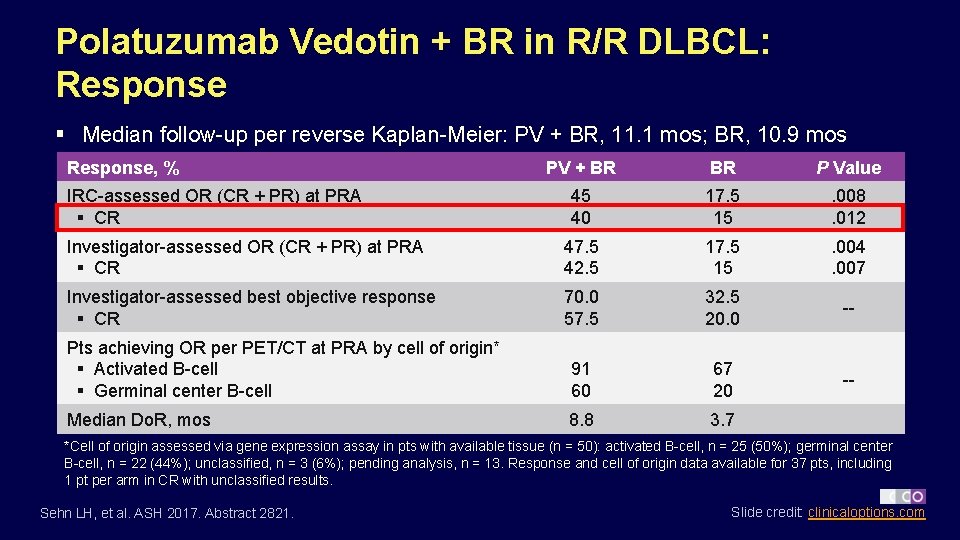
Polatuzumab Vedotin + BR in R/R DLBCL: Response § Median follow-up per reverse Kaplan-Meier: PV + BR, 11. 1 mos; BR, 10. 9 mos Response, % PV + BR BR P Value 45 40 17. 5 15 . 008. 012 Investigator-assessed OR (CR + PR) at PRA § CR 47. 5 42. 5 17. 5 15 . 004. 007 Investigator-assessed best objective response § CR 70. 0 57. 5 32. 5 20. 0 -- Pts achieving OR per PET/CT at PRA by cell of origin* § Activated B-cell § Germinal center B-cell 91 60 67 20 -- Median Do. R, mos 8. 8 3. 7 IRC-assessed OR (CR + PR) at PRA § CR *Cell of origin assessed via gene expression assay in pts with available tissue (n = 50): activated B-cell, n = 25 (50%); germinal center B-cell, n = 22 (44%); unclassified, n = 3 (6%); pending analysis, n = 13. Response and cell of origin data available for 37 pts, including 1 pt per arm in CR with unclassified results. Sehn LH, et al. ASH 2017. Abstract 2821. Slide credit: clinicaloptions. com
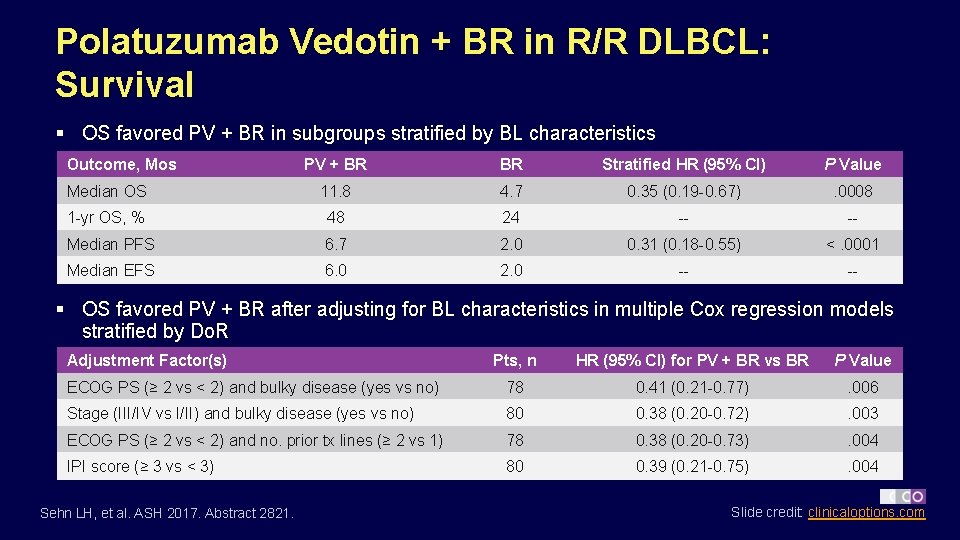
Polatuzumab Vedotin + BR in R/R DLBCL: Survival § OS favored PV + BR in subgroups stratified by BL characteristics Outcome, Mos PV + BR BR Stratified HR (95% CI) P Value Median OS 11. 8 4. 7 0. 35 (0. 19 -0. 67) . 0008 1 -yr OS, % 48 24 -- -- Median PFS 6. 7 2. 0 0. 31 (0. 18 -0. 55) <. 0001 Median EFS 6. 0 2. 0 -- -- § OS favored PV + BR after adjusting for BL characteristics in multiple Cox regression models stratified by Do. R Adjustment Factor(s) Pts, n HR (95% CI) for PV + BR vs BR P Value ECOG PS (≥ 2 vs < 2) and bulky disease (yes vs no) 78 0. 41 (0. 21 -0. 77) . 006 Stage (III/IV vs I/II) and bulky disease (yes vs no) 80 0. 38 (0. 20 -0. 72) . 003 ECOG PS (≥ 2 vs < 2) and no. prior tx lines (≥ 2 vs 1) 78 0. 38 (0. 20 -0. 73) . 004 IPI score (≥ 3 vs < 3) 80 0. 39 (0. 21 -0. 75) . 004 Sehn LH, et al. ASH 2017. Abstract 2821. Slide credit: clinicaloptions. com
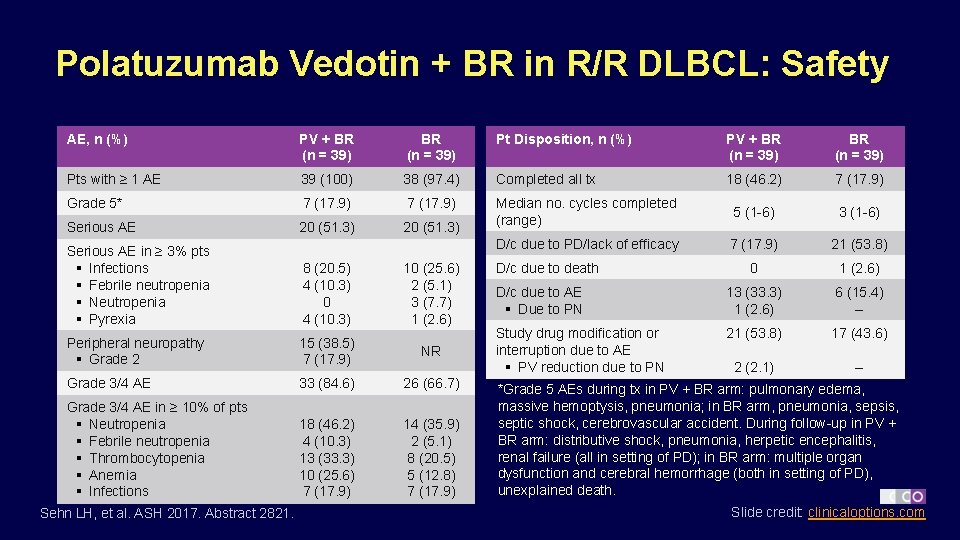
Polatuzumab Vedotin + BR in R/R DLBCL: Safety AE, n (%) PV + BR (n = 39) Pt Disposition, n (%) PV + BR (n = 39) Pts with ≥ 1 AE 39 (100) 38 (97. 4) Completed all tx 18 (46. 2) 7 (17. 9) Grade 5* 7 (17. 9) Serious AE 20 (51. 3) Median no. cycles completed (range) 5 (1 -6) 3 (1 -6) D/c due to PD/lack of efficacy 7 (17. 9) 21 (53. 8) 0 1 (2. 6) D/c due to AE § Due to PN 13 (33. 3) 1 (2. 6) 6 (15. 4) -- Study drug modification or interruption due to AE § PV reduction due to PN 21 (53. 8) 17 (43. 6) 2 (2. 1) -- Serious AE in ≥ 3% pts § Infections § Febrile neutropenia § Neutropenia § Pyrexia 8 (20. 5) 4 (10. 3) 0 4 (10. 3) 10 (25. 6) 2 (5. 1) 3 (7. 7) 1 (2. 6) Peripheral neuropathy § Grade 2 15 (38. 5) 7 (17. 9) NR Grade 3/4 AE 33 (84. 6) 26 (66. 7) Grade 3/4 AE in ≥ 10% of pts § Neutropenia § Febrile neutropenia § Thrombocytopenia § Anemia § Infections 18 (46. 2) 4 (10. 3) 13 (33. 3) 10 (25. 6) 7 (17. 9) 14 (35. 9) 2 (5. 1) 8 (20. 5) 5 (12. 8) 7 (17. 9) Sehn LH, et al. ASH 2017. Abstract 2821. D/c due to death *Grade 5 AEs during tx in PV + BR arm: pulmonary edema, massive hemoptysis, pneumonia; in BR arm, pneumonia, sepsis, septic shock, cerebrovascular accident. During follow-up in PV + BR arm: distributive shock, pneumonia, herpetic encephalitis, renal failure (all in setting of PD); in BR arm: multiple organ dysfunction and cerebral hemorrhage (both in setting of PD), unexplained death. Slide credit: clinicaloptions. com

Polatuzumab Vedotin + BR in R/R DLBCL: Conclusions § Addition of polatuzumab vedotin to BR was associated with a significantly increased response rate in R/R DLBCL pts vs BR alone – CR by IRC at PRA (primary endpoint): 40% vs 15% (P =. 012) – Preliminary biomarker data suggested that PV + BR improved response rates in pts with activated B-cell or germinal center B-cell subtypes – PV addition associated with improved Do. R, EFS, PFS, and OS – Median OS with PV + BR vs BR: 11. 8 vs 4. 7 mos (HR: 0. 35; P =. 0008) – OS benefit with PV + BR was consistent across subgroups and after adjusting for BL characteristics in multiple Cox regression models § Investigators conclude that polatuzumab vedotin addition to BR associated with clinically meaningful responses, prolonged survival, and acceptable safety profile in transplantation-ineligible R/R DLBCL pts Sehn LH, et al. ASH 2017. Abstract 2821. Slide credit: clinicaloptions. com

Go Online for More CCO Coverage of ASH 2017! Short slideset summaries of all the key data Additional CME-certified analyses with expert faculty commentary on all the key studies in: § § § Leukemias Lymphomas/CLL Myeloma Nonmalignant hematology Hot topics in hematology clinicaloptions. com/oncology
- Slides: 9